This study aims to compare the cost-effectiveness of currently approved daily PrEP in Spain, with tenofovir disoproxil fumarate plus emtricitabine (TDF/FTC), in gay, bisexual and other men who have sex with men (GBMSM) versus newer alternatives like daily tenofovir alafenamide plus emtricitabine (TAF/FTC) or injectable cabotegravir every 2 months (CAB).
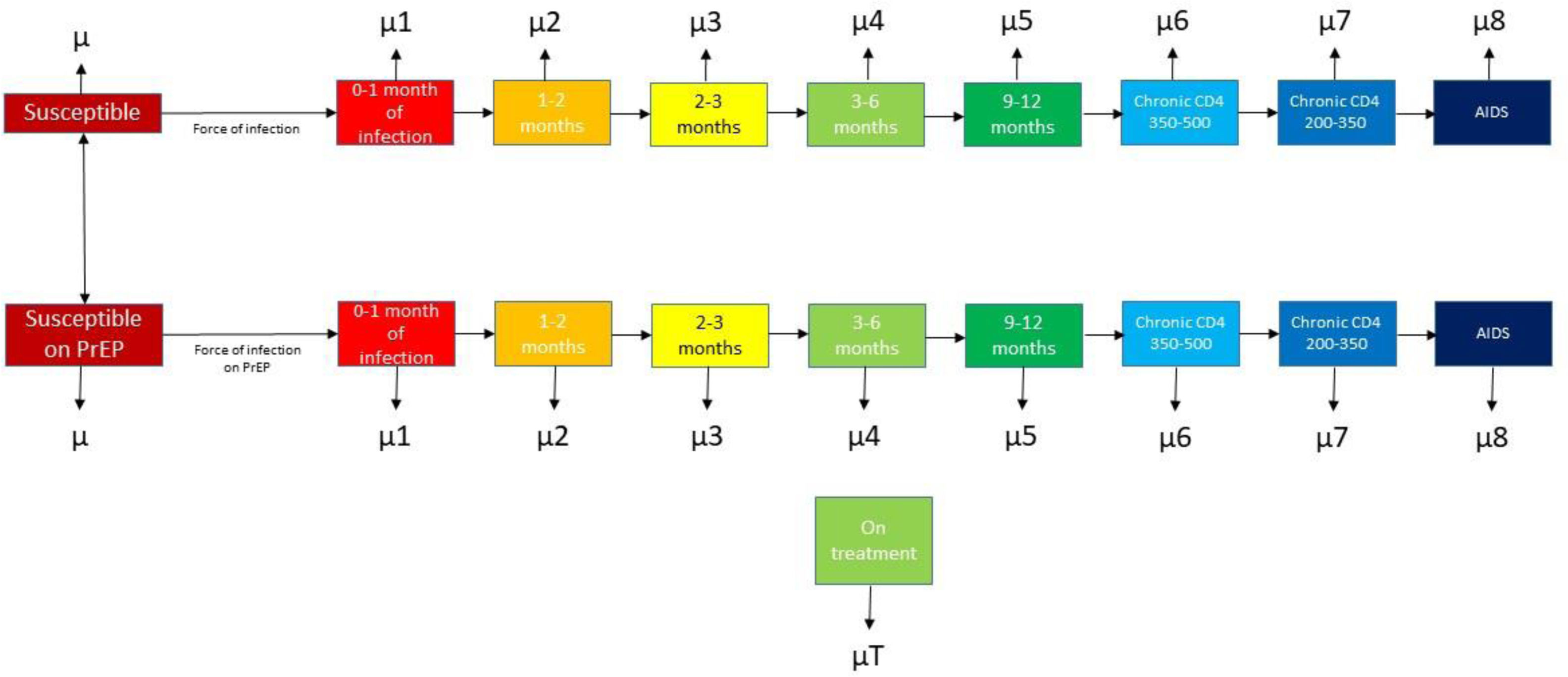
