Gonococcal infection is a current public health problem worldwide, being the second most prevalent bacterial sexually transmitted infection. The etiologic agent is Neisseria gonorrhoeae, a Gram-negative diplococcus, and mainly causes urethritis in men. In women up to 50% of infections can be asymptomatic. N. gonorrhoeae has a great ability to develop antibiotic resistance, so the last remaining therapeutic option are extended spectrum cephalosporins. Many guides recommend dual therapy with ceftriaxone and azithromycin, but in recent years the resistance to azithromycin is also increasing, so that dual treatment is being questioned by scientific societies.
La infección gonocócica es un problema de salud pública a nivel mundial, siendo la segunda infección de transmisión sexual bacteriana más prevalente. El agente etiológico es Neisseria gonorrhoeae, un diplococo gramnegativo, y causa principalmente uretritis en hombres. En mujeres, hasta un 50% de las infecciones pueden ser asintomáticas. N. gonorrhoeae tiene una gran capacidad de desarrollar resistencia antibiótica, con lo que actualmente la última opción terapéutica son las cefalosporinas de espectro extendido. Muchas guías recomiendan la terapia dual con ceftriaxona y azitromicina, pero en los últimos años la resistencia a esta última también está aumentando, con lo que el tratamiento dual se está poniendo en duda por parte de las sociedades científicas.
Gonococcal infection is caused by Neisseria gonorrhoeae (NG), a strictly aerobic, non-encapsulated, Gram-negative, facultatively intracellular diplococcus. It is an exclusively human primary pathogen which predominantly affects the columnar epithelium of the urethra, the endocervix, the rectum, the pharynx and the conjunctiva of the eyes. Unlike other species of the genus Neisseria, gonococcus is only transmitted by direct contact between mucous membranes (genital–genital, genital–anal, oral–genital or oral–anal) or from mother to child during delivery.
Gonorrhoea is usually localised and produces an intense inflammatory response and an increase in polymorphonuclear leukocytes, causing a purulent discharge, which is very characteristic of gonococcal urethritis.
EpidemiologyGonorrhoea is the second most prevalent sexually transmitted infection (STI) with bacterial aetiology after Chlamydia trachomatis infection. According to data from the European Centre for Disease Prevention and Control, in 2016, 75,349 new cases were reported in 27 European countries, representing a rate of 18.8 cases per 100,000 population and an increase of 53% compared to 2008.1 According to these data, in 2016 in Spain, 6,816 cases were reported, representing a rate of 14.7 per 100,000 population. The 15–24-year-old age group had the highest prevalence, representing 38% of cases, followed by patients aged 25–34 (34%). The cases diagnosed in men who have sex with men accounted for 65% of all male cases detected.
According to data from the Red Nacional de Vigilancia Epidemiológica [Spanish Epidemiological Surveillance Network], 6,456 cases of gonorrhoea were reported in Spain in 2016, representing an incidence of 13.89 cases per 100,000 population, with a continuous increase since 2001, which became more striking from 2012. The number of cases reported in the different autonomous regions varies widely, from 2.04 to 28.97 per 100,000 population. The highest infection rates were recorded in the Balearic Islands (15.82), Madrid (23.58), Asturias (28.14) and Catalonia (28.97).2
Signs and symptoms3–5The signs and symptoms vary according to the site of the infection, and are also often affected by the fact that coinfection with other bacteria is common.
Urethritis3–6Urethritis is symptomatic in around 90% of cases. The incubation period is generally from two to eight days (with a range of one to 14). The clinical signs are acute onset with urethral discharge. Although there may be only a little mucoid discharge to start with, it becomes frankly purulent in 80% of cases within 24h (unlike non-gonococcal urethritis, in which the clinical signs are usually less striking). Half of cases also have dysuria. If the posterior urethra is involved there may be tenesmus, terminal haematuria and urethral pain.
On examination, in addition to the exudate, there is usually erythema and oedema of the urethral meatus. Uncircumcised males may have balanitis. The urethral discharge becomes most evident from 2h since the last urination, and that is the minimum time indicated for adequate sampling. If no exudate is apparent during examination, it can be obtained by gently compressing the urethra.
The infection can affect the median raphe, the Tyson glands, the paraurethral glands and periurethral or Littre glands, and can cause abscesses in the trunk of the penis, stenosis and periurethral fistulas. Submucosal involvement of the urethral wall, the appearance of lymphangitis and thrombophlebitis may also occur. The most significant complications, caused by transluminal dissemination from the urethra, are orchiepididymitis, prostatitis and seminal vesiculitis, which can progress to chronic prostatitis. Orchiepididymitis is usually unilateral and it is important to differentiate it from testicular torsion, a very acute condition which is more common in younger people. Prostatitis and seminal vesiculitis manifest with fever, malaise, perineal discomfort, tenesmus, suprapubic pain, urinary retention or urgency, haematuria and painful ejaculation. Possible inflammation and abscess formation due to involvement of the bulbourethral or Cowper's glands is also accompanied by fever, perineal pain when moving the bowels and urinary frequency or retention.
