The introduction of the anti-tumor necrosis factorα agents (anti-TNFα) in clinical practice has greatly advanced the treatment of inflammatory bowel disease. The use of these medications results in durable remission in a subset of patients, preventing surgery and hospitalizations. However, there are some concerns about safety and costs associated with their long-term use. Therefore, anti-TNF withdrawal has emerged as an important consideration in clinical practice. Herein our goal was to discuss the available evidence about anti-TNFα discontinuation in IBD that could inform the clinician on the expected rates of relapse, the potential predictors of relapse, as well the response to re-treatment.
A introdução dos anti-TNFα na prática clínica produziu um avanço considerável no tratamento da doença inflamatória intestinal. A utilização destes medicamentos resulta numa remissão duradoura num sub-grupo de doentes, evitando cirurgias e hospitalização. No entanto, existem algumas preocupações com a segurança e o custo associados com a sua utilização a longo prazo. Assim, a descontinuação dos anti-TNFα tem emergido como uma questão importante na prática clínica. Pretendemos neste artigo, discutir a evidência disponível sobre interrupção dos anti-TNFα na doença inflamatória intestinal, proporcionando ao clínico informação actualizada sobre as taxas de recidiva após interrupção do fármaco, potenciais factores predictores de recidiva, assim como a resposta ao re-tratamento.
The introduction of anti-TNF has changed the course of inflammatory bowel disease (IBD). These drugs have proven efficacious in healing the mucosa, and in reducing surgical and hospitalizations rates. Their introduction in the clinic led to major shifts in the therapeutic paradigm that evolved from mere symptomatic clinical remission toward sustained and deep remission. In parallel with the evolution of these therapeutic goals, we have also seen a shift in treatment strategies in IBD. Central to these strategies are the early introduction of immunosuppression, the rapid escalation to anti-TNF therapies (accelerated step-up strategy) or the early introduction of combination therapy (top-down strategy), allied with a tight and frequent control of inflammatory activity, and adjustment of therapy based on that assessment (treat-to-target strategy).1,2 As a result of these strategies, there has been an exponential increase in the number of patients that are treated with anti-TNF monotherapy, or more frequently as combination therapy. However, despite their proven efficacy, anti-TNF's high cost and safety issues are among the main concerns claimed against their long-term use that may motivate doctors and patients to discontinue the drugs when sustained and deep remission has been reached. Questions on if, when and in whom we should discontinue anti-TNF are actively debated by the scientific community. Many studies have now reported on the relapse rates following drug de-escalation, in the hope of identifying a subset of patients in whom therapy could be reduced to the minimal effective therapy that could maintain remission. However, so far no definitive evidence or strategy has been defined. Herein, our goal was to review and summarize the available evidence about anti-TNF discontinuation in IBD, in a comprehensive way that could provide information and hopefully guidance in clinical practice.
2Why is anti-TNF withdrawal considered in clinical practice?It may seem counterintuitive to stop a therapy once its therapeutic goals have been achieved; both clinicians and patients may be reluctant, fearing relapse and/or loss of efficacy in the event of re-treatment. However, therapeutic de-escalation is considered in clinical practice for several reasons, namely: (1) safety concerns or side-effects, (2) cost/national regulations, and (3) special situations such as pregnancy, travel or patient preference.
2.1SafetyOverall, if used with the due precautions, anti-TNF agents have a good short and medium-term safety profile. However, we must also recognize that the follow-up time for anti-TNF and combination therapy is still very limited. An example of this can be drawn from thiopurine use in IBD. Although these drugs have been used in IBD for more than 30 years,3 only recently has it been definitely confirmed their association with the development of lymphoproliferative disorders4 and skin cancer.5 The increased risk of infections and/or malignancies is the most important concern associated with long-term anti-TNF therapy in IBD,6 especially when used in combination therapy. Anti-TNF therapies may increase the risk of infections,6 especially fungal and bacterial,7 and marginally increase the risk of melanoma.8 Patients on combination therapy may have an increased risk of developing hepatosplenic T-cell lymphoma, non-Hodgkin's lymphoma,9 and infections.6 Certain populations, such as the elderly or male pediatric patients,10–12 may be at higher risk for specific complications. Furthermore, side effects may require drug discontinuation.
