Subacute thyroiditis (De Quervain’s thyroiditis, granulomatous or viral) has been linked to numerous viruses (Coxsackievirus, influenza virus, adenovirus, ECHO virus, etc.). However, the causal virus is usually not identified, given that it does not entail changes to the treatment. A genetic predisposition and an association with certain human leukocyte antigen (HLA) haplotypes of the major histocompatibility complex have been described.1–3
This entity is more frequent in women between 30 and 50 years of age. Symptoms include neck pain, general malaise, and thyroid dysfunction; these symptoms can be mistaken for illnesses that involve pharyngeal inflammation.3
Coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), was first described in Wuhan, China, in December 2019 and rapidly spread all over the world.4 Thyroid dysfunction caused by coronaviruses other than SARS-CoV-2 has been described in patients without a prior history of thyroid disease.5,6
On June 9, 2020, a 36-year-old woman without any medical history of interest came to our clinic reporting anterior neck pain that radiated to the jaw that had been ongoing for two weeks. She also reported low-grade fever, palpitations, and fatigue. She did not present with tremor or ocular symptoms of thyrotoxicosis. A physical examination revealed that the thyroid gland was not increased in size and was sensitive to palpation as well as no adenopathy.
During the second week of April, the patient had presented with fever and head, neck, and muscle pain. These symptoms were compatible with SARS-CoV-2 infection. Her husband, a primary care physician whom she lives with, had been diagnosed with COVID-19 a few days earlier. She did not have an oropharyngeal swab for SARS-CoV-2 taken due to the limited availability of tests at that time. One month later, a serological test confirmed the diagnosis (positive IgG, negative IgM) through an immunochromatographic assay for the qualitative measurement of IgG and IgM.
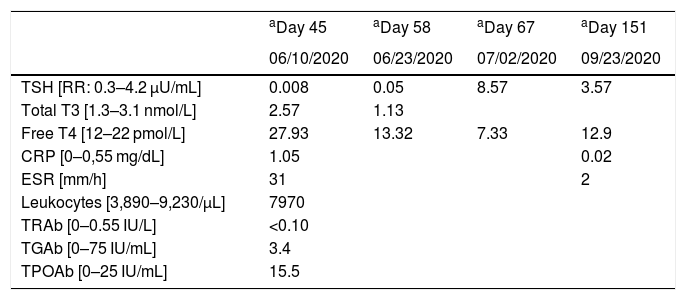
On a blood test from June, free thyroxine (FT4) was elevated with thyrotropin (TSH) suppression. Erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) levels were high and leukocyte levels were normal. Thyrotropin receptor antibodies (TRAb), thyroglobulin antibodies (TGAb), and thyroid peroxidase antibodies (TPOAb) were measured and all were negative. Table 1 summarizes the laboratory test results.
Laboratory findings: subacute thyroiditis following COVID-19 infection.
| aDay 45 | aDay 58 | aDay 67 | aDay 151 | |
|---|---|---|---|---|
| 06/10/2020 | 06/23/2020 | 07/02/2020 | 09/23/2020 | |
| TSH [RR: 0.3–4.2 µU/mL] | 0.008 | 0.05 | 8.57 | 3.57 |
| Total T3 [1.3–3.1 nmol/L] | 2.57 | 1.13 | ||
| Free T4 [12–22 pmol/L] | 27.93 | 13.32 | 7.33 | 12.9 |
| CRP [0–0,55 mg/dL] | 1.05 | 0.02 | ||
| ESR [mm/h] | 31 | 2 | ||
| Leukocytes [3,890–9,230/μL] | 7970 | |||
| TRAb [0–0.55 IU/L] | <0.10 | |||
| TGAb [0–75 IU/mL] | 3.4 | |||
| TPOAb [0–25 IU/mL] | 15.5 |
RR: reference range; TSH: thyroid stimulating hormone; T3: triiodothyronine; T4: thyroxine; ESR: erythrocyte sedimentation rate; CRP: C-reactive protein; TRAb: thyrotropin receptor antibodies; TGAb: thyroglobulin antibodies; TPOAb: thyroid peroxidase antibodies.