Cervicitis3–5,7,8The infection affects the columnar epithelium of the cervix and can reach areas of confluence with the squamous epithelium, but not the vagina, which is covered with squamous epithelium. It is asymptomatic or almost asymptomatic in 50% of cases. When there are symptoms, they are more acute and intense than those caused by chlamydia. After an incubation period of about 10 days, it usually leads to mucopurulent vaginal discharge (sometimes due to coinfection), dysuria (usually without frequency), when there is involvement of the urethral epithelium, hypogastric pain in 25% of cases and, less commonly, metrorrhagia or menorrhagia.
There may be no findings on examination, but in almost half of cases there will be mucopurulent cervical exudate or a friable cervix, which bleeds easily when taking a smear. Occasionally the pelvic examination may be slightly painful.
As complications, the infection can affect the Skene glands and the Bartholin glands, causing bartholinitis and abscesses, usually unilateral, which may drain spontaneously. In up to 14% of untreated cases, the infection can extend upwards and cause pelvic inflammatory disease (PID), which includes endometritis, salpingitis and peritonitis, often simultaneous and clinically indistinguishable from one another, causing leucorrhoea, metrorrhagia, menorrhagia, hypogastric pain, pyrexia and leucocytosis.8 In half of cases of PID, there are other bacteria involved such as C. trachomatis and anaerobes. Tubal involvement can cause obstruction in up to 20% of cases and, as a result, infertility or ectopic pregnancy. Perihepatitis or Fitz-Hugh-Curtis syndrome, although it has also been associated with lymphatic or haematogenous spread, is generally associated with PID.
Anorectal infection3–5,9,10In women it could be the result of colonisation from vaginal secretions from cervicitis, but it is rare in people who have not had anal sex. It is asymptomatic in 90% of cases, but 10% may have proctitis (pain, discomfort or anorectal pruritus, purulent discharge and, less often, tenesmus, constipation or anal bleeding). Anoscopy may show oedema, erythema and/or friability of the mucosa with a mucopurulent exudate.
Oropharyngeal infection3–5,9,10It is acquired through orogenital contact. It is usually asymptomatic, but occasionally it can cause tonsillitis and/or lymphadenopathy. Pharyngeal involvement may subside, but it has also been related to the haematogenous spread of the infection. The diagnosis is usually made by chance during an STI screen or in the contact investigation for a case of gonorrhoea.
Conjunctivitis3–5This is rare in adults but presents with inflammation and purulent exudate in one or both eyes. Without treatment, it can be complicated by keratitis and panophthalmitis, which can cause blindness.
Systemic complications3–5,11,12Haematogenous spread of infection can occur in up to 3% of cases. It is more common in women and is associated with menstruation, the final trimester of pregnancy and pharyngeal location of the infection. It has been associated with strains with greater resistance to the bactericidal action of the serum. It can present with fever, arthralgia, arthritis and skin lesions due to septic embolisms (arthritis-dermatitis syndrome). Skin lesions, present in 60% of cases, appear early, are painless and manifest on the extremities (particularly hands, feet and the distal third of the limbs) in the form of macules, which progress to erythematous papules, vesicles on a purpuric base, necrotic pustules surrounded by erythema or petechiae. Joint involvement begins with tenosynovitis, which asymmetrically affects primarily the wrists, ankles, fingers and toes. Arthritis is the most common disorder; it is usually limited to one type of joint, predominating in the knees, elbows, wrists, ankles or metacarpophalangeal joints. Cardiac involvement (which can cause severe aortic endocarditis, myocarditis or pericarditis), perihepatitis (Fitz-Hugh-Curtis syndrome) and meningitis (clinically similar to meningococcal meningitis) are very rare.
Gonococcal infection in children4,13The most common cause of gonococcal infection in children is sexual abuse. Theoretically, genital infection in girls could be acquired accidentally in conditions of poor hygiene, but both urethral infection in boys and pharyngeal and rectal infection in boys and girls are virtually always considered to be caused by sexual abuse. In girls, as the vagina has columnar epithelium, the genital infection manifests as vaginitis with purulent vaginal discharge and on rare occasions is complicated by PID. In boys, the signs and symptoms are the same as in adult urethritis.
Gonococcal infection in neonatesThis is caused by perinatal exposure to the mother's infected cervix. Onset is acute two to five days after birth. The most serious manifestations are ophthalmia neonatorum and, more rarely, sepsis with arthritis and/or meningitis. Electrodes introduced through the cervix for monitoring the foetus can cause localised infection on the scalp in the form of abscesses.
Ophthalmia neonatorum is a type of conjunctivitis of the newborn which is acquired as it passes through the birth canal. It manifests two to five days after birth with abundant purulent exudate, palpebral inflammation or even periorbital cellulitis, keratitis and perforation of the eyeball, which can lead to blindness. Prophylaxis consists of administering 0.5% erythromycin eye drops or ointment (or 1% tetracycline eye ointment or 1% silver nitrate eye drops) to each eye in the newborn after delivery, whether vaginal or caesarean.
