Nocardia infection may manifest in multiple ways, representing a significant challenge when it comes to detecting it.
With signs ranging from pulmonary to intracranial lesions, it may require an extensive differential diagnosis with other diseases such as metastatic pulmonary neoplasia, lymphoma, Wegener's syndrome, sarcoidosis, pulmonary aspergillosis and tuberculosis.
A 50-year-old patient with hypertension, diabetes and dyslipidaemia visited the emergency department due to complete amaurosis fugax with a duration of 10 min, followed by full recovery and blurred vision as well as a sensation of loss of vision in the left hemifield, associated with a headache lasting a week. Two weeks earlier, he had presented respiratory infection with left perihilar consolidation for which he had received antibiotic treatment.
He exhibited bradypsychia, inexhaustible horizontal and rotary nystagmus in dextroversion and levoversion with a vertical component in supraversion, left homonymous hemianopia on confrontation and a slightly unstable gait.
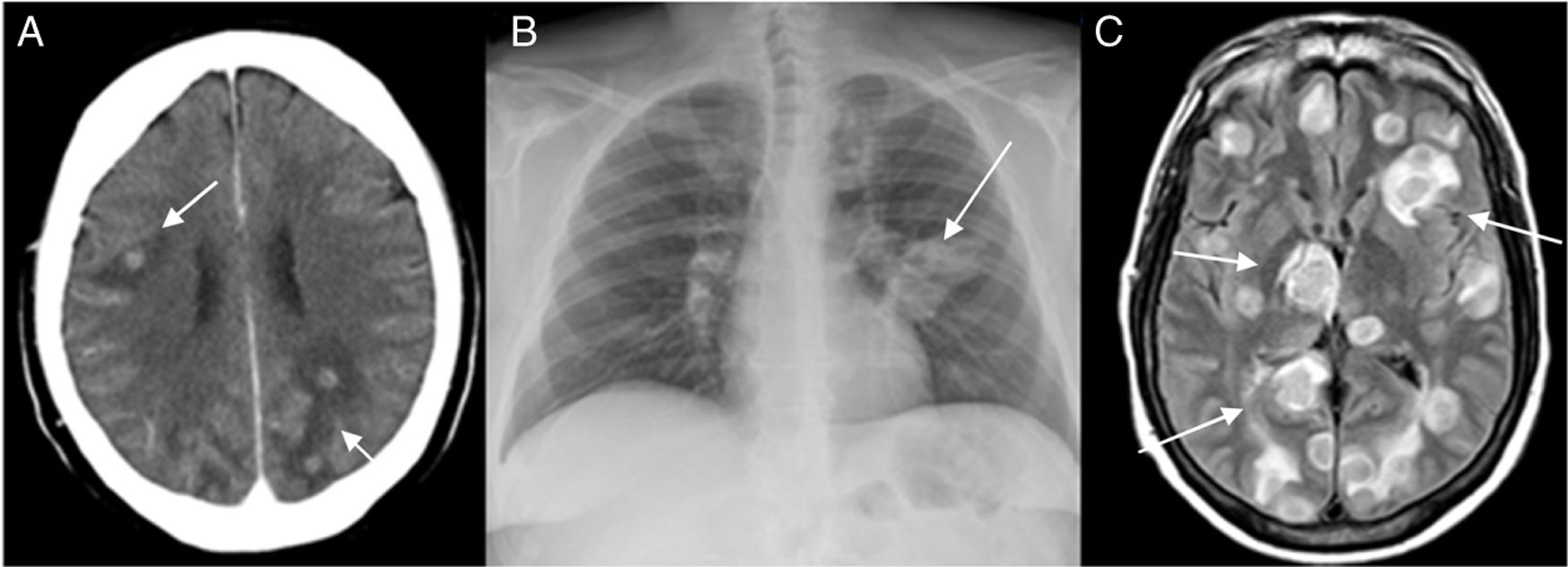
A computed tomography (CT) scan of the head (Fig. 1A) revealed multiple space-occupying lesions with contrast uptake in both cerebral hemispheres, with associated perilesional oedema and midline displacement.
