The frequency and patterns of use of scores for the assessment of endoscopic activity in inflammatory bowel disease patients are not known.
AimTo describe the prevalence of adequate use of endoscopic scores in IBD patients who underwent colonoscopy in a real-life setting.
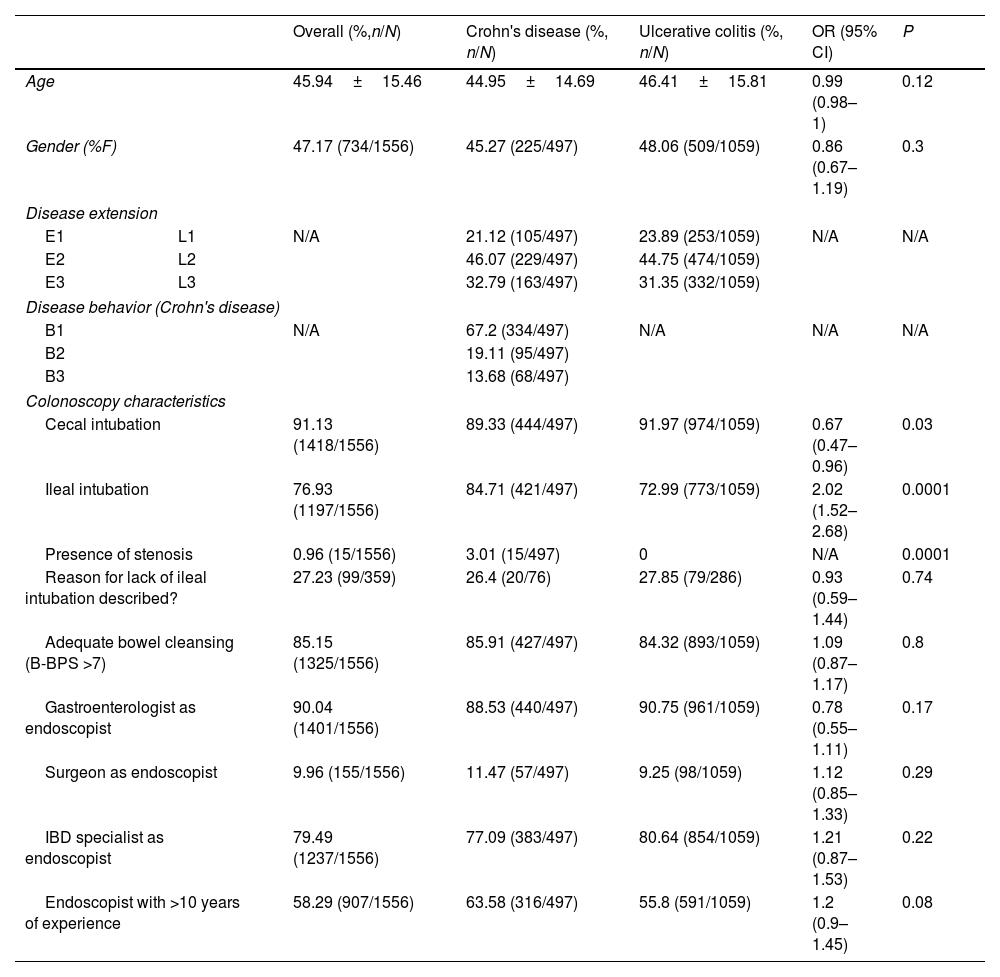
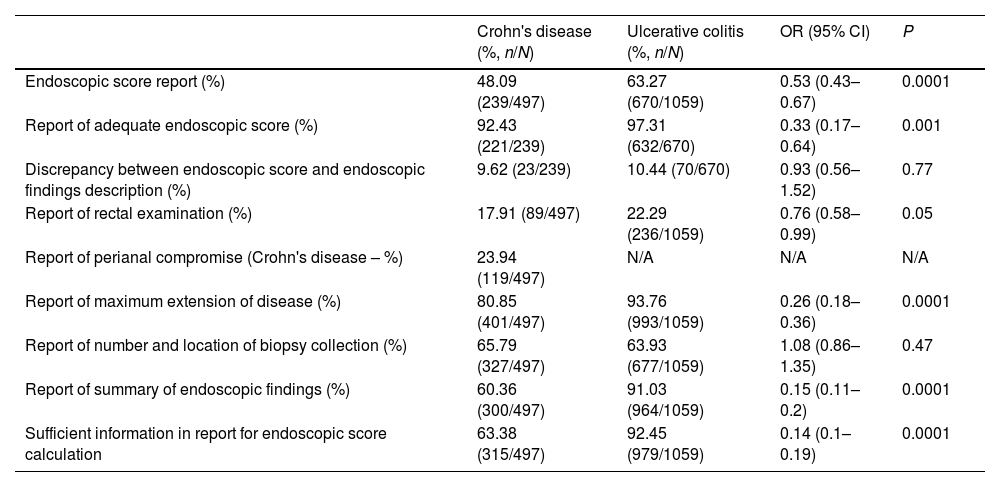
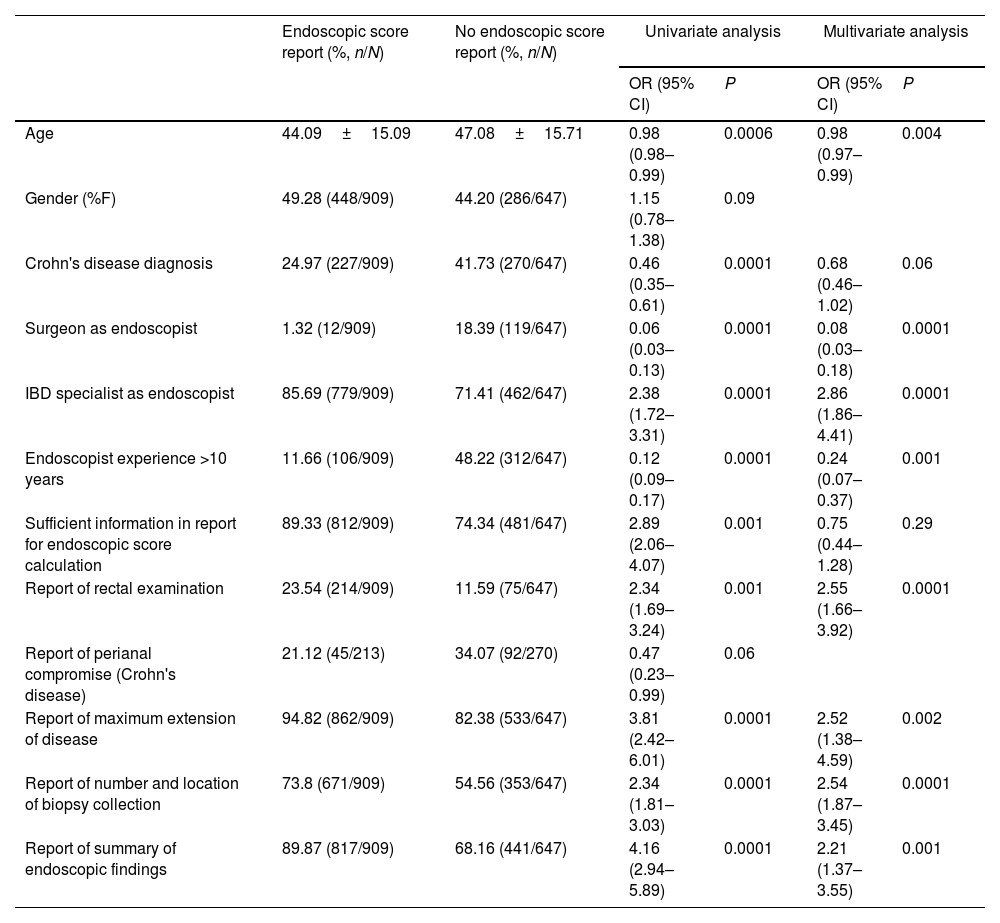
Materials and methodsA multicenter observational study comprising six community hospitals in Argentina was undertaken. Patients with a diagnosis of Crohn's disease or ulcerative colitis who underwent colonoscopy for endoscopic activity assessment between 2018 and 2022 were included. Colonoscopy reports of included subjects were manually reviewed to determine the proportion of colonoscopies that included an endoscopic score report. We determined the proportion of colonoscopy reports that included all of the IBD colonoscopy report quality elements proposed by BRIDGe group. Endoscopist's specialty, years of experience as well as expertise in IBD were assessed.