2.2Cost/national regulationsIn parallel with reduced surgery and hospitalization rates in the biological era,13 the recently published COIN study,14 demonstrated that direct healthcare costs in IBD are now led by medications. The goal of this study, performed in the Netherlands, was to provide an updated overview of general costs in IBD the current era of expanding use of biologics. This study showed that anti-TNF use accounted for 64% and 31% of the direct costs in CD and UC respectively.14 Conversely, surgery, hospitalization, and sick leave accounted for only 19%, <1% and 16% of the healthcare costs in CD, and 23%, 1%, and 39% of costs in UC patients respectively.14 Whilst these calculations do not argue against the cost-effectiveness of these therapies (rather it is the other way around), these costs are indeed substantial. In fact, a number of national societies and expert panels have recommended stopping anti-TNF after one or two year(s) of therapy, provided that clinical remission has been achieved.15–17
2.3Special situations (pregnancy, traveling, patient preference, insurance policies)An increasing number of female IBD patients conceive whilst under treatment with anti-TNF. Infliximab (IFX) and adalimumab (ADA) cross the placenta and serum drug levels in the baby may exceed those of the mother.18,19 Therefore discontinuation by the end of the second trimester is generally recommended in pregnant patients in remission.20 Other special situation refers to patients traveling for long periods in areas of the world where access to medications or medical facilities is limited, or where serious infections such as tuberculosis are endemic. The decision to continue anti-TNF in such special situations needs to be carefully balanced against the risk of relapse, taking into account patient's expectations.
3What are the relapse rates after stopping?Clinicians and patients are often faced with the question on whether it is possible to stop anti-TNF therapy given that disease remission has been conquered. However, despite all the studies that have now addressed this issue in IBD, no conclusive strategy has yet emerged.21 This may be due to large heterogeneity across different studies precluding firm conclusions on the ideal clinical scenario when the drug could be stopped. Furthermore, and most importantly, it is difficult to draw firm conclusions about the effect of anti-TNF withdrawal due to the lack of control data.
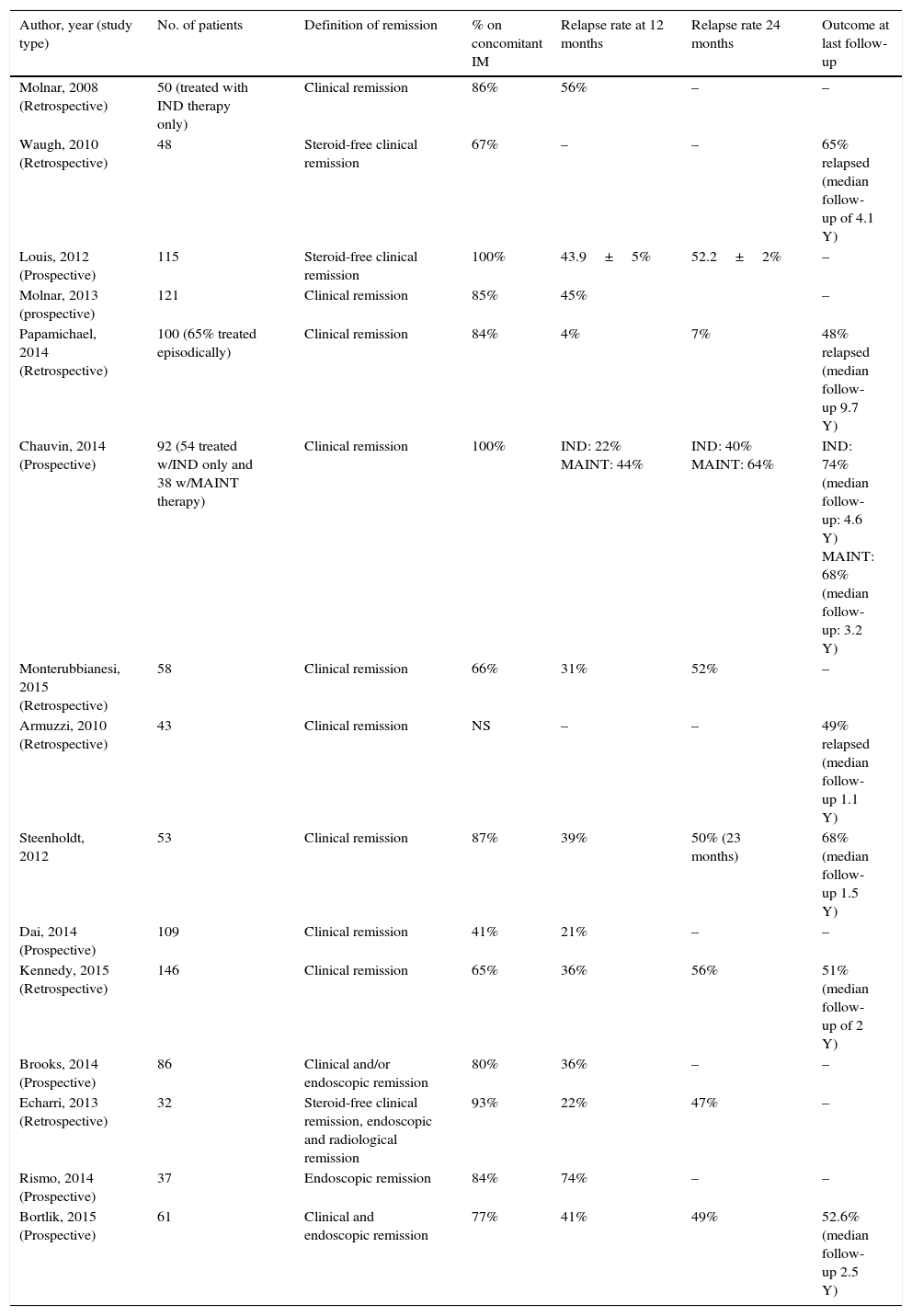