Microbiological diagnosisNG is very sensitive to environmental conditions: the way samples are taken, transported and processed is therefore particularly important in gonorrhoea, especially for culture. These days, swabs soaked in Stuart/Amies-type liquid transport medium have a high percentage of recovery, as the short perpendicularly-arranged fibres guarantee maximum absorption of the sample and good elution in the liquid transport medium. It was long thought that gonococcus survival was superior at room temperature compared to refrigeration and the recommendations offered by many textbooks are contradictory. A recent study shows that the preservation and survival of gonococcus is much better if the swabs with transport medium are kept refrigerated (4°C).14
The main diagnostic tools are as follows15:
- (a)
Microscopic examination by Gram staining or methylene blue staining. Observation at 1,000× magnification of ≥2 polymorphonuclear cells per field and Gram-negative diplococci allows rapid diagnosis of gonococcal urethritis with good sensitivity (>95%) and specificity. In contrast, the sensitivity of Gram staining in asymptomatic men, endocervical exudate and rectal and pharyngeal exudates is low and it is not therefore useful for ruling out these infections.
- (b)
Culture. The definitive diagnosis can be made through isolation and identification of the microorganism. Culture has high sensitivity and specificity at a low cost. It is also the only diagnostic test with which antimicrobial sensitivity studies can be carried out, so it is essential for detecting and monitoring antimicrobial resistance. Culture is suitable for endocervical, urethral, first-void urine, rectal, pharyngeal and conjunctival samples. It is also the recommended technique for persistent infection or suspected treatment failure.
The use of selective media containing antimicrobials such as Thayer-Martin, Martin-Lewis or New York City media is recommended. Plates should be incubated for 24–72h at 35–37°C in a 5% CO2 atmosphere. The presumptive identification is made by observing the morphology of the colony (greyish colour), the Gram staining of the colony (Gram-negative diplococci), the positive oxidase test and the positive catalase test (3% hydrogen peroxide). The definitive identification is made with presumptive identification tests and one or more techniques which demonstrate the carbohydrate utilisation patterns (for example, the API NH system) and the immunological characteristics or enzymatic profiles of the microorganisms. Mass spectrometry (MALDI-TOF) is currently a very useful tool for definitive identification. Nucleic acid amplification methods also have high sensitivity and specificity. Where disseminated infection is suspected, a blood sample should be taken for blood cultures. They must contain a sodium polyanethol sulfonate concentration no higher than 0.025%, as gonococci are sensitive to this compound. Sterile fluid samples (CSF, joint fluid) should be centrifuged (>1ml) at 1,500×g for 15min and the sediment cultured.
- (c)
Nucleic acid amplification tests (NAAT). NAAT are currently recommended for the detection of urogenital infections in females and males with or without symptoms, as they have been shown to be cost-effective. The advantage of these techniques is that they detect non-viable microorganisms, thus increasing sensitivity and facilitating the collection, transport and processing of samples. Their high sensitivity has allowed the use of less invasive samples, such as the first-void urine in men and vaginal smear in women.16 This facilitates self-collection of the sample by the patient, a procedure used in many cases in screening. It is important to note that the sensitivity of NAAT in women's urine may be 10% less than in vaginal discharge. The specificity of NAAT is lower in extragenital samples and false positives are possible. If a false positive is suspected, testing must be repeated with another method.
Although NAAT are not approved by the Food and Drug Administration as a test of cure, they can be used for this purpose, as long as it is borne in mind that residual DNA from the primary infection may be detected. In the case of gonorrhoea, it is recommended to wait two weeks from the end of treatment before testing for cure using NAAT.16,17
- (d)
Others: serology and enzyme immunoassays are not useful for the diagnosis of gonorrhoea.
Since the introduction of antimicrobials for the treatment of gonorrhoea in the early 20th century, NG has developed resistance to all those used.18
NG develops resistance to antibiotics through all the known mechanisms: (a) modification or destruction of the antimicrobial molecule through an enzymatic process; (b) modification of the therapeutic target; (c) decrease in penetration of the antibiotic into the cell; and (d) increase in expulsion out of the cell. It has also demonstrated a great capacity for horizontal transfer of genetic material. This is very significant, as it is known that pharyngeal gonorrhoea can act as a source of resistance due to the acquisition of resistance genes from other saprophytic species of Neisseria in the oropharynx. Moreover, the acquisition of resistance determinants does not affect the biological fitness of the microorganism, which is why multi-resistant strains persist even in the absence of antibiotic pressure.19
Sulfonamides, penicillins, tetracyclines, macrolides and fluoroquinolones are no longer options for empirical treatment due to the high percentage of resistance detected worldwide. Extended-spectrum cephalosporins (cefixime and ceftriaxone) are the last option for first-line empirical treatment, but over the last twenty years or so strains resistant to these agents have also been detected all over the world,20–23 meaning that gonorrhoea could become an untreatable infection. Also very alarming was the detection in early 2018 of the first case of treatment failure on dual therapy with ceftriaxone and azithromycin.24
In 2016, according to the data published by the European Gonococcal Antimicrobial Surveillance Programme (Euro-GASP),25 2,660 isolated strains of gonococci were studied in 25 participating countries. No isolates with resistance to ceftriaxone were detected, 2.1% showed resistance to cefixime, 7.5% to azithromycin and 46.5% showed resistance to ciprofloxacin.
