A 41-year-old woman from Colombia was recently diagnosed with HIV-1 infection. A co-formulated antiretroviral treatment started when her CD4+ count was 13cells/mm3 (1.4%) and the HIV-1 viral load was 2,800,289copies/ml.
In March 2017, the patient was admitted to the Emergency Department (ED) because she had dyspnea, fatigue and fever (39°C). Thorax radiography revealed a multiple bilateral pulmonary interstitial and nodular pattern. A polymerase chain reaction (PCR) in plasma for cytomegalovirus (CMV) with 26,485copies/ml and culture for Mycobacterium avium (MAC) in bone marrow aspirate were found positive. Therapy with gancyclovir, ethambutol, rifabutin and clarithromycin was started showing good clinical response. Nevertheless, the patient developed a unilateral peripheral CMV retinitis, and intravitreal foscarnet was therefore associated. The patient has a favorable clinical outcome and she was discharged after 45 days.
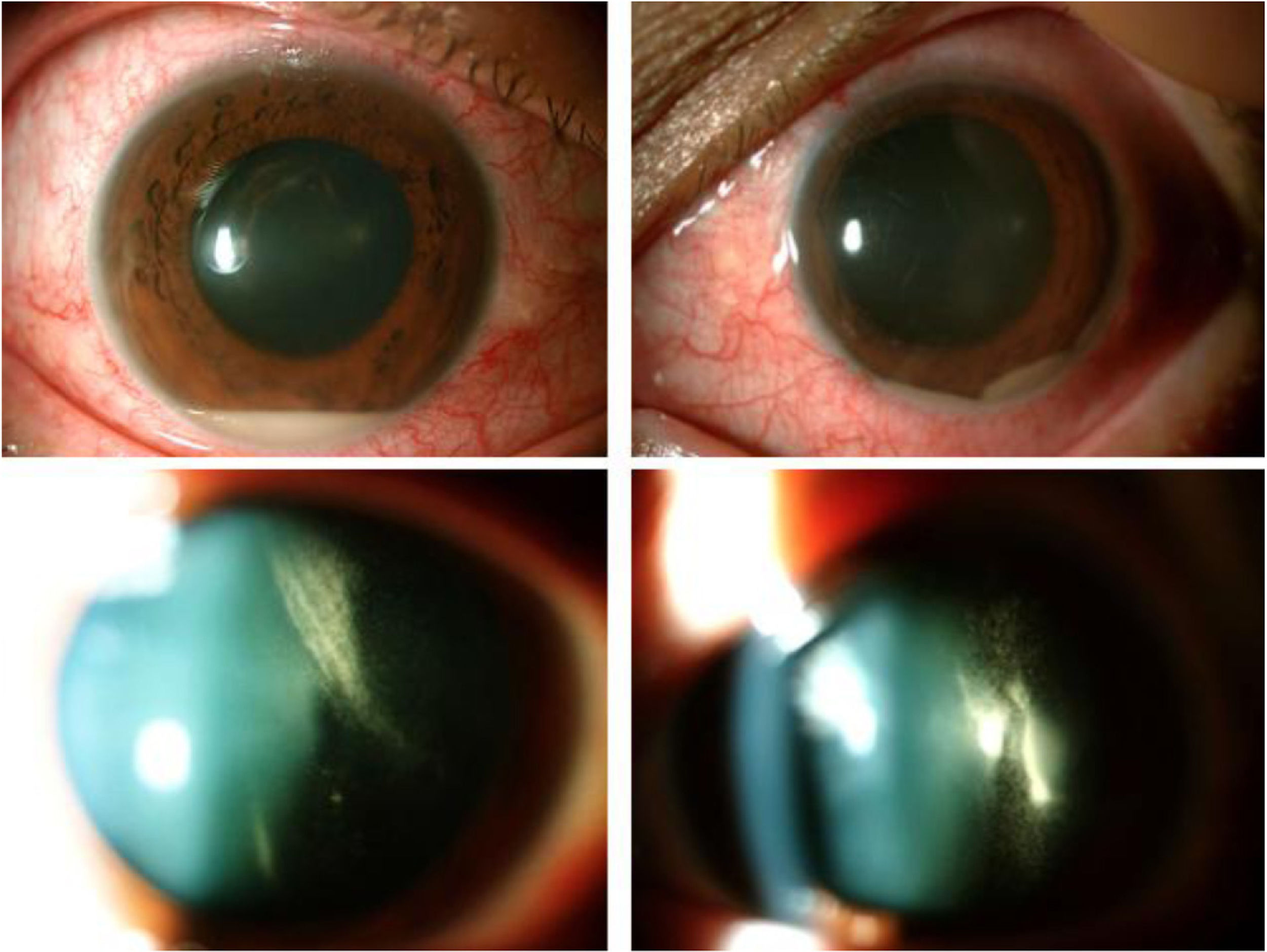