3.1Anti-TNF discontinuation in Crohn's diseaseMany studies have now looked into anti-TNF withdrawal in Crohn's disease (see Table 1 for a summary of most relevant studies).22–33 The STORI trial (Infliximab diSconTinuation in Crohn's disease patients in stable Remission on combined therapy with Immunosuppressors) was the pivotal study boosting clinical research in this topic, being thereafter followed by many studies. This was a multicenter study led by the GETAID, and it was the first prospective study specifically designed to assess the risk of relapse, and to identify predictors of relapse following anti-TNF maintenance therapy withdrawal.34 Patients entering the trial had to be in steroid-free remission for a minimum of 6 months, while on at least 1 year of scheduled IFX combined with a stable dose of immunomodulators (azathioprine, 6-mercaptopurine, or MTX). Among the 115 CD patients with luminal disease that were enrolled (perianal CD was excluded), there was a 43.9% (±5.0%) rate of relapse over 1 year and a 52.2% (±5.2%) rate of relapse over 2 years after stopping IFX. Relapse occurred after a median of 16.4 months. Following STORI, many other retrospective and prospective cohorts followed. Importantly, it is worth mentioning, that for most of these studies, patients had the anti-TNF discontinued while they were in clinical remission (with variable definitions of clinical remission and variable duration of remission before drug withdrawal). Interestingly, the relapse rates among those studies are fairly homogenous, ranging from 21 to 56% at 12 months and from 47 to 64% at 24 months respectively24,29,31,34–41 (see Table 1). An exception is the study by Papamichael et al42 that reported lower relapse rates after drug discontinuation; the cumulative probability of maintaining sustained clinical remission after the first, second, third, fourth, and fifth year was 96%, 93%, 88%, 79.9%, and 72.8%, respectively. However, by the end of follow-up (median 10 years), 52% of patients had relapsed. Studies with long follow-up periods have reported high relapse rates ranging from 27%42 to 65%36 at 5 years, and 88% at 10 years.37 The long-term (median 6.9 years) follow-up of the STORI trial, recently presented, showed that 25% of patients who discontinued anti-TNF in the original trial eventually developed bowel damage (need for surgical resection, new perianal fistula).43
Summary of most relevant studies specifically reporting on the relapse rates after discontinuation of anti-TNF therapy in adult populations with Crohn's disease, following a variable period of therapy (Note: only studies with >30 patients were included).
