A high incidence of postoperative nausea and vomiting has been observed in patients undergoing laparoscopic cholecystectomy (20–40%). This causes an increase in patient discomfort, a delay in recovery, and an increase in hospital stay.
ObjectiveCompare the efficacy of ondansetron plus metoclopramide and ondansetron alone in the control of postoperative nausea and vomiting in patients undergoing laparoscopic cholecystectomy.
Material and methodsA comparative, prospective, experimental, longitudinal double blind study was performed with 30 patients programmed for laparoscopic cholecystectomy. The Apfel score was applied postoperatively. Patients were divided into two groups with 15 participants. Metoclopramide 10mg and ondansetron 4mg were administered in the first Group (A), and ondansetron 4mg in the second Group (B), after induction of anesthesia. Nausea and vomiting were evaluated for 24h postoperatively using a Likert scale.
ResultsOf the patients who presented nausea, the condition was mild. The patient who presented vomiting, vomited only once. Neither group required rescue drugs. Regarding the Apfel score, there was no correlation between the percentage of stratified risk and the patients who presented postoperative nausea and vomiting.
ConclusionsNo statistically significant difference was observed between ondansetron plus metoclopramide and ondansetron alone in patients subjected to laparoscopic cholecystectomy.
Laparoscopic cholecystectomy is the golden choice in the treatment of gallbladder disease and has been proven to be better in the cost-effectiveness-efficiency relation when compared to open surgery.1,2
Out of 100 laparoscopic cholecystectomies performed, between 20 and 42% present a complication, postoperative nausea and vomiting (PONV), which delays the patients’ discharge. It translates into greater postoperative discomfort and distress for patients, on top of the fact that they usually remain 50% longer in the recovery room, thus altering the patient flow of the hospital and increasing internment costs. PONV can also increase suture tension and the risk of pulmonary aspiration, which could lead to pneumonia or pneumonitis and the increase of intracranial pressure. Its etiology includes dehydration by fasting, the use of opioids, nitrous oxide, inhaled anesthetics in general anesthesia, pneumoperitoneum, CO2 and handling of the parietal peritoneum with vagal stimulation. The risk of PONV ought to be stratified preoperatively and reduced, adapting anesthetic techniques or using the available pharmacological resources.3–5
There are different PONV predictive models that, if applied, can help prevent its presence. When ranked by sensitivity, the Apfel score is the most used in the reviewed bibliography (see Table 1).6
Apfel risk assessment scale for nausea and vomiting.
| Risk factors | Points | Risk |
|---|---|---|
| Female | 1 | Low risk (0–1 points) 10–20% |
| Non-smoker | 1 | Moderate risk (2 points) 40% |
| Previous history of postoperative vomiting and/or motion sickness | 1 | High risk (3–4 points) 60–80% |
| Use of postoperative opioids | 1 |
I. Bel Marcoval. Estratificación del riesgo, profilaxis y tratamiento de las náuseas y vómitos postoperatorios. Rev. Esp. Anestesiol. Reanim. 2006; 53: 301–311.
The different vomit stimuli are related to the dopaminergic, histaminergic, serotoninergic, muscarinic and opioid receptors located in the central pathways (reticular formation of the medulla) and peripheral pathways (gastrointestinal system), thus their blockage is a mechanism of antiemetic medications.
There are different antiemetic families; H1 antihistamines, anticholinergic, steroids, phenothiazine, butyrophenones, dopamine antagonists and 5-HT3 receptor antagonists. These medications are currently being utilized in an isolated or combined form. Metoclopramide, an antiemetic from the benzamide group with anti-dopaminergic and central and peripheral antiemetics, acts in the upper gastrointestinal tracts for 2h (10–20mg for every 70kg on average in adults). Ondansetron is an antagonist of the serotonin receptor (5-HT3). It has been used for some years to control nausea and vomiting induced by chemotherapy and radiotherapy. It has central and peripheral effects. Doses of 4mg peak plasmatic concentrations are reached in approximately 7–11min depending of the speed of administration.7–9
The objective of our study was to compare the clinical effectiveness of ondansetron with metoclopramide in a single dose and ondansetron in an isolated form in the control of postoperative nausea and vomiting in patients undergoing laparoscopic cholecystectomy.
Methods and materialsThe current study included patients who were programmed for elective laparoscopic cholecystectomies at the surgical ward from the “Dr. José E. Gonzalez” University hospital of the Autonomous University of Nuevo León, México. The study consisted of a comparative, prospective, experimental, longitudinal double blind study. Our institution's Ethics Committee reviewed and authorized the project. The inclusion criteria were patients undergoing elective laparoscopic cholecystectomy, between 18 and 60 years old, who weighted between 50 and 90kg. ASA I-II patients and both genders were included. The exclusion criteria were ASA III-IV patients, under 18 years old, allergic to the medications included in this study, pregnant women, patients who were chronically taking prokinetics, patients with an Apfel score of 3–4 and patients who required any other medication during induction. Within exclusion criteria were patients who presented allergic reactions during the procedure, those with conversions to open surgery and those who required the use of other rescue antiemetic medications.