In October 2017, the patient was seen in the Ophthalmology ED for sudden bilateral blurring of vision, eye redness and ocular pain in both eyes. At this moment her CD4+ count was 165cells/μl and her plasma viral load was undetectable (<50copies/ml). The ophthalmologic exploration revealed a bilateral vitritis with no focus of chorioretinitis or vasculitis. The rifabutin based treatment for MAC was stopped and topical therapy with dexamethasone plus cycloplegic was started. After 24h, visual acuity decreased in patient's right eye and bilateral hypopyon was appeared with increased vitritis in both eyes (Fig. 1).
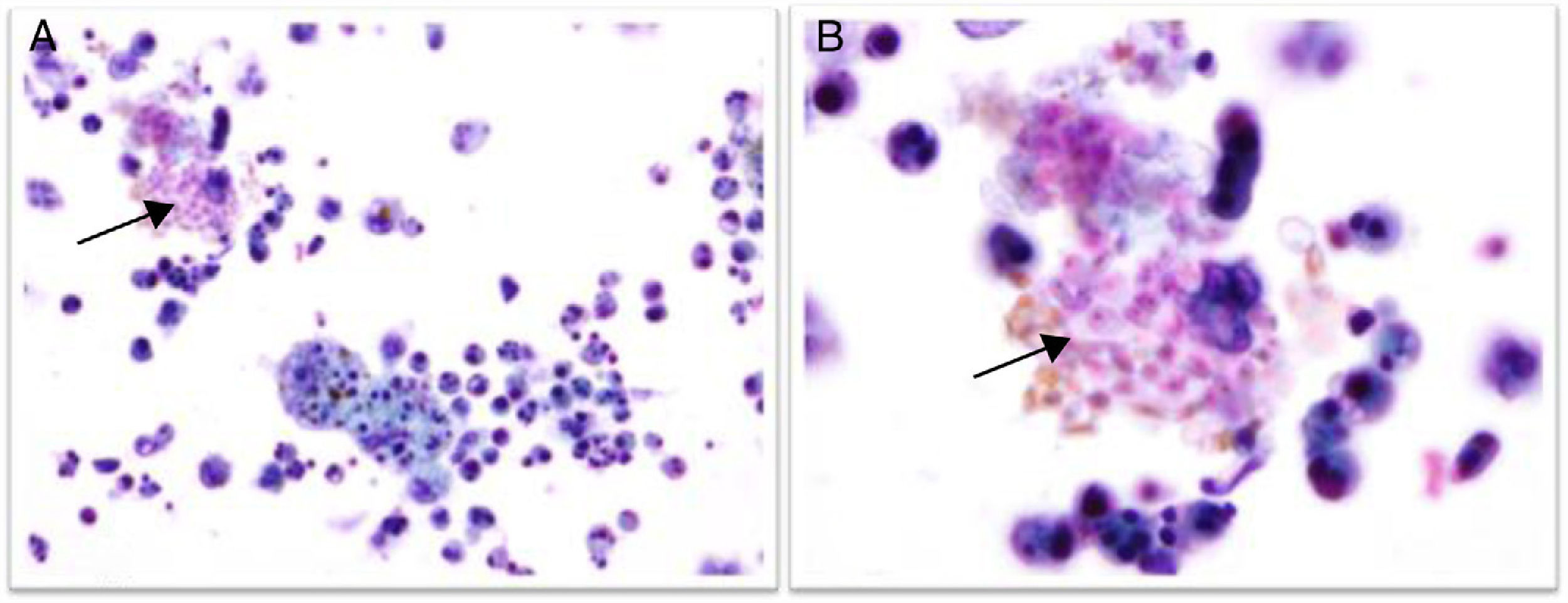
EvolutionAnterior chamber (AC) paracentesis was performed and samples were sent to the Pathology and Microbiology Department. Bacteriological and mycological cultures were negative after 48h and 30 days of incubation respectively. In the Pathology Department two cytospin preparations were stained with Papanicolau and Giemsa stain respectively. In both smears, numerous polymorphonuclear leukocytes and macrophages were observed, but only in the Papanicolau stain a significant amount of spores consistent with microsporidia were observed (Fig. 2). These findings were compatible with the acute infectious/inflammatory process.