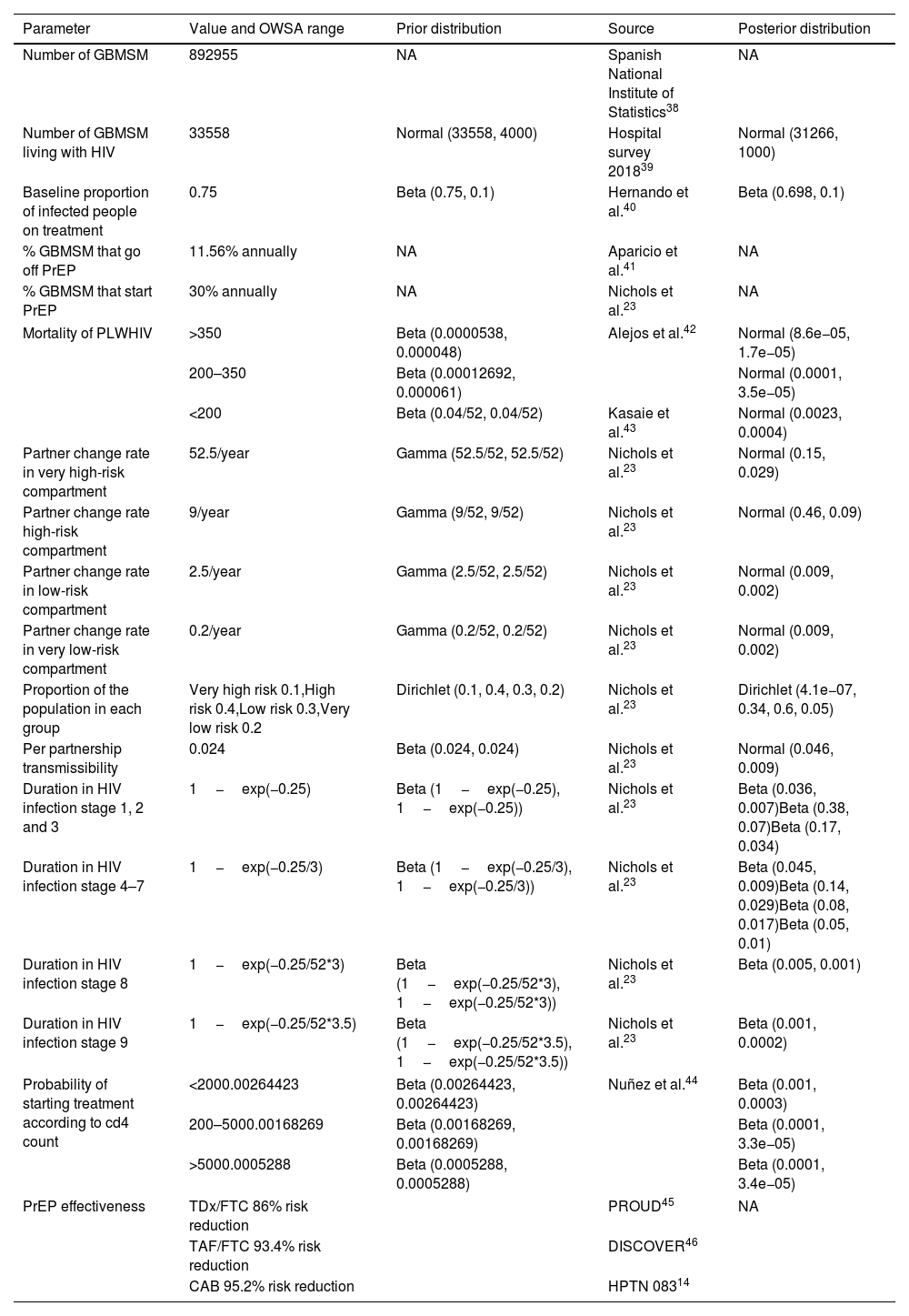
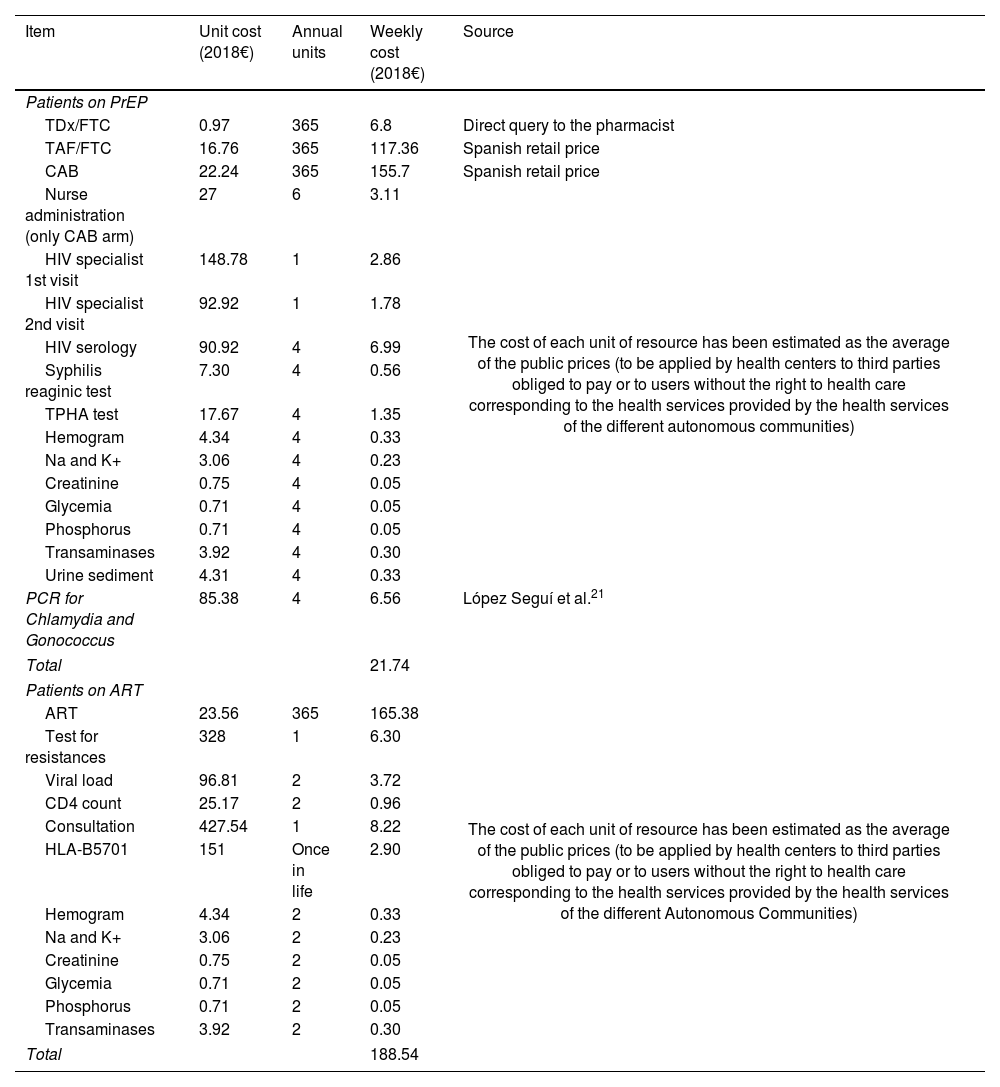
MethodsWe fitted a dynamic compartmentalized Markov model that represents the dynamics of HIV-1 transmission in GBMSM in Spain. The model was calibrated to replicate the epidemiological data of the HIV epidemic in GBMSM in Spain from 2013 to 2018. We used the perspective of the national health system and applied a 40-year time horizon. Quality-adjusted life years (QALYs) were the health outcome variable, and the cost was accounted for in 2018 Euros (€). Our outcome variable was the incremental cost-effectiveness ratio (ICER) for PrEP. We used the 2018 gross domestic product per capita of Spain (€25,854) as the willingness-to-pay (WTP) threshold.
