Arterial and venous thromboses are very prevalent diseases and it has been discovered that they share some common pathways, but their presentation in young patients is rare and should raise concern for thrombophilia. Therefore, the case is presented of a 34 year-old female with no prior pertinent history and with no predisposing factors. She was diagnosed with a pulmonary embolism, with an incidental finding of a left ventricular thrombus. A coronary artery thrombosis was also found. The patient met the criteria for a thrombophilia work-up, but the authors would like to express their concern on whether these tests are truly necessary, as they would not have change the patient management.
La trombosis arterial y la trombosis venosa son enfermedades altamente prevalentes, y se ha visto que comparten algunas vías en su formación. Sin embargo, su presentación en pacientes jóvenes es rara y debe generar sospechas por trombofilias. Por esto queremos describir el caso de una mujer de 34años sin antecedentes clínicos pertinentes y sin factores predisponentes que fue diagnosticada de tromboembolismo pulmonar y con hallazgo incidental de un trombo en el ventrículo izquierdo y trombosis de la arteria coronaria. La paciente cumplía criterios para realizar estudios de trombofilia, pero abrimos la discusión de si estos son verdaderamente necesarios, ya que no modificarían el manejo de la paciente.
Thrombosis is the feared complication of Virchow's triad: hypercoagulability, endothelial injury and stasis. A thorough patient history and physical examination are important to help identify the systems and organs most likely affected.1 Timely recognition and intervention are critical for patients presenting with symptoms of thrombosis. Although work-up and management can slightly vary based on past medical history, predisposing factors, location, and clot burden, stabilizing the patient and initiating anticoagulation (unless contraindicated) are essential.2
Among many other factors, the location of the clot is of most importance to pinpoint the underlying mechanism. Certain factors predispose the patient to form clots in either arterial or venous circulation, with some shared factors like systemic inflammation or cardiac arrhythmias. Other predisposing factors include genetic mutations such as Factor V Leiden and prothrombin gene mutation, non-genetic intrinsic factors deficiencies such as Protein C and S deficiencies, and external factors such as oral contraceptive pills (OCP), tobacco use, recent surgery, prolonged immobility, and obesity.3
Currently in the US, venous and arterial thromboses carry incidence rates ranging from 1518 for ischemic heart disease cases to 200 for venous thromboembolism cases per 100,000 people. Globally in 2010 one in four deaths were attributed to an arterial or venous thrombosis, placing these disease among the top 10 contributors to morbidity and mortality.4 Secondary to the burden of these diseases, the recognition and awareness of them has gradually increased among health care providers and the general population, especially due to the disability and financial burden that they present in the health systems.3
Here we discuss the case of a young female patient in her early 30s who presented simultaneous thromboses of the coronary artery, pulmonary arteries and the left ventricular (LV) cavity. Patient verbal and written informed consent for publishing her case was obtained previous submission of the article
Case reportA 34-year-old female with a medical history of hypertension and anxiety, presented herself to the emergency department with shortness of breath and left sided chest pain associated cough accompanied by occasional streaks of blood. The patient also reported sudden onset of paroxysmal nocturnal dyspnea (PND) and orthopnea, which started the day prior to the ED visit. She denied oral contraceptive pill (OCP) use. Patient had a BMI of 47.5, was tachypneic, and had low oxygen saturation (80–84%) requiring supplemental oxygen with high flow nasal cannula (HFNC). There were no other abnormalities of her vital signs and no pertinent findings throughout the rest of physical exam. Initial laboratory studies revealed a troponin I of 0.35 and 0.45 (ng/ml), a BNP of 1460 (pg/ml), and no other abnormalities. A 12 lead EKG showed sinus rhythm with Q waves in leads V1 to V4.