| Author, year (study type) | No. of patients | Definition of remission | % on concomitant IM | Relapse rate at 12 months | Relapse rate 24 months | Outcome at last follow-up |
|---|---|---|---|---|---|---|
| Molnar, 2008 (Retrospective) | 50 (treated with IND therapy only) | Clinical remission | 86% | 56% | – | – |
| Waugh, 2010 (Retrospective) | 48 | Steroid-free clinical remission | 67% | – | – | 65% relapsed (median follow-up of 4.1 Y) |
| Louis, 2012 (Prospective) | 115 | Steroid-free clinical remission | 100% | 43.9±5% | 52.2±2% | – |
| Molnar, 2013 (prospective) | 121 | Clinical remission | 85% | 45% | – | |
| Papamichael, 2014 (Retrospective) | 100 (65% treated episodically) | Clinical remission | 84% | 4% | 7% | 48% relapsed (median follow-up 9.7 Y) |
| Chauvin, 2014 (Prospective) | 92 (54 treated w/IND only and 38 w/MAINT therapy) | Clinical remission | 100% | IND: 22% MAINT: 44% | IND: 40% MAINT: 64% | IND: 74% (median follow-up: 4.6 Y) MAINT: 68% (median follow-up: 3.2 Y) |
| Monterubbianesi, 2015 (Retrospective) | 58 | Clinical remission | 66% | 31% | 52% | – |
| Armuzzi, 2010 (Retrospective) | 43 | Clinical remission | NS | – | – | 49% relapsed (median follow-up 1.1 Y) |
| Steenholdt, 2012 | 53 | Clinical remission | 87% | 39% | 50% (23 months) | 68% (median follow-up 1.5 Y) |
| Dai, 2014 (Prospective) | 109 | Clinical remission | 41% | 21% | – | – |
| Kennedy, 2015 (Retrospective) | 146 | Clinical remission | 65% | 36% | 56% | 51% (median follow-up of 2 Y) |
| Brooks, 2014 (Prospective) | 86 | Clinical and/or endoscopic remission | 80% | 36% | – | – |
| Echarri, 2013 (Retrospective) | 32 | Steroid-free clinical remission, endoscopic and radiological remission | 93% | 22% | 47% | – |
| Rismo, 2014 (Prospective) | 37 | Endoscopic remission | 84% | 74% | – | – |
| Bortlik, 2015 (Prospective) | 61 | Clinical and endoscopic remission | 77% | 41% | 49% | 52.6% (median follow-up 2.5 Y) |
Y, years; IND, induction; MAINT, maintenance; NS, non-specified.
The results from the STORI trial suggested that patients with deep remission (clinical and endoscopic) had a very low chance for relapse.34 These results were felt to be encouraging, as they suggested that a subset of patients who had reached deep remission could be good candidates for stopping therapy. Therefore, it was anticipated that stopping anti-TNF in patients presenting full endoscopic and biologic remission, could lead to even higher remission rates. However, recent studies that recruited patients in deep remission (clinical and endoscopic remission) have shown somehow similar relapse rates between 22–74% at 12 months,44,45 47–49% at 24 months,44,46 and 50% at 36 months,27 very similar to those when anti-TNF was discontinued while in clinical remission. For example, in a recently published cohort, patients who discontinued the anti-TNF after endoscopic remission (absence of ulcerations) was attained, were prospectively followed.46 The cumulative probability of relapse at 2 years was 49%.46 The authors compared the relapse rates between the subset of patients that besides endoscopic remission also presented biological remission (FC<150mg/kg and CRP levels ≤5mg/L) to those who only presented clinical and endoscopic remission only. The relapse rates at 2 years were 60% and 52% respectively (non-significant). Although these data require further confirmation, in appropriately powered cohorts, they are somehow disappointing, raising the possibility that even after deep remission has been achieved, the disease will eventually resume its trajectory and the inflammatory cascade will relapse, after the drug has been discontinued.
