On 11th March 2020, the World Health Organization (WHO) declared a global pandemic status due to the current outbreak of coronavirus disease (COVID-19), which was first reported in Wuhan (China) on 31 December 2019.1 At present, COVID-19 is severely impacting a large number of countries, including those that are supposed to be most resilient. In 2009, Castro et al. indicated the need to create a triage system to adapt our healthcare system to possible influenza pandemics.2 This article written for Influenza A (H1N1) already showed the need to adapt protocols to the different phases of the pandemic, to avoid the spread of the virus, the saturation of healthcare centres with mild patients and the contamination of healthcare facilities with the virus, as well as reducing the morbidity and mortality of critically ill patients.
In this same sense, with respect to COVID-19, the Center for Disease Control and Prevention (CDC), recommends decreasing and taking extreme caution with aerosol-generating procedures,3 such as: Ventilation with mask and self-inflating bag, aspiration of secretions, orotracheal intubation (OTI), application of nebulisations, use of non-invasive ventilation (NIV) and cardiopulmonary resuscitation (CPR) manoeuvres.
The results of the analysis of a cohort of COVID-19 patients throughout China have shown that 3.4% of infected patients presented with acute respiratory distress syndrome (ARDS), representing 40% of the total number of patients who showed severe pathologies.4
ARDS causes exudate alveolar invasion in the lung and a decrease in lung compliance, generating mainly hypoxemic respiratory failure. In prehospital care, diagnosis is based on detecting an ineffective respiratory pattern with impaired ventilatory auscultation, a decrease in oxygen saturation, and an increase in respiratory rate.
In these patients, oxygenation with a reservoir mask will be a priority as the first step in the treatment of ARDS. In the second step, the use of non-invasive ventilation (NIV) will be considered as risk due to possible leaks that may appear,4,5 especially during ambulance transport, with invasive mechanical ventilation and extracorporeal membrane oxygenation (ECMO) being relegated to the last step of treatment. The administration of inhaled therapy must be strictly supervised to avoid external contamination.5
At the prehospital level, we are faced with the problem of how to ventilate patients who are inefficient in oxygenation by means of a mask with a reservoir bag. In this case, the next step would be the use of NIV, but we have the risk of external contamination. As it is a high-flow ventilation, its use is limited to hospital rooms with negative pressure.
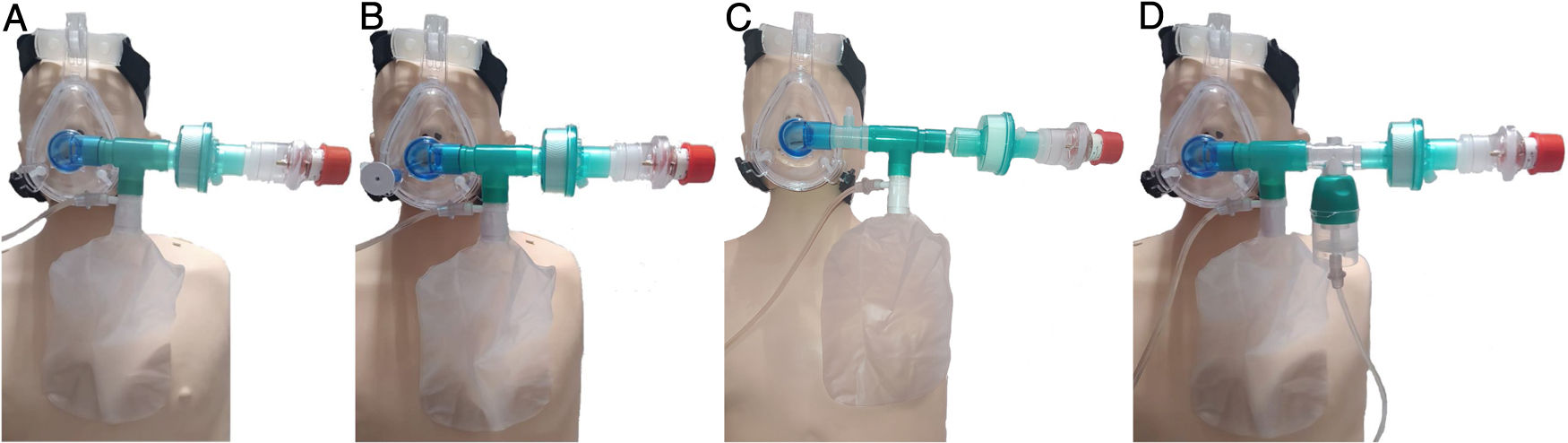
In this exceptional context that COVID-19 has generated, it is where we propose to assess the use of our device that, although we do not have randomized clinical trials (RCTs) that support its use, we think that it can be an alternative when there is no chance of performing mechanical ventilation, and especially for patients in whom the usual oxygen therapy techniques are not being effective enough, designed not to replace NIV but as an alternative in this context. With the 'oxygenation device with reservoir and PEEP' (ODRPEEP) (Fig. 1) we can oxygenate the patient with a reservoir bag in the inspiratory phase, and in the expiratory phase the inlet of the reservoir bag will be closed with the built-in valve and the exhalation will be done through a virus and bacteria-proof filter with a > 99.9% efficacy and a PEEP valve avoiding alveolar collapse thanks to a spring system. In addition, the device will allow us to safely apply inhaled drugs.
Diagram of the different options related to the oxygenation device with reservoir and PEEP (ODRPEEP).
A: It consists of an appropriately sized, one-piece "T-shaped" NIV mask with closed elbow connected to a reservoir bag with its 15 L/min oxygen connection, a filter for bacteria and viruses with a filtering efficiency of > 99.99 % and a PEEP valve. The necessary inspiration volume will be made from the reservoir bag while the patient's expiration is done through the PEEP valve with an antibacterial and viral filter.
B: In the case of needing to administer inhaled drugs, we use the pressurized cartridge fitting, needing an O2 extension end for its connection which we will snap into one of the holes in the mask.
C: We can apply an inhaled drug without nebulization. We will use the diagram of option «a» adding a T-fitting for a pressurized cartridge between the elbow and the T of the reservoir bag.
D: It will allow us to nebulize reducing the risk of aerosol leaks. We will use the diagram of option «a» adding a T-piece with a nebulization cup and extension. Without nebulization, we will provide the reservoir with a flow of 15 L/min and during nebulization, the flow must be distributed between the reservoir and the nebulizer cup to avoid hyperoxygenation.
The inherent safety of this device is based on the fact that there is no external contamination thanks to the NIV mask, the low pressures inside and the exhalation through the filter. Even so, the authors recommend caution when applying and conducting RCTs that compare the results of ODRPEEP with NIV and determine if both options can be considered to have a certain therapeutic equivalence.
FundingThis article has not received any type of funding.
Conflict of interestsThe authors declare no conflict of interest.
Please cite this article as: Segura Alba O, Nieto Fernández-Pacheco A, Pardo Ríos M. Dispositivo de oxigenación con reservorio y PEEP (DORPEEP) en pacientes con dificultad respiratoria aguda por COVID-19. Med Clin (Barc). 2020;155:133–134.