The data published in Spain are very limited. In 2011, Serra-Pladevall et al. studied the sensitivity of 100 strains isolated in Barcelona and found 3% resistance to ceftriaxone and 10% to cefixime.26 The same group studied 1,355 strains of NG from August 2012 to July 2016 and found that the rate of resistance to penicillin over these years ranged from 20% to 30%. The rate of resistance to ceftriaxone was low (0–2.8%). In contrast, 8.3% of strains isolated in 2013 were resistant to cefixime. Around 50% showed resistance to ciprofloxacin and 3–5.3% to azithromycin.27
Consequently, it is very important to diagnose the infection using cultures in order to isolate the microorganism and perform the antibiogram. This allows resistance to be monitored at a local level and treatment guidelines to be adjusted, as the World Health Organisation recommends updating these guidelines if the resistance rate to the antibiotics used exceeds 5%.28
TreatmentThe aim of treatment is to eliminate the infection, avoid complications and reduce the length of time with capacity of transmission to sexual partners and newborns in pregnant women. Treatment therefore has both a clinical and public health benefit, which justifies starting an empirical treatment when urethritis is diagnosed.
The initial treatment of urethritis should cover gonorrhoea and chlamydia (main causes of urethritis). However, except in cases with high probability of infection and risk that the patient will not return for the results, treatment should not be started unless there are clear clinical signs and laboratory criteria for urethritis are met.
Given the high diagnostic sensitivity of Gram staining, the observation of intracellular Gram-negative diplococci in urethral samples is sufficient to start a specific treatment for gonorrhoea without the need to wait for microbiological confirmation by culture or PCR.
It is recommended that the treatment be carried out in situ in order to ensure adherence.
In terms of the recommended initial regimen, the continual development of antibiotic resistance means that frequent changes have had to be made to the therapeutic guidelines all over the world. Quinolones, widely used in the early 21st century, should not now be used in the initial treatment of gonorrhoea without a prior antibiogram confirming sensitivity. More recently, a gradual reduction in sensitivity to cephalosporins led to a change in strategy with the intention of delaying the development and spread of antibiotic resistance. Since 2011, all guidelines recommend a dual regimen using two antibiotics with different mechanisms of action to increase the effectiveness of the treatment. An increase in the drug dose that enables a sustained high level of bactericidal activity in plasma is the second key point of this strategy. The regimens currently recommended by reference national and international scientific bodies and societies are shown in Tables 1–6.17,29–33. All recommend the combination of ceftriaxone and azithromycin as initial therapy of choice for gonorrhoea, at doses which vary according to the epidemiology of antibiotic resistance in the region and gastrointestinal tolerance to the different doses of azithromycin. The Centers for Disease Control and Prevention guidelines recommend lower doses of cephalosporins, as in the United States the MIC are at more favourable levels than in Europe in general and in Spain in particular.25–27,34 It is worth mentioning the lack of studies from which to make sufficiently solid treatment recommendations aimed at delaying the emergence of resistance and confirming the real benefits of a dual therapy. No synergistic effect between ceftriaxone and azithromycin has been convincingly demonstrated. Azithromycin at the dose of 1g may be insufficient to eliminate the infection and at 2g, in addition to worse gastrointestinal tolerance, it may be ineffective against gonococci with high resistance to azithromycin. There are also data suggesting that the use of azithromycin can induce and propagate resistance to Treponema pallidum and Mycoplasma genitalium. Although most infections are curable with the currently recommended doses, a higher dose of ceftriaxone would ensure the eradication of strains with increased MIC by obtaining sustained plasma levels sufficiently above the MIC. The British Association for Sexual Health and HIV has recently issued a new draft on the treatment of gonorrhoea and states that ceftriaxone monotherapy at a dose of 1g may be the best option in the initial treatment of uncomplicated gonorrhoea.35 Other scientific bodies and societies, such as the International Union against Sexually Transmitted Infections, are in the process of reviewing their recommendations.
Treatment recommendations for uncomplicated gonorrhoea in urethra, cervix or rectum.