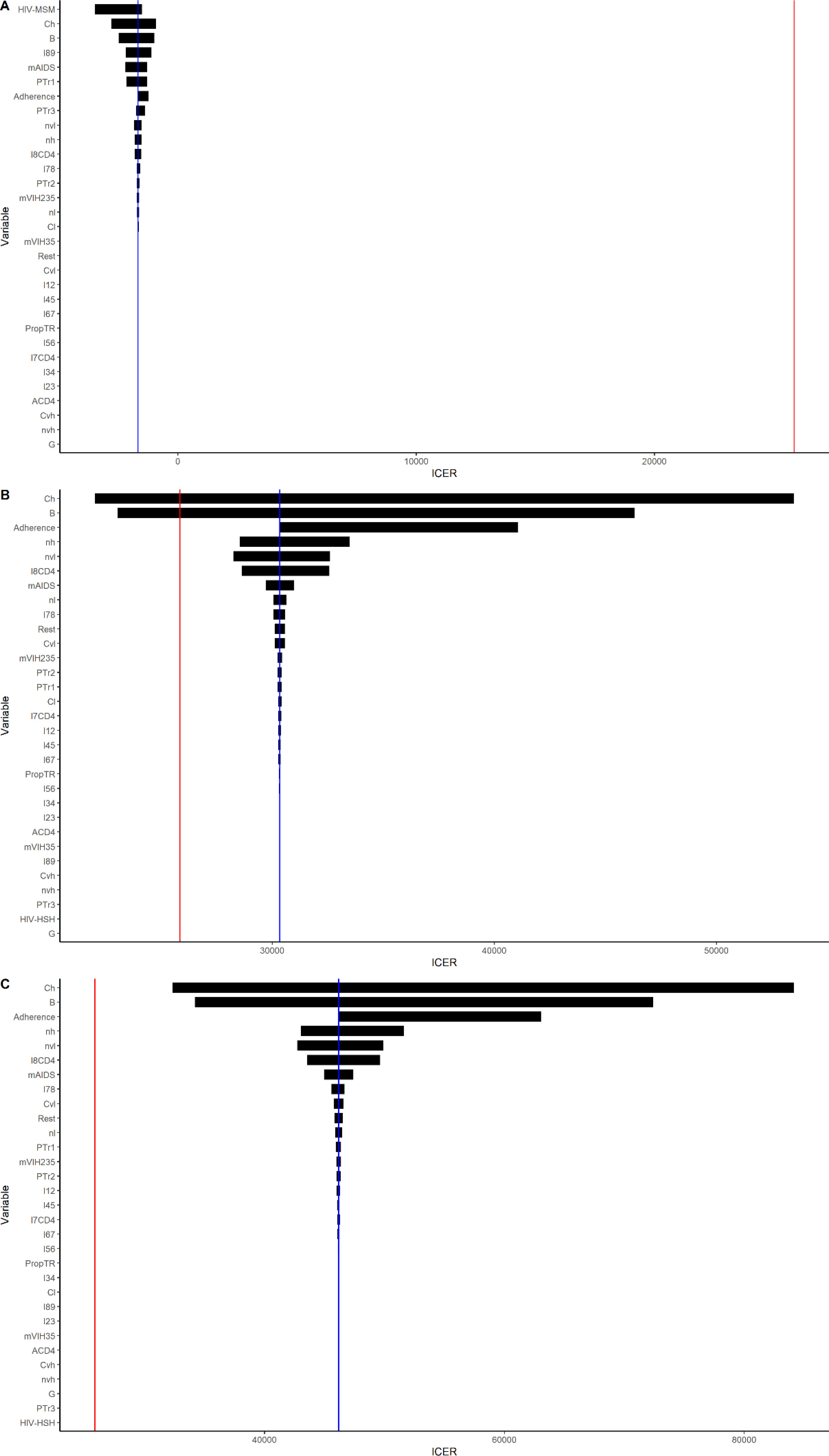
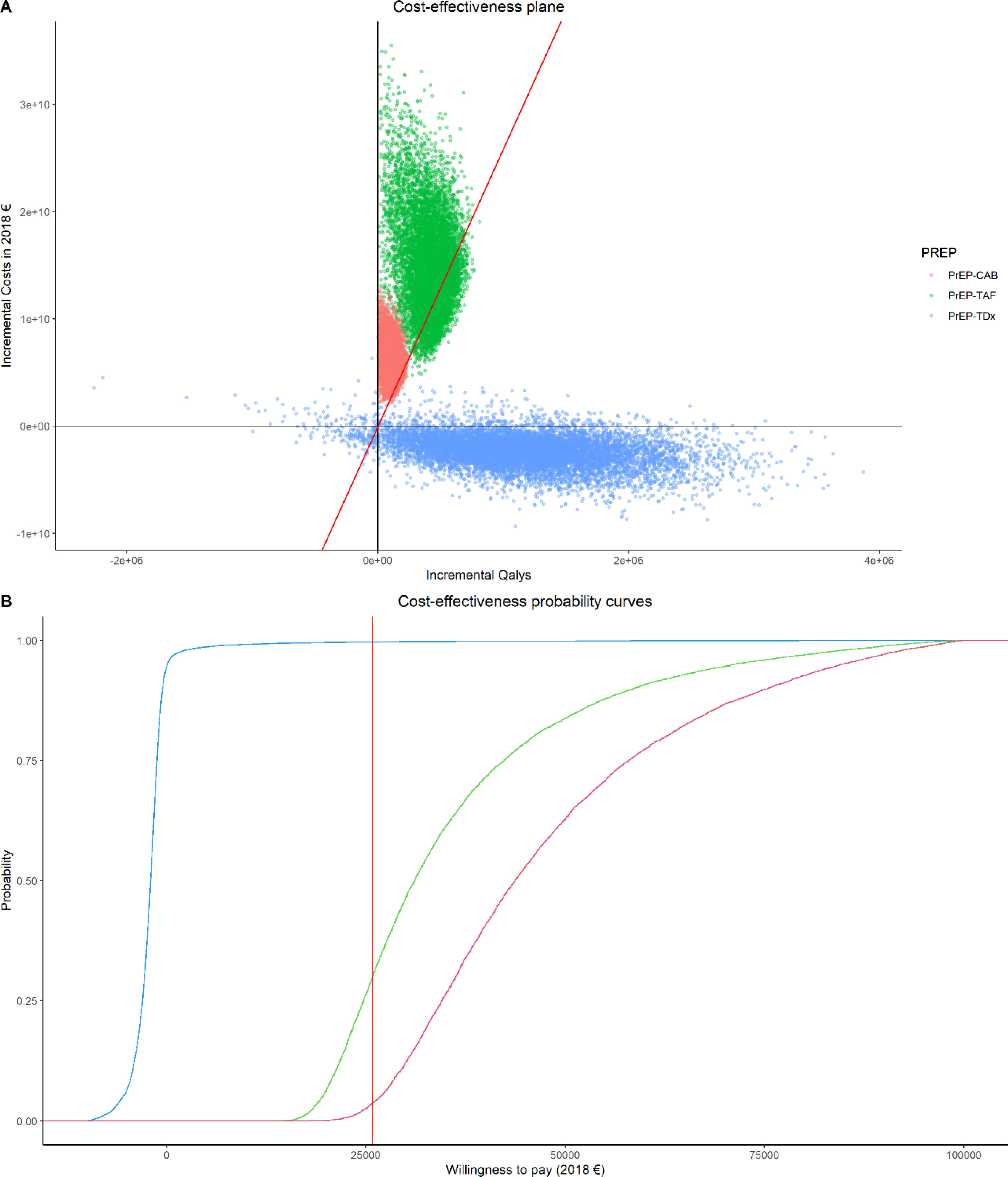
ResultsThe 2019 scenario with no PrEP in place would yield 17,424,891 QALYs and a cost of 2018 €17,345,310,254. The present scenario of daily TDF/FTC yields 18,615,325 QALYs, an increase of 1,190,434 QALYs at a cost of 2018 €15,354,878,534 (decrease of −2018 €1,990,431,719), generating an ICER of −2018 €1672 per QALY gained. The introduction of daily TAF/FTC in the present scenario would yield an increase in 449,392 QALYs at an additional cost of 2018 €13,634,260,217. The ICER would thus be 2018 €30,339 per QALY gained. Introducing CAB in the present scenario would yield an increase of 573,007 QALYs at an additional cost of 2018 €16,754,471,790 (average cost-effectiveness ratio=29,239). Compared to TAF, the increase in QALYs would be 123,614 at an additional cost of 2018 €5,707,367,590, yielding an ICER of 2018 €46,170/QALY gained. A one-way sensitivity analysis and a probabilistic sensitivity analysis was carried out.