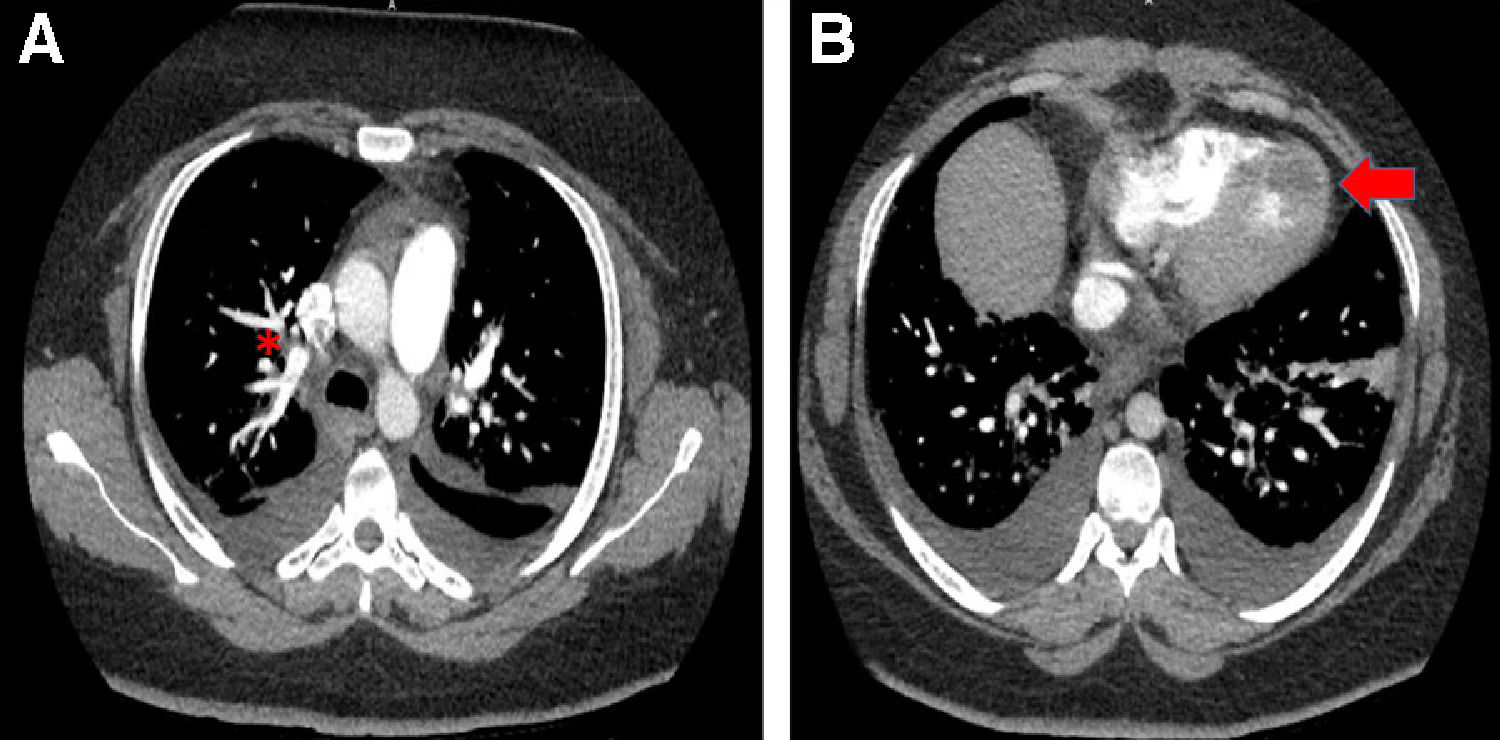
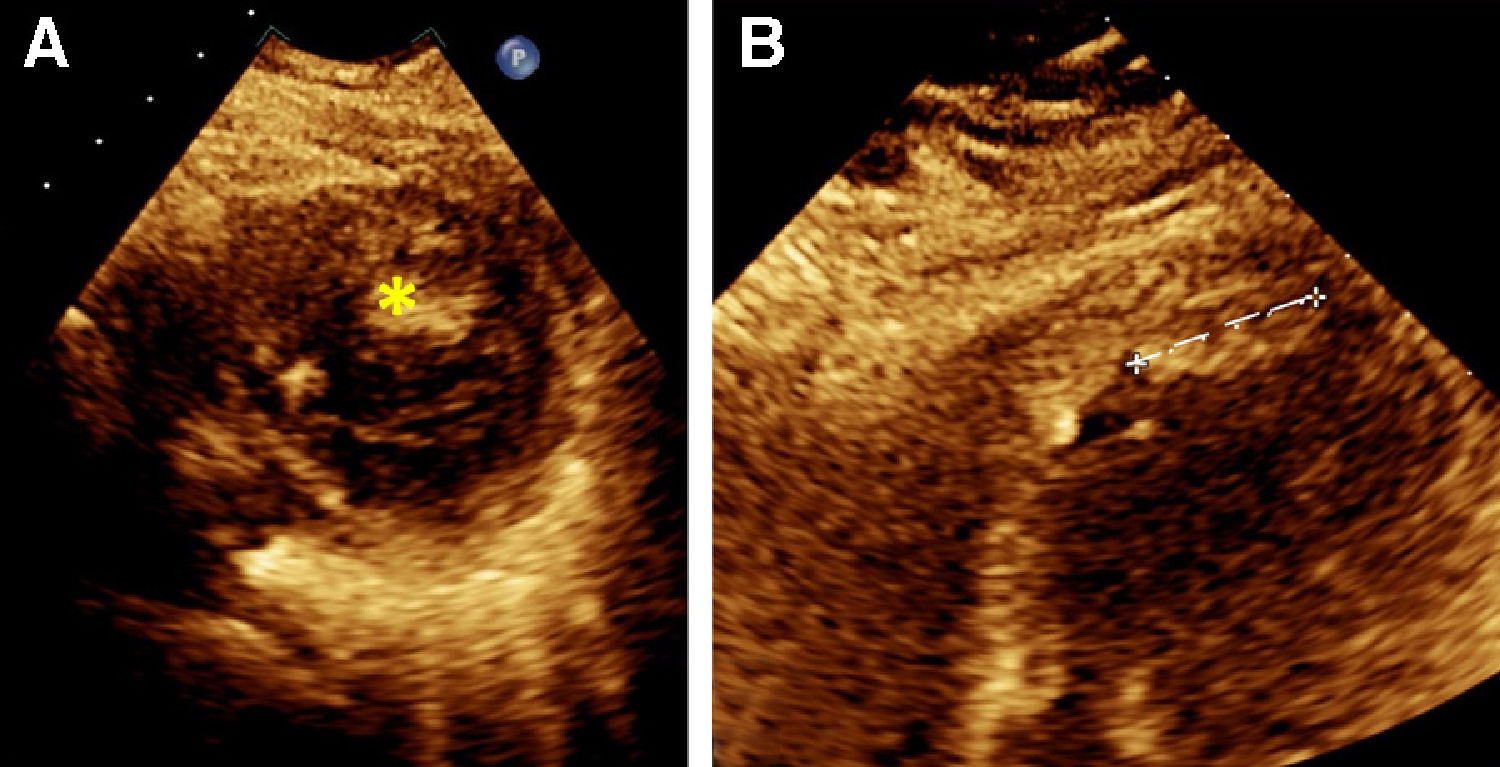
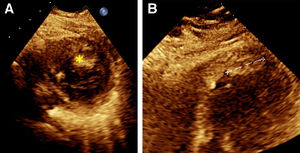
A chest CT with IV contrast identified a single pulmonary embolus in the right upper lobe, a 1.8×2cm hypoechoic finding in the inferior left ventricular apex concerning for a thrombus, and mild bilateral pulmonary edema (Fig. 1). The patient was started on a heparin drip and cardiology was consulted. A transthoracic echocardiogram confirmed a 2.25cm layered thrombus at the left ventricular apex, as well as the identification of global hypokinesia, mostly in the mid and distal segments suggestive of stress-induced cardiomyopathy. The transthoracic echocardiogram also revealed a left ventricular ejection fraction of 30% (Fig. 2). The patient was started on furosemide due to new onset heart failure with reduced ejection fraction and raised concern for pulmonary edema.
