Different measures are recommended to reduce pancreatitis after endoscopic retrograde cholangiopancreatography (ERCP). We conducted a study in patients with ERCP treated with rectal diclofenac or lactated Ringer’s solution, or both interventions, to assess whether there is a decrease in the number of cases of post-ERCP pancreatitis.
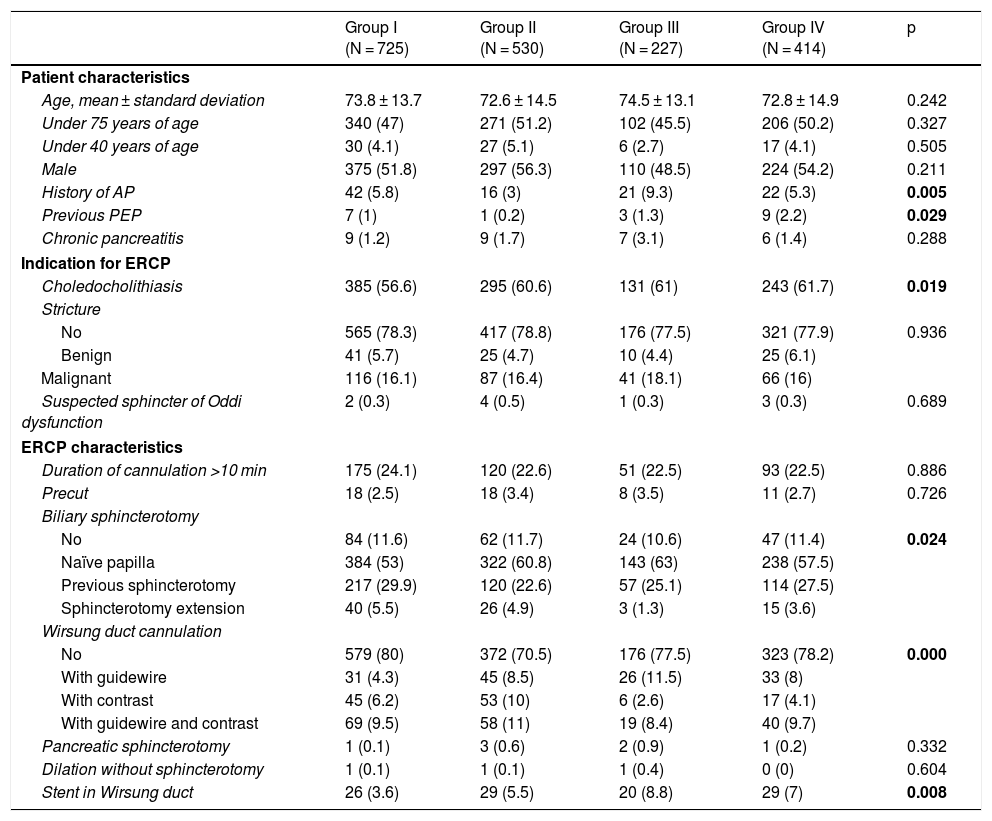
Material and methodsA mixed cohort study involving 1896 patients from 2009 to 2018. Up to June 2012 without treatment (Group I). Subsequently, 100 mg of rectal diclofenac (Group II). Since 2016, lactated Ringer’s solution 200 ml/h during the procedure and 4 h after it, in addition to 500 ml over 30 min when the pancreas was cannulated (Group III). Since 2017, lactated Ringer’s solution plus Diclofenac (Group IV). There were 725 patients in group I, and 530, 227 and 414 patients in groups II, III and IV, respectively. Factors predisposing to post-ERCP pancreatitis and post-ERCP pancreatitis cases that were defined by consensus criteria have been collected.