| GESIDA, SPNS, GeITS, AEDV and SEIP, 2017 | BASHH, 2011 | IUSTI Europe, EADV, EDF, UEMS, ECDC and WHO-Europe, 2012 | CDC, 2015 | |
|---|---|---|---|---|
| Of choice | IM ceftriaxone 500mg SD+oral azithromycin 1–2g SD | IM ceftriaxone 500mg SD+oral azithromycin 1g SD | IM ceftriaxone 500mg SD+oral azithromycin 2g SD | IM ceftriaxone 250mg SD+oral azithromycin 1g SD |
| Alternatives | Allergy to cephalosporins:IM gentamicin 240mg SD+oral azithromycin 2g SDororal gemifloxacin 320mg SD+oral azithromycin 2g SDResistance to cephalosporins:IM ceftriaxone 1g SD+oral azithromycin 2g SDorIM gentamicin 240mg SD+oral azithromycin 2g SDRejection, IM contraindicated or non-availability of ceftriaxone:Oral cefixime 400mg SD+oral azithromycin 2g SDPregnancy:IM ceftriaxone 250mg SD+oral azithromycin 1g SD | Allergy to cephalosporins:IM Spectinomycin 2g+oral azithromycin 1g SDorIM gentamicin 240mg SD+oral azithromycin 2g SDorOral gemifloxacina 320mg SD+oral azithromycin 2g SDResistance to cephalosporins:IM ceftriaxone 1g SD+oral azithromycin 2g SDorIM gentamicin 240mg SD+oral azithromycin 2g SDRejection, IM contraindicated or non-availability of ceftriaxone:Oral cefixime 400mg SD+oral azithromycin 1g SDOther:IM Spectinomycin 2g+oral azithromycin 1g SDorIM cefotaxime 500mg SD or IM cefoxitin 2g SD with 1g of oral probenecid ororal cefpodoxime 200–400mg SD+oral azithromycin 1g SDSensitive to quinolones:Oral ciprofloxacin 500mg SD or oral ofloxacin 400mg SDSensitive to macrolides:Oral azithromycin 2g SDPregnancy:IM ceftriaxone 500mg SD+oral azithromycin 1g SDorIM spectinomycin 2g+oral azithromycin 1g SD | Allergy or resistance to cephalosporins:IM Spectinomycin 2g+oral azithromycin 2g SDSensitive to quinolones:Oral ciprofloxacin 500mg SD or oral ofloxacin 400mg SDSensitive to macrolides:Oral azithromycin 2g SDResistance to cephalosporins:IM ceftriaxone 1g SD+oral azithromycin 2g SDorIM gentamicin 240mg SD+oral azithromycin 2g SDRejection, IM contraindicated or non-availability of ceftriaxone:Oral cefixime 400mg SD+oral azithromycin 2g SDAzithromycin not available or unable to take:IM ceftriaxone 500mg SD | Allergy or resistance to cephalosporins:Oral gemifloxacin 320mg SD+oral azithromycin 2g SDorIM gentamicin 240mg+oral azithromycin 2g SDOther option when allergic to cephalosporins:IM Spectinomycin 2gAllergic to azithromycin:Doxycycline 100mg/12h 7 days with ceftriaxone or cefiximeNon-availability of ceftriaxone:Oral cefixime 400mg SD+oral azithromycin 1g SD (oral doxycycline 100mg/12h 7 days if allergic to azithromycin)Pregnancy:IM ceftriaxone 500mg SD+oral azithromycin 1g SDorIM spectinomycin 2g |
AEDV: Academia Española de Dermatología y Venereología [Spanish Academy of Dermatology and Venereology]; BASSH: British Association for Sexual Health and HIV; CDC: Centers for Disease Control and Prevention; ECDC: European Centre for Disease Prevention and Control; EDF: European Dermatology Forum; GeITS: Grupo de Estudio de ITS de la SEIMC (Sociedad Española de Enfermedades Infecciosas y Microbiología Clínica) [Spanish Society of Infectious Diseases and Clinical Microbiology STI Study Group]; GESIDA: Grupo de Estudio del SIDA de la SEIMC [SEIMC AIDS Study Group]; IM: intramuscular; IUSTI: International Union against Sexually Transmitted Infection; SD: single dose; SEIP: Sociedad Española de Infectiología Pediátrica [Spanish Society of Paediatric Infectious Diseases]; SPNS: Secretaría del Plan Nacional sobre el SIDA [Secretariat for the National Plan on AIDS]; UEMS: European Union of Medical Specialists; WHO: World Health Organisation.
Azithromycin, gentamicin and spectinomycin monotherapy can facilitate the emergence of resistance. Spectinomycin does not guarantee the eradication of pharyngeal gonorrhoea and is not presently available in Spain. Category B of the Food and Drug Administration regarding pregnancy, but use with caution during breastfeeding due to lack of information about its effect. There is a possibility of treatment failure with azithromycin and it is poorly tolerated at the dose of 2g. Cefixime should only be used if the intramuscular route is refused or contraindicated. The other cephalosporins offer no advantages over ceftriaxone or cefixime.
Treatment recommendations in gonococcal infection of the pharynx in adults and adolescents.
| IUSTI Europe, EADV, EDF, UEMS, ECDC and WHO-Europe, 2012 | BASHH, 2011 | CDC, 2015 | |
|---|---|---|---|
| Of choice | IM ceftriaxone 500mg SD+oral azithromycin 2g SD | IM ceftriaxone 500mg SD+oral azithromycin 1g SD (IV, C) | IM ceftriaxone 250mg SD+oral azithromycin 1g SD |
| Alternative | Sensitive to quinolones:Oral ciprofloxacin 500mg SD or oral ofloxacin 400mg SDSensitive to macrolides:Oral azithromycin 2g SDNot available or impossible to take orallyIM ceftriaxone 500mg SD in monotherapy | Sensitive to quinolonesOral ciprofloxacin 500mg SD or oral ofloxacin 400mg SD or oral azithromycin 2g SD |
AEDV: Academia Española de Dermatología y Venereología [Spanish Academy of Dermatology and Venereology]; BASSH: British Association for Sexual Health and HIV; CDC: Centers for Disease Control and Prevention; ECDC: European Centre for Disease Prevention and Control; EDF: European Dermatology Forum; IM: intramuscular; IUSTI: International Union against Sexually Transmitted Infection; SD: single dose; UEMS: European Union of Medical Specialists; WHO: World Health Organisation.
