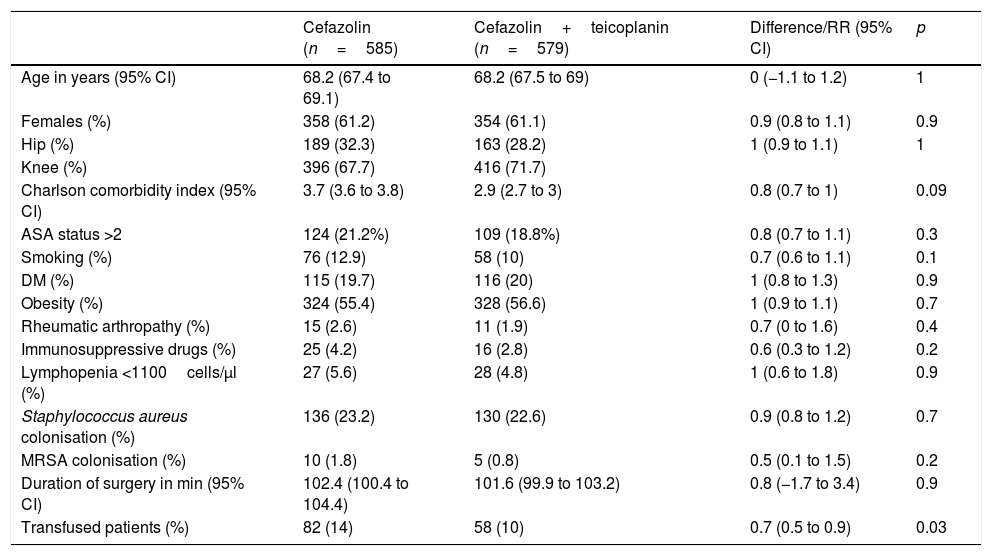
There is a growing increase in prosthetic joint infection (PJI) incidence due to cephalosporin-resistant bacteria, used in surgical prophylaxis. The replacement of these with glycopeptides has not been shown to improve the results, but they have been shown to improve with their combination.
MethodsComparative study of combination of teicoplanin and cefazolin before arthroplasty surgery against cefazolin alone from a previous control group.
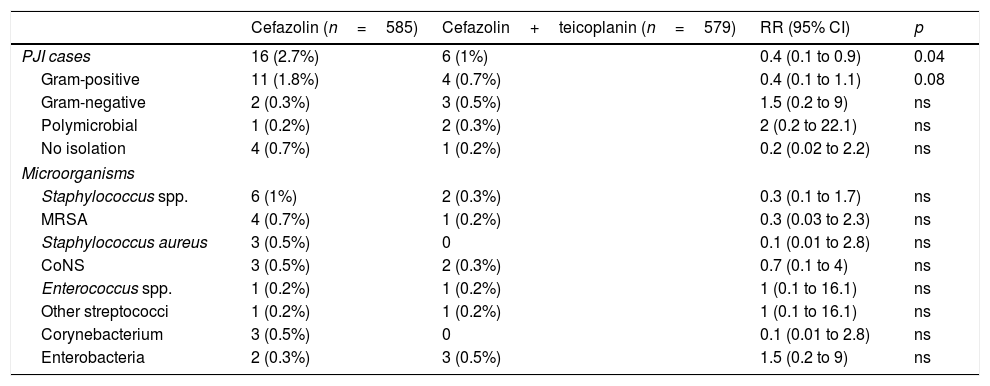
ResultsDuring the control period, there were 16 PJIs from 585 surgeries, while in the intervention group there were 6 from 579 (incidence 2.7% vs. 1.03%, RR 0.4, p=0.04). In control group, 11 of the infections were caused by Gram-positive bacteria vs. 4 in the intervention group (1.8% vs. 0.7%, p=0.08).
ConclusionsThe addition of teicoplanin to cefazolin in the prophylaxis of arthroplasty surgery was associated with a reduction in the incidence of PJI, thanks to a decrease in infections caused by Gram-positive bacteria.
Existe un creciente aumento de las infecciones de prótesis articular (IPA) por bacterias resistentes a las cefalosporinas utilizadas en la profilaxis quirúrgica. La sustitución de estas por glucopéptidos no ha demostrado mejorar los resultados pero sí su asociación.
MétodosEstudio comparativo de la asociación de teicoplanina y cefazolina antes de la cirugía de artroplastia frente a cefazolina sola de un grupo control previo.
ResultadosEn el periodo control hubo 16 IPA de 585 cirugías, mientras que en el grupo de intervención fueron 6 de 579 (incidencia 2,7% vs. 1,03%; RR 0,4, p=0,04). En el grupo control, 11 de las infecciones fueron causadas por bacterias grampositivas frente a 4 en el de intervención (1,8% vs. 0,7%, p=0,08).
ConclusionesLa adición de teicoplanina a cefazolina en la profilaxis de la cirugía de artroplastia se asoció a una reducción de la incidencia de IPA, a expensas de un descenso de las causadas por grampositivos.
Article
Socio de la Sociedad Española de Enfermedades Infecciosas y Microbiología Clínica

Para acceder a la revista
Es necesario que lo haga desde la zona privada de la web de la SEIMC, clique aquí
Para realizar los cursos formativos
La actividad estará abierta para socios de la SEIMC. IMPORTANTE, recuerde que requiere registro previo gratuito. Empezar aquí