In regard to imaging tests, both the thyroid ultrasound and gammagraphy were compatible with the suspected subacute thyroiditis. As initial treatment, she used a nonsteroidal anti-inflammatory drug (NSAID) (ibuprofen 600 mg), a beta blocker (propranolol 10 mg), and a stomach protector (omeprazole 20 mg). The doses of the drugs were reduced according to clinical and analytical progress. After 15 days, the inflammatory parameters had significantly decreased, with normal levels of free T4 and suppressed TSH. The patient did not report any symptoms.
At the beginning of July, the patient’s blood test results were compatible with hypothyroidism; she did not have any symptoms. The dose of ibuprofen was again decreased; the stomach protector was maintained. Levothyroxine was not prescribed given that she was asymptomatic. In a later follow-up appointment in September, thyroid function and the inflammatory parameters had normalized and, finally, treatment was suspended.
Cases of subacute thyroiditis following SARS-CoV-2 infection have been reported in other countries: Italy (Brancatella et al.,7 Ippolito et al.,8 Ruggeri et al.9), Turkey (Asfuroglu et al.10), the United States of America (Chong et al.11), Mexico (Campos-Barrera et al.12), and Singapore (Mattar et al.13). The onset of this pathology, related to a viral infection or a postviral inflammatory reaction in subjects with a genetic predisposition, tends to occur two to eight weeks after said infection.1
It has been demonstrated that the thyroid gland contains a significant amount of angiotensin-converting enzyme 2 (ACE2), which is essential for SARS-CoV-2 to be able to invade human cells. This could be a plausible mechanism for the physiopathology of thyroiditis in COVID-19.14 Similar cases have recently been published7–13 that allow for considering SARS-CoV-2 as the probable viral trigger of our specific case.
Subacute thyroiditis is a self-limiting autoinflammatory disease characterized by a change in the thyroid follicles with inflammatory infiltration of giant cells, lymphocytes, and neutrophils.3 Its incidence is 12.1 cases per 100,000 people per year and it is more frequent among women. The reported cases of thyroiditis following SARS-CoV-2 infection are all in women between 18 and 69 years of age.7–13 The patients reported neck pain, mild fever, and constitutional symptoms. In the described cases, including ours, symptoms started from one to six weeks after infection. Only two cases occurred at the same time.8,10
Thyroid dysfunction in this disease is characterized by three phases: thyrotoxicosis, hypothyroidism, and euthyroidism.3 During the initial phase of follicle destruction, thyroid hormones are released into the blood, which is accompanied by the typical symptoms of hyperthyroidism (palpitations, weight loss, fatigue, etc.) and characteristic laboratory findings (ESR and CRP elevation, T3 not disproportionally elevated, and thyroid autoimmunity (TPO and antithyroglobulin) that is undetectable or present in low titers).3 The thyroid ultrasound is characterized by diffuse bilateral hypoechoic areas and low or absent vascularization on a color Doppler ultrasound. The thyroid gammagraphy showed a block or decrease in uptake of the radiotracer.15
Several weeks after the onset of symptoms, the thyroid hormone reserve is exhausted and the hypothyroid phase sets in, which will be permanent in a small percentage of cases.3,9 A return to a state of euthyroidism can take from one to six months.
Accepted treatment for this entity includes NSAIDs or steroids. Patients with palpitations, tremors, or jitteriness improve with beta blockers.3 Thioamides must not be used.
Unlike the majority of reported cases, we treated our patient with an NSAID and propranolol; symptoms remitted after two weeks of treatment. Similarly, Chong et al.11 used acetylsalicylic acid and propranolol.
We still have a lot to learn about the clinical manifestations of SARS-CoV-2 infection. We believe possible thyroid involvement must be taken into account given that it can go unnoticed in the face of the magnitude of this pandemic. Likewise, SARS-CoV-2 should be considered a possible causative agent of the thyroiditis cases we see in our clinical practice.
FundingThis work has not received any type of funding.
Conflicts of interestThe authors declare that they do not have any conflicts of interest.
Please cite this article as: de la Higuera López-Frías M, Perdomo CM, Galofré JC. Tiroiditis subaguda tras infección por COVID-19. Rev Clin Esp. 2021;221:370–372.