A) Brain CT scan showing juxtacortical lesions (arrows), in both cerebral hemispheres, with perilesional oedema. B) Chest X-ray showing left perihilar opacity (arrow) with probable associated lymphadenopathy. C) Brain MRI scan, FLAIR sequence, showing numerous ring-shaped hyperintense intraparenchymal lesions (arrows) in both cerebral hemispheres, with a hypointense peripheral ring and surrounding oedema.
A chest X-ray confirmed left perihilar opacity with probable associated lymphadenopathy (Fig. 1B), consistent with a primary tumour lesion and secondary brain metastases. The patient was started on intravenous dexamethasone.
A chest CT scan was performed, which revealed a paramediastinal cavitary lesion in the apical segment of the left upper lobe (LUL) measuring 7 cm × 4 cm × 5 cm, with nodular lesions in the LUL and lingula, raising suspicion of tuberculosis. A fibrobronchoscopy was performed in which no macroscopic findings were visualised, and samples were sent for cytology and microbiology.
A brain MRI scan (Fig. 1C) showed intraparenchymal lesions in both cerebral hemispheres, arranged juxtacortically and in the basal ganglia. The lesions had a ring shape, featuring a peripheral ring with FLAIR hyposignal and surrounding oedema, in addition to lesions in the cerebral peduncle and left cerebellar hemisphere.
Microbiological studies for atypical micro-organisms and human immunodeficiency virus (HIV) as well as the QuantiFERON TB® interferon-gamma release assay (IGRA) were negative, and the patient was confirmed to have good blood sugar control with a glycosylated haemoglobin level of 5.2%. Basic immunological studies and measurement of immunoglobulin and complement levels were normal.
Nocardia farcinica sensitive to imipenem, amikacin, levofloxacin, trimethoprim/sulfamethoxazole and linezolid was isolated in a bronchoalveolar lavage culture. As a result, treatment was started with imipenem and trimethoprim/sulfamethoxazole, and the patient's steroid treatment was suspended. In the following 24 h, the patient showed clinical worsening, with a low level of consciousness, lack of response to verbal stimuli and hypotonia and hypoparesis of his left leg. Given the possibility of increased cerebral oedema, as well as the likelihood of a Jarisch–Herxheimer reaction, the patient was put back on the steroid regimen and subsequently followed a favourable clinical course.
The patient completed the cycle of imipenem and trimethoprim/sulfamethoxazole for 3 weeks and his corticosteroids could gradually be reduced. He was then kept on treatment with trimethoprim/sulfamethoxazole for 2 years, with full neurological recovery and pulmonary radiological resolution on a CT scan with minimal residual lesions on a brain MRI scan.
Central nervous system infection with Nocardia is rare, occurring primarily in immunosuppressed patients, though it has been reported in immunocompetent patients. In our case, no apparent risk factor for developing this infection was detected.
Nocardiosis is an infection caused by an aerobic Gram-positive bacterium belonging to the genus Actinomyces.1,2 In most cases, it is contracted by inhalation through the respiratory tract.2 Involvement of the central nervous system is rare. The most common presentation is a single lesion, with a higher rate of mortality compared to other aetiologies of brain abscess (30%).
Most cases reported in the literature correspond to immunosuppressed patients,2 but it has also been reported in immunocompetent patients1,3 and patients with chronic obstructive pulmonary disease.4
Treatment of forms of cerebral involvement should be maintained over a prolonged period of time. N. farcinica usually shows resistance to trimethoprim/sulfamethoxazole, so the antibiogram is essential when adjusting the antibiotic therapy. It is advisable to start treatment with two drugs, subsequently reducing to one of them, and maintain this regimen for a minimum of 6–12 months (at least 12 months in immunosuppressed patients). In serious cases, the addition of a third drug, normally linezolid, can be contemplated.5
It is important to consider nocardiosis, specifically N. farcinica, in patients with pneumonia who follow an unfavourable course with conventional antibiotic treatment,6 especially in pseudotumour and cavitary forms, as well as in differential diagnosis of single or multiple focal cerebral lesions, regardless of whether patients are immunocompetent.
Please cite this article as: Ordieres-Ortega L, Pulfer MD, Cano-Ballesteros JC. Nocardia: la gran simuladora. Enferm Infecc Microbiol Clin. 2021;39:159–160.