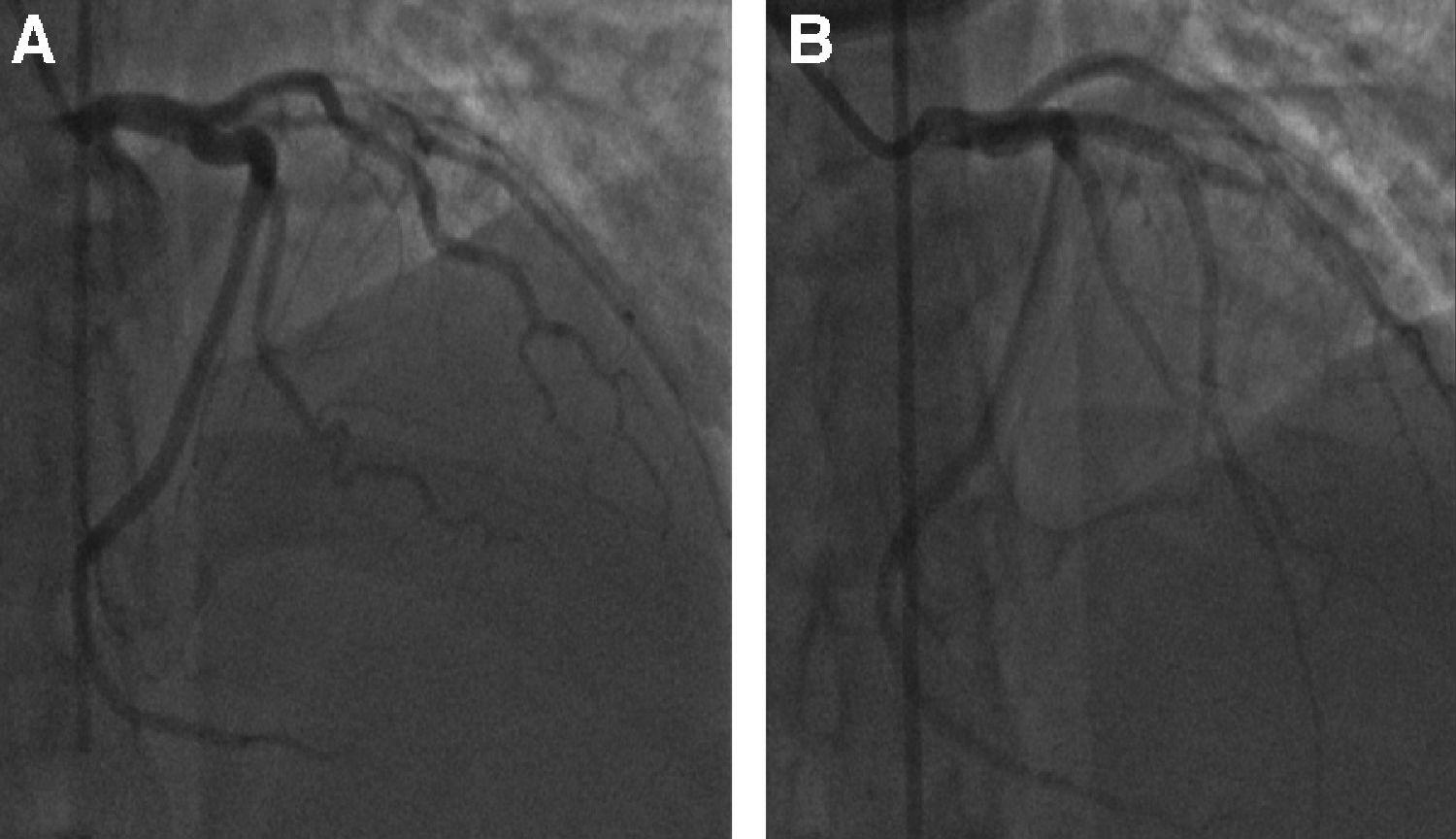
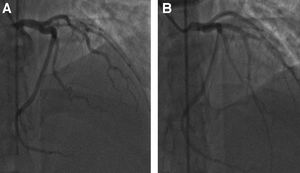
Bilateral lower extremity Doppler imaging ruled out deep venous thrombosis (DVT). Due to the underlying cardiomyopathy and positive troponins, patient underwent left heart catheterization. The study identified total occlusion of left anterior descending artery (LAD) (Fig. 3), which was treated with mechanical thrombectomy followed by the insertion of five drug eluting stents (Fig. 3). The rest of the coronary circulation did not show any abnormalities. After the procedure, the patient was placed on medical management for ischemic cardiomyopathy with metoprolol, lisinopril, atorvastatin, and dual antiplatelet with aspirin and clopidogrel. A hematology consult was requested, and the recommendation was to transition the patient to warfarin with a goal INR of 2–3. Hypercoagulable work-up was deferred as the patient had an active thrombosis and was on anticoagulation drugs, which would interfere with the non-genetic hypercoagulable tests. The consultant recommended indefinite-duration anticoagulation due to the fact that the patient appeared to have had unprovoked thrombi, and also due to her ongoing risk factor of morbid obesity. Warfarin was chosen as the optimal anticoagulant because of the patient's intracardiac thrombus and data that indicated that warfarin was the preferred agent.5
DiscussionUnprovoked arterial and venous thrombosis are often due to underlying hypercoagulable states or disease6 diagnosed in people younger than 40 years of age such as in this patient. Left ventricular (LV) thrombus is one of the common consequences of ischemic heart disease and is highly associated with arterial thrombosis. It has been associated with secondary arterial thrombosis like ischemic strokes and renal ischemia.7,8 Despite these associations, it is very difficult to establish the chronology of the events – whether the ischemic heart disease triggered the appearance of the left ventricular thrombus or vice versa.
The presence of concomitant pulmonary artery thrombosis as well as arterial thrombosis in a young individual increases the suspicion of underlying hypercoagulable conditions like antiphospholipid syndrome (APS), protein C and S deficiency and other hypercoagulable conditions that have been described in the literature.7,9 Patient did not have any personal risk factors that could explain the hypercoagulable state nonetheless, patient had strong family history for hypercoaguable disorders as her father had an episode of DVT when he was young (unclear age) and her father's brother also had multiple episodes of thrombosis (unclear locations and age). Even with this strong association, it has been shown that the prevalence of these conditions in the general population is less than 5%.10