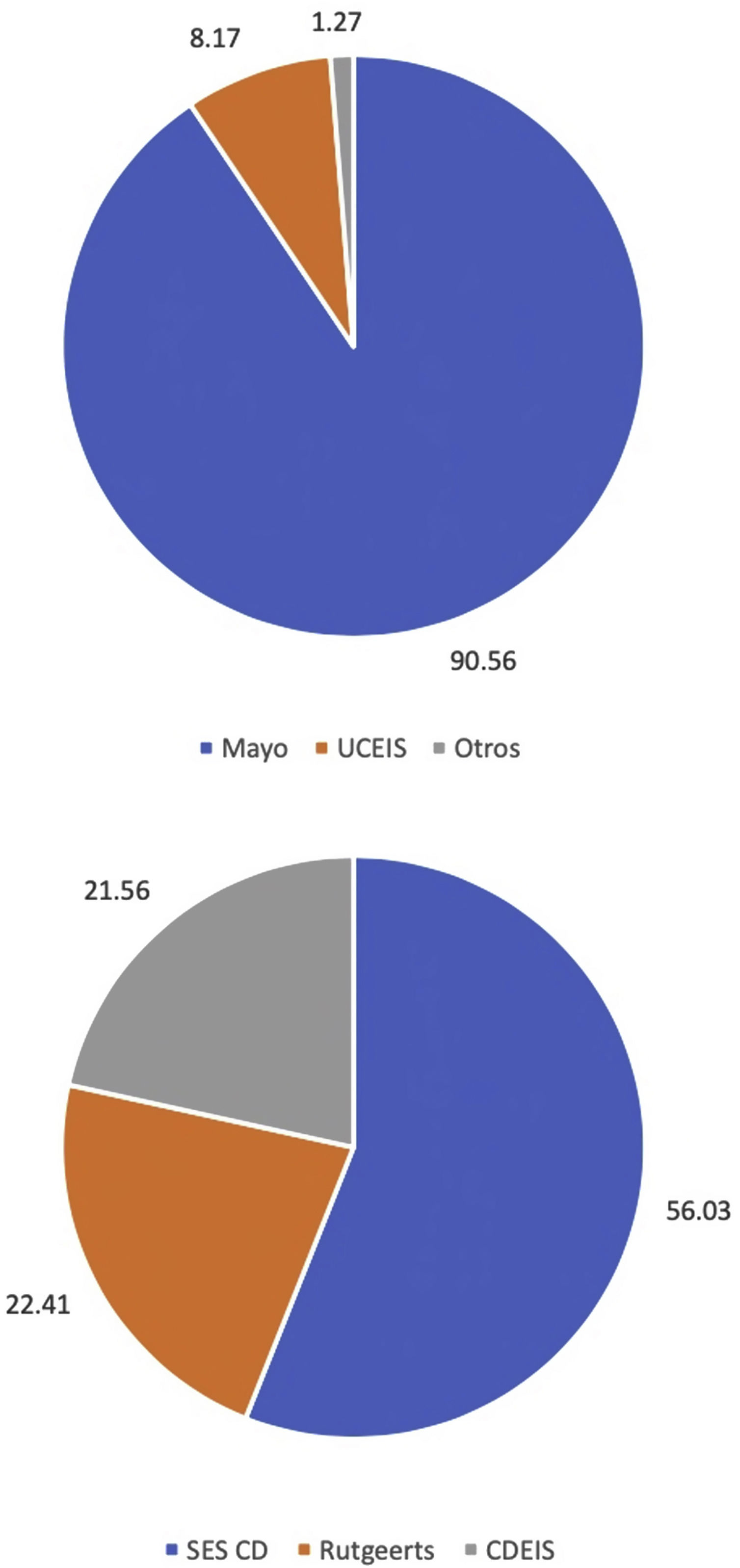
ResultsA total of 1556 patients were included for analysis (31.94% patients with Crohn's disease). Mean age was 45.94±15.46. Endoscopic score reporting was found in 58.41% of colonoscopies. Most frequently used scores were Mayo endoscopic score (90.56%) and SES-CD (56.03%) for ulcerative colitis and Crohn's disease, respectively. In addition, 79.11% of endoscopic reports failed to comply with all recommendations on endoscopic reporting for inflammatory bowel disease.
ConclusionsA significant proportion of endoscopic reports of inflammatory bowel disease patients do not include the description of an endoscopic score to assess mucosal inflammatory activity in a real-world setting. This is also associated with a lack of compliance in recommended criteria for proper endoscopic reporting.
La frecuencia y el patrón de uso de puntajes para evaluar la actividad endoscópica en pacientes con enfermedades inflamatorias intestinales no se conoce bien.
ObjetivoDescribir la prevalencia de uso adecuado de puntajes de actividad endoscópica en colonoscopias de pacientes con enfermedades inflamatorias intestinales en la vida real.
Materiales y métodosSe realizó un estudio multicéntrico observacional en seis hospitales de comunidad en Argentina. Se incluyeron pacientes con enfermedad de Crohn o colitis ulcerosa sometidos a colonoscopia para evaluación de la actividad endoscópica entre 2018 y 2022. Se revisaron los reportes de colonoscopias de sujetos incluidos evaluando la proporción de reportes que incluyeron algún puntaje de actividad endoscópica. Se evaluó la proporción de reportes que incluyeron los elementos de calidad en reporte de colonoscopia en enfermedades inflamatorias intestinales sugeridos por el grupo BRIDGe. Se evaluó la especialidad de los operadores, sus años de experiencia y su experiencia en el manejo de enfermedades inflamatorias intestinales.
ResultadosSe incluyeron 1556 pacientes (31.94% con enfermedad de Crohn). La edad promedio fue de 45.94±15.46. El reporte de algún puntaje endoscópico se identificó en el 58.41% de las colonoscopias. Los puntajes más utilizados fueron el puntaje endoscópico de Mayo (90.56%) y el SES-CD (56.03%). El 79.11% de los reportes de colonoscopias no contaban con todas las recomendaciones para el correcto reporte de colonoscopias en enfermedades inflamatorias intestinales.
ConclusionesUna proporción signifivativa de reportes de colonoscopias en sujetos con enfermedades inflamatorias intestinales no incluyen puntajes endoscópicos de actividad.