A second AC paracentesis was performed for the detection of Toxoplasma gondii by PCR (RealCycler T. gondii, Valencia, Spain), which yielded negative result. A specific PCR for microsporidia (aqueous humor “in house” PCR) was positive, reinforcing the presence of microsporidia spores. Thereafter, albendazole 400mg/12h was administered for 7 days regressing both redness and ocular pain. However, visual acuity did not improve because of anterior segment inflammation and dense vitritis; therefore, oral and topic steroids were administered. After 6 weeks of albendazole plus dexamethasone and cycloplegic eye drops and 4 weeks of associated oral prednisone, the patient's visual function improved following her 5th month check up. Other opportunistic infections, inflammatory diseases related to HLA B27/B5 and pharmacology adverse reactions, were ruled out.
DiscussionIn HIV patients a wide spectrum of differential diagnosis ought to be considered when uveitis appers with hypopion. Pharmacologic causes must be considered, as rifabutin bilateral associated-uveitis1 is a well-known entity. In this case, symptoms did not resolve after suspension of rifabutin.
Differential diagnosis of posterior infectious uveitis should be ruled out in an immunocompromised patient: toxoplasmosis and CMV are the most frequent, although herpes zoster and herpes simplex virus play a role as well. Tuberculosis or serpiginous like chorioretinopathy and syphilis should equally be taken into account.2 Recognized forms of uveitis include intraocular inflammations with occur during or following some emerging infectious diseases: Chikungunya fever, dengue, Zika or Ebola virus disease.3
Microsporidiosis has been identified in immunocompromised and immunocompetent hosts.4 Different species of microsporidia were involved with a broad range of clinical presentations (diarrhea, keratoconjuctivitis, disseminated disease…). Microsporidia are intracellular spore forming ubiquitous funguses with a controversial taxonomy.
Ocular involvement with punctuate/stromal keratitis has been reported due to several risk factors.5 Save where uveal involvement has been described in animals,6 to the best of our knowledge, this is the first case in an HIV patient. Only one case has been reported in an immunocompetent patient concerning uveal involvement.7
During the diagnostic process of uveitis, the aqueous humor analysis is mandatory to diagnose of infectious uveitis. A pars plana vitrectomy (PPV) is mainly valuable for diagnosing lymphoma and non-infectious uveitis of unknown cause.8 Microsporidiosis detection is challenging and usually remains underdiagnosed. Laboratories should be alerted to the suspected diagnosis, and specific stains (periodic acid-Schiff, silver, Giemsa, acid-fast stains, modified Gram chromotrope and modified trichrome stains)9 should be performed to identify spore-forming shapes, since routine parasites or fungal examination does not detect microsporidia. Immunofluorescence assays or molecular methods are also useful but they are still not commercialized. PCR assays for detection and identification of microsporidia species have been developed,10 although they are only performed in specialized laboratories.9
Albendazole is effective against microsporidia. The treatment usually lasts for approximately four weeks. In case of confirmed ocular microsporidiosis, urine and respiratory samples should be examinated.6
FundingThis research did not receive any specific grant from fundingagencies in the public, commercial, or not-for-profit sectors.
Conflict of interestThe authors declare that there is no conflict of interest.
We gratefully acknowledge Boris Revollo from the Infectious Diseases Department of the Germans Trias i Pujol Hspital and Fernando Izquierdo and Ángela Magnet from the Parasitology Department of the San Pablo CEU University (Madrid) for the technical support and the performance of the molecular techniques.