The sample calculation was performed based on the article “Risk stratification, nausea and vomiting prophylaxis” postoperative, published by Dr. I Bel Marcoval et al., in the Spanish magazine of Anesthesiology in 2006, using the following formula, ((2*σ2)/(−Δ+μd)2)*(Z1−α2+Z1−β)2. We obtained a sample size of 30 patients, via a non-inferiority trial and with a confidence level of 5% (p<0.05). The sample was divided into two groups of 15 patients each. Metoclopramide 10mg and ondansetron 4mg were administered in the first Group (A), and ondansetron 4mg in the second Group (B), after induction of anesthesia.
The general objective of our study was to compare the clinical effectiveness of ondansetron with metoclopramide in a single dose and ondansetron in an isolated form in the control of postoperative nausea and vomiting in patients undergoing laparoscopic cholecystectomy. Within our secondary objectives were the application and comparison of the Apfel scale for the risk of nausea and vomiting in the preoperative of both groups, to assess the antiemetic efficacy of both medications for their study groups, and the requirement of rescue medications.
In the pre-surgical area, demographic data was collected, such as age, gender and body mass index (BMI). Their ASA was determined based on their pathological and non-pathological background. In addition, we obtained the signature of the patient (or tutor) for the informed consents. The patients were pre-medicated with ranitidine and midazolam, and were evaluated with the Apfel score. After the patient entered the operation room (OR), monitoring and the taking of basal vital signs were conducted. Anesthetic induction consisted of fentanyl 3mcg/kg, etomidate 0.2mg/kg and atracurium 0.4mg/kg with isoflurane-based anesthetic maintenance. The first group (A) received ondansetron 4mg and metoclopramide 10mg in a single dose in a slow infusion for 20min after anesthetic induction and the second group (B) received ondansetron 4mg in the same fashion. The degree of nausea and vomiting was evaluated with the Likert scale of 5 points (0=none, 1=preview, 2=mild, 3=moderate, 4=high, 5=intense retching and presence of vomit). Patients were evaluated immediately and 24h after the postoperative period. Basal time measurements were conducted 4, 8, 12 and 24h later.
Statistical analysis was performed via a non-inferiority trial; qualitative and quantitative variables were used. An alpha value of 0.05 was also used and null hypothesis was rejected when the critical value was under 0.05. Nonparametric tests were used (x2 and parametric (t student)). The electronic data processing and statistical methods (descriptive and inferential) were performed using STATA-IC-10-2010 software.
ResultsIn the period, a total number of 30 patients were obtained and divided into 2 groups; group A (metoclopramide and ondansetron) with 15 patients and group B (ondansetron) with 15 patients.
Group A consisted of 2 male patients (13.3%), and 13 female (86.6%) patients, while group B consisted of 1 male (6.66%) and 14 females (93.3%). The difference between the two groups was not statistically significant. Regarding age range, 11 patients were between the ages of 18 and 29, 8 of them were between 30 and 39, 5 were between 40 and 49 and 6 were between 50 and 59. Group A consisted of 4 patients between the ages of 18 and 29, 5 between 30 and 39, 3 between 40 and 49 and 3 were between 50 and 59. On the other hand, Group B consisted of 7 patients between the ages of 18 and 29, 3 were between 30 and 39, 2 were between 40 and 49 and 3 were between 50 and 59. A statistically significant difference was not found in the mean age of the groups. Regarding BMI, 8 of the patients who participated in the study had a normal BMI (18.5–24.9), 15 were overweight (25–29.9) and 7 had grade I obesity (30–34.9). Within Group A, there were 4 patients with a normal BMI, 10 overweight and 1 with grade I obesity, whereas in Group B there were 4 patients with normal BMI, 5 overweight and 6 with obesity. Once again no significant differences between Group A and Group B were observed. It was noted that 20 of the patients included in the study were cataloged as ASA I and 10 as ASA II. No statistically significant difference was observed in this parameter.
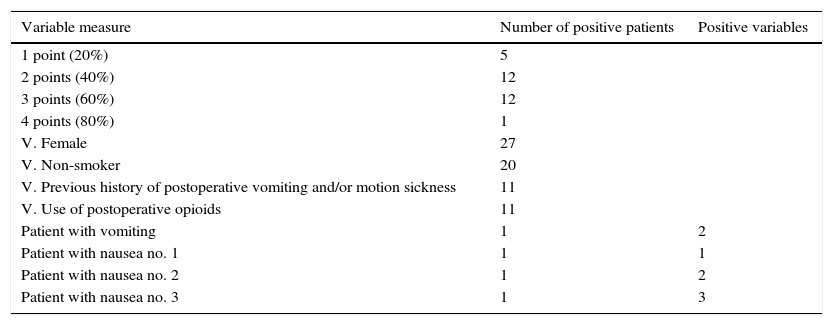
As for the Apfel score, of the 30 patients included in the study, 5 presented 1 risk factor out of the 4 of the evaluation, representing a risk of 20% of suffering PONV. 12 patients presented 2 factors (a risk of 40%); 12 patients presented 3 factors (a risk of 60%) and 1 presented 4 factors (a risk of 80%). It was also noted that out of the 30 individuals of the study, 27 were women, 20 were non-smokers, 11 did not have a PONV or motion sickness background and postoperative opioids were used in 11 patients. The patient who presented a vomiting episode had 2 positive variables in the Apfel score, and the 3 subjects of study who presented mild nausea had 1, 2 and 3 positive variables respectively (see Table 2). No statistically significant differences were observed in this parameter either.