Spectinomycin has limited efficacy and it is not recommended.
Treatment recommendations in gonococcal orchiepididymitis and pelvic inflammatory disease.
| IUSTI Europe, EADV, EDF, UEMS, ECDC and WHO-Europe, 2012 | BASHH, 2011 | ||
|---|---|---|---|
| Orchiepididymitis | Of choice | IM ceftriaxone 500mg SD+oral doxycycline 100mg/12h 10–14 days | IM ceftriaxone 500mg SD+oral doxycycline 100mg/12h 10–14 days |
| Alternative | Alternative to ceftriaxone if sensitive to quinolones:Oral ciprofloxacin 500mg SD | ||
| Pelvic inflammatory disease | IM ceftriaxone 500mg single dose+oral doxycycline 100mg/12h 14 days+oral metronidazole 400mg/12h 14 days | IM ceftriaxone 500mg SD+oral doxycycline 100mg/12h 14 days+oral metronidazole 400mg/12h 14 days |
AEDV: Academia Española de Dermatología y Venereología [Spanish Academy of Dermatology and Venereology]; BASSH: British Association for Sexual Health and HIV; ECDC: European Centre for Disease Prevention and Control; EDF: European Dermatology Forum; IM: intramuscular; IUSTI: International Union against Sexually Transmitted Infection; SD: single dose; UEMS: European Union of Medical Specialists; WHO: World Health Organisation.
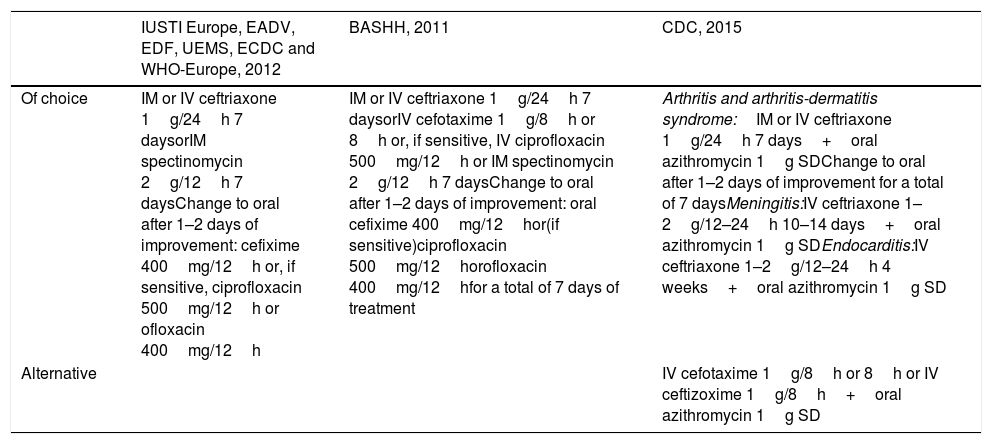
Treatment recommendations in disseminated gonococcal infection.
| IUSTI Europe, EADV, EDF, UEMS, ECDC and WHO-Europe, 2012 | BASHH, 2011 | CDC, 2015 | |
|---|---|---|---|
| Of choice | IM or IV ceftriaxone 1g/24h 7 daysorIM spectinomycin 2g/12h 7 daysChange to oral after 1–2 days of improvement: cefixime 400mg/12h or, if sensitive, ciprofloxacin 500mg/12h or ofloxacin 400mg/12h | IM or IV ceftriaxone 1g/24h 7 daysorIV cefotaxime 1g/8h or 8h or, if sensitive, IV ciprofloxacin 500mg/12h or IM spectinomycin 2g/12h 7 daysChange to oral after 1–2 days of improvement: oral cefixime 400mg/12hor(if sensitive)ciprofloxacin 500mg/12horofloxacin 400mg/12hfor a total of 7 days of treatment | Arthritis and arthritis-dermatitis syndrome:IM or IV ceftriaxone 1g/24h 7 days+oral azithromycin 1g SDChange to oral after 1–2 days of improvement for a total of 7 daysMeningitis:IV ceftriaxone 1–2g/12–24h 10–14 days+oral azithromycin 1g SDEndocarditis:IV ceftriaxone 1–2g/12–24h 4 weeks+oral azithromycin 1g SD |
| Alternative | IV cefotaxime 1g/8h or 8h or IV ceftizoxime 1g/8h+oral azithromycin 1g SD |
AEDV: Academia Española de Dermatología y Venereología [Spanish Academy of Dermatology and Venereology]; BASSH: British Association for Sexual Health and HIV; CDC: Centers for Disease Control and Prevention; ECDC: European Centre for Disease Prevention and Control; EDF: European Dermatology Forum; IM: intramuscular; IUSTI: International Union against Sexually Transmitted Infection; IV: intravenous; SD: single dose; UEMS: European Union of Medical Specialists; WHO: World Health Organisation.