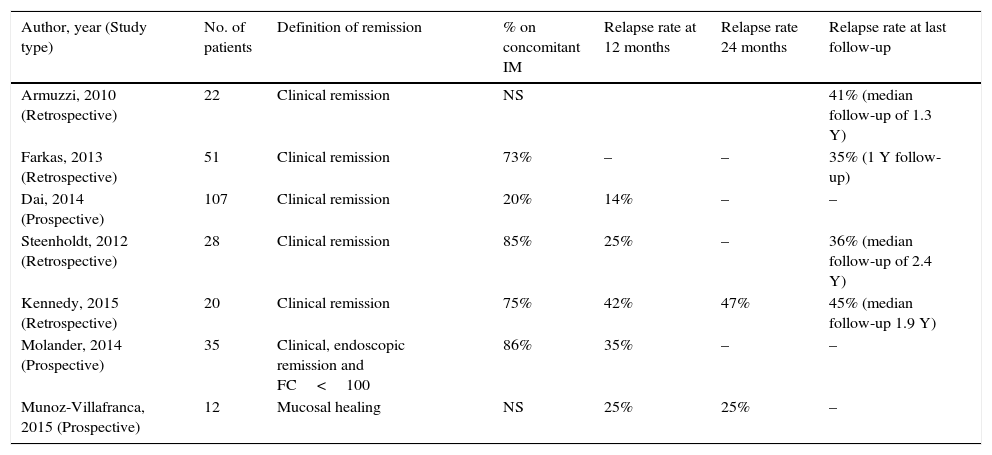
3.2Anti-TNF discontinuation in ulcerative colitisLike in CD, most of the studies that have been made studying anti-TNF discontinuation in UC are very heterogeneous (see Table 2). Nevertheless, the reported relapse rates are somehow similar, perhaps slightly lower than CD. Studies in adult populations have reported relapse rates that range between 14–42% at 12 months, and 25–47% at 24 months24,37,38,41,47–50 (see Table 2). Studies where anti-TNF was discontinued after mucosal healing had been attained, report similar relapse rates at 12 and 24 months of 17–25% and 25–35% respectively47,50. The study with longest follow-up by Steenholdt et al37 reported relapse rates of 60% at 4.5 years.37
Summary of most relevant studies specifically reporting on the relapse rates after discontinuation of anti-TNF therapy in adult populations with ulcerative colitis/inflammatory bowel disease undetermined, following a variable period of therapy.
| Author, year (Study type) | No. of patients | Definition of remission | % on concomitant IM | Relapse rate at 12 months | Relapse rate 24 months | Relapse rate at last follow-up |
|---|---|---|---|---|---|---|
| Armuzzi, 2010 (Retrospective) | 22 | Clinical remission | NS | 41% (median follow-up of 1.3 Y) | ||
| Farkas, 2013 (Retrospective) | 51 | Clinical remission | 73% | – | – | 35% (1 Y follow-up) |
| Dai, 2014 (Prospective) | 107 | Clinical remission | 20% | 14% | – | – |
| Steenholdt, 2012 (Retrospective) | 28 | Clinical remission | 85% | 25% | – | 36% (median follow-up of 2.4 Y) |
| Kennedy, 2015 (Retrospective) | 20 | Clinical remission | 75% | 42% | 47% | 45% (median follow-up 1.9 Y) |
| Molander, 2014 (Prospective) | 35 | Clinical, endoscopic remission and FC<100 | 86% | 35% | – | – |
| Munoz-Villafranca, 2015 (Prospective) | 12 | Mucosal healing | NS | 25% | 25% | – |
Y, years; FC, fecal calprotectin; NS, non-specified.
Since anti-TNF discontinuation is associated with high relapse rates, it is crucial to be able to identify risk factors for recurrence (Table 3) that could inform on the risk/benefit of withdrawing. However, it is important to recognize that most studies on anti-TNF discontinuation were not adequately powered for determining risk factors for recurrence, and therefore clinicians should be cautious on relying exclusively in these factors for making their decision. Stopping anti-TNF (or any other therapy) should instead be a personalized and individualized decision, that should take into account the presence of risk factors for relapse, patient phenotype, patient preference, patient specific risk factors for side-effects, weighing them against the consequences of relapse in the individual patient21 (Fig. 1).