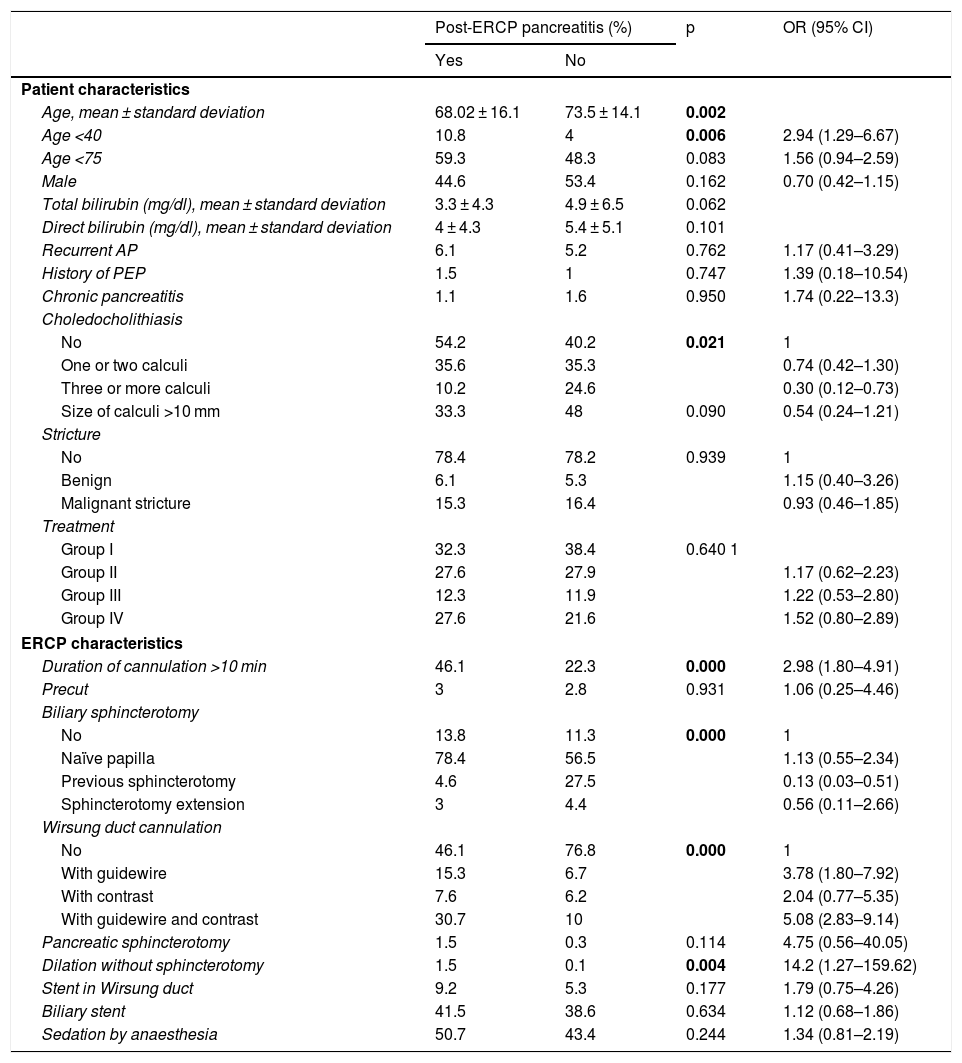
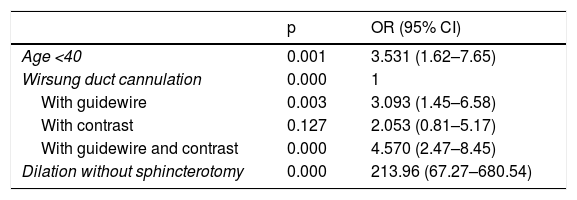
ResultsThere were 65 cases of post-ERCP pancreatitis (3.4%); 2.9%, 3.4%, 3.1% and 4.3% in groups I, II, III and IV, respectively (p = .640). In group I, there was 4.2% of post-ERCP pancreatitis in naïve papillae and 4%, 4.9% and 6.3% in groups II, III and IV, respectively (p = .585). The severity of post-ERCP pancreatitis and adverse effects were similar in all groups. 38.4% were high-risk patients. There were also no differences in post-ERCP pancreatitis in this group (p = .501).
ConclusionIn this work, no benefit was obtained with diclofenac plus hydration in reducing the number and severity of cases of post-ERCP pancreatitis nor with the other prophylactic measures.
Se aconsejan diferentes medidas para disminuir la pancreatitis post-colangiopancreatografía retrógrada endoscópica (PPCPRE). Efectuamos un estudio en pacientes con CPRE tratados con Diclofenaco rectal o Ringer Lactato o bien ambas intervenciones para valorar si existe una disminución en el número de PPCPRE.
Material y métodosEstudio de cohortes mixto con 1.896 pacientes desde 2009 hasta 2018. Hasta junio de 2012 sin tratamiento (grupo I). Posteriormente 100 mg de diclofenaco rectal (grupo II). Desde 2016 Ringer Lactato 200 ml/h durante el procedimiento y 4 h después del mismo, además 500 ml en 30 min cuando se canuló el páncreas (grupo III). Desde 2017 Ringer Lactato más diclofenaco (grupo IV). Hubo 725 pacientes en el grupo i, 530, 227 y 414 pacientes en grupos II, III y IV. Se han recogido factores predisponentes a PPCPRE y los casos de PPCPRE que fue definida por criterios de consenso.
ResultadosHubo 65 PPCPRE (3,4%); 2,9; 3,4; 3,1 y 4,3% en los grupos I, II, III y IV respectivamente (p = 0,640). En el grupo I hubo un 4,2% de PPCPRE en papilas naïve y un 4; 4,9% y 6,3% en los grupos II, III y IV respectivamente (p = 0,585). La gravedad de PPCPRE y los efectos adversos fueron similares en los grupos. El 38,4% eran pacientes de alto riesgo. Tampoco hubo diferencias de PPCPRE en este grupo (p = 0,501).
ConclusiónEn este trabajo no se ha obtenido beneficio con diclofenaco más hidratación en la disminución del número y gravedad de la PPCPRE. Tampoco con las otras medidas profilácticas.