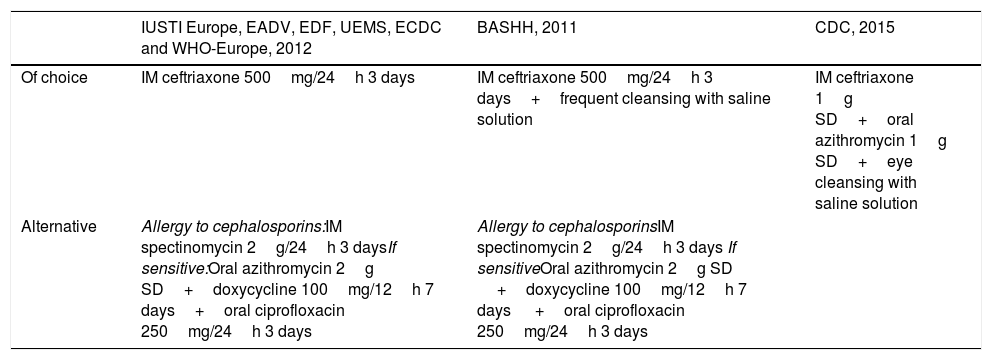
Treatment recommendations for gonococcal conjunctivitis in adults.
| IUSTI Europe, EADV, EDF, UEMS, ECDC and WHO-Europe, 2012 | BASHH, 2011 | CDC, 2015 | |
|---|---|---|---|
| Of choice | IM ceftriaxone 500mg/24h 3 days | IM ceftriaxone 500mg/24h 3 days+frequent cleansing with saline solution | IM ceftriaxone 1g SD+oral azithromycin 1g SD+eye cleansing with saline solution |
| Alternative | Allergy to cephalosporins:IM spectinomycin 2g/24h 3 daysIf sensitive:Oral azithromycin 2g SD+doxycycline 100mg/12h 7 days+oral ciprofloxacin 250mg/24h 3 days | Allergy to cephalosporinsIM spectinomycin 2g/24h 3 days If sensitiveOral azithromycin 2g SD +doxycycline 100mg/12h 7 days +oral ciprofloxacin 250mg/24h 3 days |
AEDV: Academia Española de Dermatología y Venereología [Spanish Academy of Dermatology and Venereology]; BASSH: British Association for Sexual Health and HIV; CDC: Centers for Disease Control and Prevention; ECDC: European Centre for Disease Prevention and Control; EDF: European Dermatology Forum; IM: intramuscular; IUSTI: International Union against Sexually Transmitted Infection; SD: single dose; UEMS: European Union of Medical Specialists; WHO: World Health Organisation.
Treatment recommendations for gonococcal infection in children.
| IUSTI Europe, EADV, EDF, UEMS, ECDC and WHO-Europe, 2012 | BASHH, 2013 | CDC (2015) GESIDA, SPNS, GeITS, AEDV and SEIP, 2017 | ||||
|---|---|---|---|---|---|---|
| Ophthalmia neonatorum | IM or IV ceftriaxone 25–50mg/kg (max. 125mg) SD and cleansing with sterile saline solution | IM or IV ceftriaxone 25–50mg/kg (max. 125mg) SD Assess and treat mothers and their partners | ||||
| Disseminated gonococcal infection and scalp abscesses in the newborn | IM or IV ceftriaxone 25–50mg/kg/24h (max. 125mg) 7 days (10–14 if meningitis) or IM or IV cefotaxime 25mg/kg/12h 7 days (10–14 if meningitis) Assess and treat mothers and their partners | |||||
| Uncomplicated vulvovaginitis, cervicitis, urethritis, pharyngitis or proctitis in children | <45kg (1 month–2 years) | >45kg (2–12 years) | >12 years | <45kg | >45kg | |
| IM ceftriaxone 125mg SD oIM spectinomycin 40mg/kg SD | IM ceftriaxone 250mg/kg SD or IM spectinomycin 40mg/kg SD | As in adults (except ciprofloxacin in still-growing children) | IM or IV ceftriaxone 25–50mg/kg (max. 125mg) SD | IM ceftriaxone 250mg/kg SD+oral azithromycin 1g SD or oral cefixime 400mg SD+oral azithromycin 1g SD | ||
| Pelvic inflammatory disease | <45kg (2–12 years) | >12 years | ||||
| IM ceftriaxone 125mg SD followed by 2 weeks of oral erythromycin 250mg/12h+oral metronidazole (dose according to weight) | As in adults (oral cefixime 400mg as alternative if IM is refused) | |||||
| Bacteraemia or arthritis in children | <45kg | >45kg | ||||
| IM or IV ceftriaxone 50mg/kg (max. 1g) every 24h 7 days | IM or IV ceftriaxone 1g every 24h 7 days | |||||
AEDV: Academia Española de Dermatología y Venereología [Spanish Academy of Dermatology and Venereology]; BASSH: British Association for Sexual Health and HIV; CDC: Centers for Disease Control and Prevention; ECDC: European Centre for Disease Prevention and Control; EDF: European Dermatology Forum; GeITS: Grupo de Estudio de ITS de la SEIMC (Sociedad Española de Enfermedades Infecciosas y Microbiología Clínica) [Spanish Society of Infectious Diseases and Clinical Microbiology STI Study Group]; GESIDA: Grupo de Estudio del SIDA de la SEIMC [SEIMC AIDS Study Group]; IM: intramuscular; IUSTI: International Union against Sexually Transmitted Infection; IV: intravenous; SD: single dose; SEIP: Sociedad Española de Infectiología Pediátrica [Spanish Society of Paediatric Infectious Diseases]; SPNS: Secretaría del Plan Nacional sobre el SIDA [Secretariat for the National Plan on AIDS]; UEMS: European Union of Medical Specialists; WHO: World Health Organisation.