Factors predictive of relapse in IBD (adapted from Torres et al21). Note: only factors identified through multivariable analysis across different studies are presented in this table.
| Factors predictive of relapse |
|---|
| Reflective of disease activity at de-escalation or during follow-up |
| • Elevated inflammatory markers (elevated leukocyte count,34,41 elevated CRP,24,31,34 escalation,51,52 elevated FC25,34,41 |
| • Laboratorial markers suggestive of ongoing inflammation (low hemoglobin)34 |
| • Absence of mucosal healing24,34 |
| Factors reflective of disease poor prognostic features |
| • Smoking29,31 |
| • Perianal disease29 |
| • Disease location (ileocolonic disease35; colonic versus ileal or ileocolonic disease46) |
| • Young age at diagnosis41,42 |
| Previous disease course |
| • Prior disease course marked by higher therapeutic requirements (higher steroid use,34 prior anti-TNF course*,31,35,49 need for dose-escalation prior to discontinuation,31 prior immunomodulator failure29) |
| Other |
| • Male sex (HR 3.7 [1.9–7.4])34 |
| • Elevated/detectable IFX trough levels34 |
Most studies reporting on predictive factors for relapse were made on Crohn's disease, and less evidence is available in UC. Generally speaking, risk factors for disease recurrence can be grouped into four categories: (1) factors indicating (sub-clinical) active disease, (2) prognostic factors for poor disease course, (3) prior disease course, and (4) other mixed factors (Table 3).21 Since many studies have included patients that were discontinued while on clinical remission, it comes as no surprise that evidence of active but sub-clinical disease (reflected as elevated inflammatory markers [elevated C-reactive protein – CRP, elevated fecal calprotectin – FC, high leukocyte count, etc.], presence of ulcers on endoscopy or bowel wall thickening on MRI), is consistently identified across studies as predictive for relapse.25,34,41,51,52 Therefore, it is legitimate to state that clinical remission per se is not enough of an argument to stop a drug in IBD. Physicians that are considering on discontinuing anti-TNF should always make a full work-up on their patients, including an endoscopic and/or radiological examination before making that decision. Those patients presenting risk factors that have been associated with a poor disease course, such as young age at diagnosis, complicated phenotype, and disease located in the terminal ileum, more extensive disease, smoking, etc., will be more likely to relapse when the drug is discontinued.29,31,35,42,46 Likewise, prior disease course may be informative on relapse. Patients that had in the past a disease that was more difficult to control, or that had higher therapeutic requirements, need for dose-escalation to achieve and/or maintain remission, etc. may reflect a group with more severe disease, and therefore may be at higher risk for relapsing.31,35,49 The type and duration of therapy and of disease may impact recurrence rates, as shorter anti-TNF duration and longer disease duration before anti-TNF have been identified as predictors for relapse in some studies, whereas a shorter interval between disease diagnosis and starting anti-TNF, and maintaining the treatment with an immunomodulator were associated with remission.42,53Other factors have also been identified. A very interesting finding, that has now been replicated in some studies is that those patients who have lower or undetectable trough levels before drug discontinuation have a lower chance of relapse.34,42,54 In a retrospective cohort, patients with IBD who discontinued anti-TNF while in remission (endoscopic or radiological remission documented in 95% of patients) had their trough levels measured before discontinuation. The odds for clinical relapse by 12 months of follow-up was 30 times higher (95% CI: 5.8–153) for those who presented detectable trough levels as compared to those who did not.54 This finding had also been observed in the STORI trial, where presenting infliximab trough level >2mg/L (HR 2.5 [1.1–5.4)] was predictive for relapse.34 This may seem counterintuitive, as absence or low trough levels have been associated with worse outcomes,55 boosting the use of therapeutic drug monitoring in the clinic for therapy optimization. However, for the subset of patients in long-lasting remission, low/absent trough levels may indicate that remission is no longer driven by the anti-TNF, and therefore that these patients no longer need the drug to maintain the remission state. On the same topic, it is very interesting to note that for patients stopping the immunomodulator and maintaining the anti-TNF, the presence of good anti-TNF trough levels is protective from relapse.56 Therefore, therapeutic drug monitoring may in the future be a powerful tool to select patients for anti-TNF withdrawal.
