Since its emergence at the end of 2019, the pathophysiology and clinical presentation of SARS-CoV-2 infection is becoming increasingly well understood as time goes by.1 One aspect to consider is the possible association with antiphospholipid antibodies, due to growing evidence of its association with an increased systemic prothrombotic effect. There are studies on the determination of these antibodies during the acute phase of infection2,3; however, the aim of our study is to determine the presence of these antibodies in patients who have overcome the infection and the persistence or disappearance of these antibodies during follow-up.
A specialist consultation follow-up was carried out on 189 patients admitted to the inpatient ward of the Hospital Virgen de las Nieves in Granada between March and October 2020, all of them with confirmed SARS-CoV-2 infection by PCR or serology, excluding unconfirmed suspects. In addition to the clinical and radiological re-evaluation, lupus anticoagulant and antiphospholipid antibodies (anti-cardiolipin IgM and IgG; anti-beta2 glycoprotein IgM and IgG) were determined in 2 cut-off values. The cut-off value for lupus anticoagulant positivity was set at 1.2 in at least one of the tests used in our hospital laboratory (Russell’s viper venom test and activated partial thromboplastin); antiphospholipid antibodies were divided into weak (10–40IU/ml) and strong (>40IU/ml) positivity.
Of the 189 patients included, 44.4% were women and the rest men (55.6%), with a mean age of 59 years (SD 15). Medical history included: hypertension (43.3%), diabetes mellitus (18.9%), hypercholesterolemia (26.7%), asthma (13.3%), chronic obstructive pulmonary disease (5%), SAHS (8.9%) and previous venous thromboembolic disease (4.8%). 64.5% had a body mass index greater than 25. 6.3% required admission to Intensive Care and 4.2% invasive mechanical ventilation. None of the included patients had a known history of systemic autoimmune disease. 5.8% were active smokers and 23.8% former smokers.
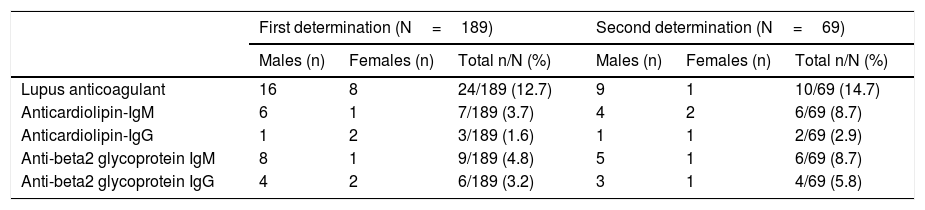
The results of the 2 determinations are shown schematically in Table 1. The first was performed on average 89 days after a PCR positive result and the second after 180 days.
Results of the 2 determinations and percentage of the total.
| First determination (N=189) | Second determination (N=69) | |||||
|---|---|---|---|---|---|---|
| Males (n) | Females (n) | Total n/N (%) | Males (n) | Females (n) | Total n/N (%) | |
| Lupus anticoagulant | 16 | 8 | 24/189 (12.7) | 9 | 1 | 10/69 (14.7) |
| Anticardiolipin-IgM | 6 | 1 | 7/189 (3.7) | 4 | 2 | 6/69 (8.7) |
| Anticardiolipin-IgG | 1 | 2 | 3/189 (1.6) | 1 | 1 | 2/69 (2.9) |
| Anti-beta2 glycoprotein IgM | 8 | 1 | 9/189 (4.8) | 5 | 1 | 6/69 (8.7) |
| Anti-beta2 glycoprotein IgG | 4 | 2 | 6/189 (3.2) | 3 | 1 | 4/69 (5.8) |
It is noteworthy that these antibodies remain positive 6 months after acute SARS-CoV-2 infection, even with high titres. In addition, 3 patients with triple positivity (lupus anticoagulant, anticardiolipin, and anti-beta2 glycoprotein) and 3 patients with double positivity (combination of 2 of them) were observed.
Another aspect that we consider important is the higher prevalence of positive results for lupus anticoagulant, anticardiolipin-IgM and anti-beta2 glycoprotein (IgM and IgG) in men, with a greater difference than in women for lupus anticoagulant at 180 days.
During the follow-up of these patients no thromboembolic events have been observed after hospital discharge, so the potential pathogenic role of these antibodies is still unknown. We do not know whether, in the future, a percentage of them will meet criteria for antiphospholipid syndrome and develop thrombotic events,4 or whether it is a transient immune reaction associated with the viral infection itself.5
Conflict of interestsThe authors declare that they have no conflict of interest.
Please cite this article as: Rosales-Castillo A, Sabio JM. Determinación de anticuerpos antifosfolípidos durante el seguimiento de pacientes tras infección SARS-CoV-2. Med Clin (Barc). 2022;158:437–438.