The BASHH recommends cefotaxime as an alternative to ceftriaxone for neonates born before week 41 with uncomplicated gonorrhoea. Spectinomycin does not guarantee cure in pharyngeal infection.
Given the complexity of the treatment of gonorrhoea, if treatment failure is detected and once reinfection is ruled out, an STI expert should be consulted and the case should be reported to local public health officials as a matter of urgency. The alternative treatment options are shown in Tables 1–6.17,29–33 None of the second-line drugs has proved to be superior to ceftriaxone. In monotherapy, drugs such as spectinomycin, cefixime or gentamicin may contribute to the emergence of resistance without guaranteeing the eradication of pharyngeal gonorrhoea.17,30,31 The loss of sensitivity to azithromycin detected more recently25,27 makes it recommendable to restrict its use in monotherapy to avoid selection or acquisition of resistance. Several studies have shown the efficacy of gentamicin and gemifloxacin in combination with azithromycin in genital infection.36,37
For the treatment of gonorrhoea in people coinfected with HIV, no different regimen is required.17,29–31
Follow-up17,29–31Repeat microbiological testing is recommended to check for cure three weeks after finishing the treatment if using PCR or after at least three days if cultures are taken. Test of cure is recommended for infection in any location, but is imperative for pharyngeal infection, persistence of symptoms, second-line treatment, suspected noncompliance or reinfection, and in cases with probable acquisition in areas of high resistance such as Southeast Asia. In cases of treatment failure, culture and antibiogram should always be performed.
Given the high rates of reinfection in these patients, it is advisable to repeat testing at three months.
Prevention and control5,17,29–31In order to interrupt the chain of transmission, contact investigations are recommended (see below), in addition to the case and their partners abstaining from sexual relations for seven days after treatment (provided the symptoms have disappeared).
Screening for other STIs, especially chlamydia, syphilis and HIV, should always be performed. The possibility of coinfection with HIV varies according to risk practices, reaching 39% in men who have sex with men.38
It should be noted that in the case of gonorrhoea, as with any other STI, other preventive and sexual health promotion measures have to be implemented, such as: reviewing viral hepatitis serological status and updating hepatitis A, hepatitis B and HPV vaccines in the at-risk population; carrying out health education; and promoting the use of barrier prevention methods, such as condoms.
It is also important to remember that gonorrhoea is notifiable in Spain, so it has to be reported according to the circuit established in each region.
Treatment of sexual partners5,17,29–31The sexual partners of the patient in the 2–3 months prior to the onset of symptoms or at the date of sample collection in asymptomatic patients should be assessed. If the last partner was more than three months previously, they should be studied. In symptomatic gonococcal urethritis in men, the study and treatment of sexual partners can be limited to those of the two weeks prior to the onset of symptoms. However, if the last sexual partner was prior to this period, they should always be studied.
The treatment regimen of the contact should be the same as that of the patient and it is advisable that they also maintain sexual abstinence for seven days post-treatment. With the current diagnostic resources, based on PCR techniques, epidemiological treatment of asymptomatic sexual contacts could be given only to those who are assessed within the first two weeks after sexual exposure to the index case, because in that period a negative PCR may not rule out infection.33,35 If such cases assessed within the first two weeks after exposure did not receive epidemiological treatment, they would then need to be re-examined after that window period.35,39
The contacts assessed beyond two weeks after the last contact can be treated only based on the results of the tests, abstaining from having sexual relations until confirming the absence of infection or more than seven days after treatment.35,39
Regarding the option of providing epidemiological treatment to contacts through the index case, it has only been recommended in heterosexual couples in whom there is no other chance of access to the healthcare system. In Spain, there is no specific legislation in this regard and this strategy involves risks such as the possibility that the contact has allergies or contraindications to drugs that are unknown to the prescribing doctor or to the index case. This strategy is particularly discouraged in the case of men who have sex with men due to a higher likelihood of co-infections, especially HIV, which would not be diagnosed.
Conflicts of interestThe authors declare that they have no conflicts of interest.
Please cite this article as: Barberá MJ, Serra-Pladevall J. Infección gonocócica: un problema aún sin resolver. Enferm Infecc Microbiol Clin. 2019;37:458–466.