One very important message that arose from a sub-study of the STORI trial and was later confirmed in another cohort is that clinical relapse is preceded by elevation of inflammatory markers.51,52 In STORI's51 prospective follow-up, 113 patients had bimonthly measurements of CRP and FC; those who later relapsed, presented a marked rise in these inflammatory markers occurring 4 months before relapse. The median value for FC and CRP before relapse was 534μg/g and 8mg/L for relapsers and 66.9μg/g and 3.7mg/L for non-relapsers, respectively.51 These findings have been confirmed in another cohort52, and indicate that in patients stopping anti-TNF, a close follow-up with regular laboratorial assessment, may allow to diagnose relapse even before symptoms occur, offering the possibility for early intervention.
5Can we re-treat after we stop?A reassuring message arising from some studies is that if relapse occurs, re-treatment may be successfully and safely re-instituted. A significant proportion of the patients that relapsed across studies were re-treated. Most studies only provided the rates of clinical remission/response after re-treatment with no further details.30,31,36,41,44,57,58 Also, most studies have only reported short-term results, frequently for the induction therapy phase. Apart from some exceptions,31,36 the short-term remission/response rates to re-treatment (often using the same biologic agent) were generally very good, ranging from 78% to 100% in CD and 54–100% in UC.38,41,47,49 Studies reporting longer follow-up of at least one year reported clinical remission rates between 80 and 92% at one year35,50,53 for CD.
6Unanswered questions and perspectivesThere are many unanswered questions that are worth mentioning. The first and most important is that so far there is no controlled study that can really answer the question on whether maintaining the anti-TNF is superior to maintain remission as opposed to discontinuing the drug. In second place, there is a lack of studies appropriately powered, looking into the possibility of reducing the dose of the anti-TNF once remission has been attained, although this could be an interesting approach for reducing costs and perhaps adverse effects.59 In rheumatoid arthritis for example, a meta-analysis on de-escalation of anti-TNF in randomized and controlled clinical trials in patients with rheumatoid arthritis (RA), showed that while reducing the drug dose (etarnecept data only) was not associated with worse outcomes, stopping the drug (etarnecept or adalimumab data) led to higher rates of clinical activity and worse radiological and functional outcomes.60 In IBD, three small studies have explored this possibility with apparently good results.22,25,61 Third, stopping the anti-TNF when the drug has been used as monotherapy has also not been adequately studied in IBD, which reflects the clinical practice where these agents are often used in combination with an immunomodulator agent. Fourth, it is also very interesting to acknowledge that are no studies specifically studying anti-TNF discontinuation in early disease. Evidence from rheumatology suggests that studies focusing on drug withdrawal after early drug treatment with anti-TNF may be more fruitful.62 Finally, the topic of personalizing discontinuation, taking into account the specific patient risk for complications development needs to be further explored.10
Clearly, the topic of drug de-escalation is a very important one in IBD and warrants further research. A large European Union funded trial (Biocycle project) will soon start and likely solve many of the answered questions.63 This large multicenter project will for the first time study different discontinuation strategies in a controlled fashion. Patients with CD in sustained steroid-free remission on combination therapy will be randomized to continuing therapy, discontinuing the anti-TNF or the immunomodulator. The results of this trial will be then used to inform the concept of treatment cycles in IBD, characterized by periods where both anti-TNF and immunomodulators are administered alternating with periods where either of the drug is withdrawn after remission has been reached. Hopefully this large trial will inform us on how to make the best use of the current therapeutic strategies we currently have, shedding some light into this important question in clinical practice.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that no patient data appear in this article.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
Conflict of interestThe authors have no conflicts of interest to declare.
Author's contributionJT has made substantial contributions to conception and design, acquisition and analysis and interpretation of data and has drafted the article. MC and JFC have revised the manuscript critically for important intellectual content. All authors have provided final approval of the version to be published.