ConclusionThe present Spanish policy of PrEP is a cost-saving strategy. TAF/FTC and CAB are not cost-effective at current market prices.
Este estudio tiene como objetivo comparar el coste-efectividad de la profilaxis preexposición (PrEP) diaria actualmente aprobada en España, con tenofovir disoproxil fumarato más emtricitabina (TDF/FTC), en hombres gais, bisexuales y otros hombres que tienen sexo con hombres (GBHSH) frente a alternativas más recientes, como tenofovir alafenamida más emtricitabina (TAF/FTC) de administración diaria o cabotegravir inyectable cada 2 meses (CAB).
MétodosSe ajustó un modelo de Markov dinámico compartimental que representa la dinámica de transmisión del VIH-1 en GBHSH en España. El modelo fue calibrado para replicar los datos epidemiológicos de la epidemia de VIH en GBHSH en España desde 2013 hasta 2018. Se utilizó la perspectiva del sistema nacional de salud y se aplicó un horizonte temporal de 40 años. Los años de vida ajustados por calidad (AVAC) fueron la variable de resultado de salud, y el coste se contabilizó en euros de 2018 (€). Nuestra variable de resultado fue la razón incremental de coste-efectividad (ICER) para la PrEP. Se empleó el producto interior bruto per cápita de España en 2018 (25.854€) como umbral de disposición a pagar (WTP).
ResultadosEl escenario de 2019 sin PrEP generaría 17.424.891 AVAC y un coste de 17.345.310.254€ (2018). El escenario actual de PrEP diaria con TDF/FTC genera 18.615.325 AVAC, un incremento de 1.190.434 AVAC a un coste de 15.354.878.534€ (2018), lo que supone una disminución de −1.990.431.719€ (2018), generando un ICER de −1.672€ (2018) por AVAC ganado. La introducción de TAF/FTC diaria en el escenario actual generaría un aumento de 449.392 AVAC a un coste adicional de 13.634.260.217€ (2018). El ICER sería de 30.339€ (2018) por AVAC ganado. La introducción de CAB en el escenario actual generaría un incremento de 573.007 AVAC a un coste adicional de 16.754.471.790€ (2018) (razón media de coste-efectividad=29.239). En comparación con TAF, el incremento en los AVAC sería de 123.614, con un coste adicional de 5.707.367.590€ (2018), lo que resulta en un ICER de 46.170€ (2018)/AVAC ganado. Se realizó un análisis de sensibilidad univariado y un análisis de sensibilidad probabilístico.
ConclusiónLa política actual de PrEP en España es una estrategia que ahorra costes. TAF/FTC y CAB no son rentables a los precios actuales de mercado.
Article
Socio de la Sociedad Española de Enfermedades Infecciosas y Microbiología Clínica

Para acceder a la revista
Es necesario que lo haga desde la zona privada de la web de la SEIMC, clique aquí
Para realizar los cursos formativos
La actividad estará abierta para socios de la SEIMC. IMPORTANTE, recuerde que requiere registro previo gratuito. Empezar aquí