Regarding the hypercoagulable state studies, it is essential to identify who will benefit from such costly work up. There has been a discussion about appropriate timeframe for the work up and associated benefits and drawbacks.
There are clear recommendations about which patients should undergo hypercoagulable studies; but before performing any studies, it is advised to avoid performing the hypercoagulable work up in the acute setting of the thrombus or in the presence of anticoagulants as the results would be altered and the results could be misleading and misinterpreted.6 Criteria for testing for thrombophilia has been established and it is recommended in cases with strong family history of venous thrombosis, thrombosis with none or very weak provoking factors at ages younger than 40–50, recurrent episodes of venous thrombosis and thrombosis in unusual sites as cerebral venous sinuses or intra abdominal veins. The patient described above meets most of the recommended criteria such as a young age, no precipitating factors, and with strong family history thrombotic conditions.2,6 Despite meeting these criteria, the prevalence of hypercoagulable syndromes is not high and. the most important question is whether the result will change the treatment plan or impact the prognosis.7 In the case of this patient, she meets criteria for lifelong anticoagulation and the results of the hypercoagulable work up will not change the treatment plan.
Statement of ethicsThe patient gave verbally and written consent for the use and publication of her case. A copy of the consent would be sent upon request
Author contributionsRGR and TV obtained all the clinical information and made the first draft of the manuscript, as well as further versions.
KA was the expert in charge of guiding the approach of the manuscript, gave feedbacks and actively participated in obtaining more detailed family history from the information.
FundingNo funding sources to declare.
Conflict of interestsThe authors declared no conflict of interests.
We would like to acknowledge the initial approach of writing this paper to Dr. Maria del Pilar Pardi, faculty from Saint Agnes Hospital.