Data obtained in relation to the Apfel scale.
| Variable measure | Number of positive patients | Positive variables |
|---|---|---|
| 1 point (20%) | 5 | |
| 2 points (40%) | 12 | |
| 3 points (60%) | 12 | |
| 4 points (80%) | 1 | |
| V. Female | 27 | |
| V. Non-smoker | 20 | |
| V. Previous history of postoperative vomiting and/or motion sickness | 11 | |
| V. Use of postoperative opioids | 11 | |
| Patient with vomiting | 1 | 2 |
| Patient with nausea no. 1 | 1 | 1 |
| Patient with nausea no. 2 | 1 | 2 |
| Patient with nausea no. 3 | 1 | 3 |
Regarding the efficacy of the antiemetic in study, 1 patient presented vomiting on a single occasion immediately after extubation. He was a part of Group B (ondansetron). 2 of the 3 patients who presented nausea (number 2 in the Likert scale) belonged to Group A (metoclopramide and ondansetron) and presented it in the basal hour and at 4h. It ceased without rescue medication. The third of the 3 patients with nausea (number 2 in the Likert scale) belonged to Group B (ondansetron) and only presented it during the basal hour; it ceased without rescue medication. However, no differences were found in the severity of the nausea or the satisfaction of the patients between groups. Thus, the difference between both groups was not statistically significant.
DiscussionPONV is among the most frequent complications following anesthesia in the postoperative of laparoscopic cholecystectomies; 42% of post-operated laparoscopic cholecystectomy patients present nausea and vomiting, which translates into a greater postoperative discomfort and distress for patients, on top of the fact that they usually remain 50% longer in the recovery room, thus altering the patient flow of the hospital and increasing the internment costs of each patient. This is reduced using effective antiemetics in a single dose previous to its presentation. The origin of this complication is complex and depends on a wide variety of factors previously mentioned.
In this study, the treatment groups are similar in relation to the demographic data (inter-group homogeneity), surgical procedure, and type of anesthesia and analgesia utilized in the postoperative.
The Apfel score was used to assess the risk of presenting PONV, in order to observe its ability to forecast this event and because it includes the predisposed factors which have been observed, most in different studies, such as the Lenka Doubravskaa in 2010, which included 1954 patients. Here, they noted the fact that being a woman increased the risk of presenting PONV, with an occurrence of 24.5% in females and 6.3% in males. On the other hand, smoking seemed to be a protective factor. The incidence of PONV in smokers was 8.7%, compared to non-smokers, which was 17.7%. Also, a high PONV incidence was observed in patients with obesity (BMI over 30), at 11.8%. In our study, we were able to observe that the 4 patients who did not present PONV were all women; 2 smokers and 2 non-smokers; 2 with a PONV or motion sickness background; and none were administered opioids in postoperative. The patient who presented vomiting in the postoperative had 2 positive variables in the Apfel score, and the 3 patients who presented mild nausea had 1–3 variables, which cast doubt on the efficacy of the Apfel score as the most sensitive PONV predictor.10
Different studies have been conducted around the world, where different managements for PONV prevention are proposed. The use of combined therapies for a better management is recommended in many of them, stressing it in patients with high risk factors.11,12
In a study conducted in Saudi Arabia by Dr. Mohamed Naguib et al. they included 132 patients undergoing laparoscopic cholecystectomies, who were randomly administered ondansetron 4mg, tropisetron 5mg, granisetron 3mg, metoclopramide 10mg or a placebo 10min before anesthetic induction. The percentages of patients free of vomiting were 65.5%, 52%, 48%, 29.2% and 27.6% in the ondansetron, granisetron, tropisetron, metoclopramide and placebo groups, respectively. Prophylactic antiemetic treatment with ondansetron resulted in a lower PONV incidence (p=0.02) than with metoclopramide or placebos. In our study, we were able to observe that the antiemetic therapy with ondansetron with metoclopramide in a single dose is not superior to that of just ondansetron.13
ConclusionsAntiemetic combined therapy has proven in some studies to be more effective than monotherapy in the control of PONV. In our study, a statistically significant difference was not found in the efficacy of polytherapy with ondansetron with metoclopramide (4mg+10mg) in a single dose and monotherapy with ondansetron (4mg) in patients undergoing laparoscopic cholecystectomies under general anesthesia. However, both groups of patients presented a good control of PONV without the need for rescue medications.
Regarding the Apfel score to assess the risk of presenting PONV, in the present study we did not find a correlation between the risk percentage stated by the score and the patients who presented PONV.
Therefore, based on the results obtained in our study we accept the established hypothesis that the combination of ondansetron and metoclopramide in a single dose is as effective as just ondansetron for the control of postoperative nausea and vomiting.
Conflict of interestThe authors have no conflicts of interest to declare.
FundingNo financial support was provided.