Improper use of antibiotics increases antimicrobial resistance.
ObjectiveEvaluate the use of antibiotics and the impact of an intervention designed to improve antibiotic prescription for surgical prophylaxis in 6 hospitals of Monterrey, Mexico.
Material and methodsDesign: A prospective multicenter survey and a pretest–postest experimental study. Phase 1: Survey to evaluate the use of antibiotics through an especially designed guide. Phase 2: Intervention designed to improve antibiotic prescription for surgical prophylaxis by the medical staff by using printed, audiovisual and electronic messages. Phase 3: Survey to evaluate the impact of the intervention. Analysis: Frequencies, percentages, medians, ranges and χ2 test.
ResultsPhase 1: We evaluated 358 surgical patients, 274 prophylactic antibiotic regimens. A total of 96% of antibiotics regimens began with inappropriate timing (290/302), 82.8% were inappropriate regimens (274/331), 77.7% were in inappropriate dosage (230/296), 86% of inadequate length (241/280), and in 17.4% restricted antibiotics were used (52/299). Phase 2: 9 sessions including 189 physicians (14 department chairs, 58 general practitioners and 117 residents). Phase 3: We evaluated 303 surgical patients, 218 prophylactic antibiotics regimens. Inappropriate treatment commencement was reduced to 84.1% (180/214) (p<0.001), inappropriate regimens to 75.3% (162/215) (p=0.03), inappropriate dosages to 51.2% (110/215) (p<0.001), and use of restricted antibiotics to 8.3% (18/215) (p=0.003).
ConclusionsInappropriate use of prophylactic antibiotics in surgery is a frequent problem in Monterrey. The intervention improved the antibiotic prescription for surgical prophylaxis by reducing inappropriate treatment commencement, regimens, dosages, and overuse of restricted antibiotics. It is necessary to strengthen strategies to improve the prescription of antibiotics in surgical prophylaxis.
El uso inadecuado de antibióticos incrementa la resistencia antimicrobiana.
Objetivos Evaluar el uso de antibióticos y el impacto de una intervención dirigida a modificar la conducta prescriptiva en profilaxis quirúrgica en 6 hospitales de Monterrey.
Material y métodosDiseño: encuesta multicéntrica prolectiva y estudio de intervención antes y después. Etapa 1: Evaluación del uso de antibióticos con base en una guía especialmente diseñada. Etapa 2: Intervención dirigida a modificar la conducta prescriptiva mediante entrega de material impreso, audiovisual y electrónico. Etapa 3: Evaluación del impacto de la intervención. Análisis: Frecuencias absolutas, porcentajes, medianas, rangos y prueba de χ2.
ResultadosEtapa 1: Se evaluó a 358 pacientes quirúrgicos, 274 esquemas de antibióticos profilácticos. El 96% iniciaron en momento inadecuado (290/302), el 82.8% fueron esquemas inadecuados (274/331), el 77.7% aplicaron dosis inadecuada (230/296), el 86% con duración inadecuada (241/280) y en el 17.4% se usaron antibióticos restringidos (52/299). Etapa 2: Hubo 9 sesiones que incluyeron 189 médicos (14 directivos, 58 médicos de base y 117 residentes). Etapa 3: Se evaluó a 303 pacientes quirúrgicos con 218 esquemas profilácticos. Los momentos de inicio inadecuados se redujeron a 84.1% (180/214) (p<0.001), los esquemas inadecuados a 75.3% (162/215) (p=0.03), las dosificaciones inadecuadas a 51.2% (110/215) (p<0.001) y los antibióticos restringidos a 8.3% (18/215) (p=0.003).
ConclusionesEl uso inadecuado de antibióticos en profilaxis quirúrgica es prevalente en Monterrey. La intervención mejoró la conducta prescriptiva al reducir momentos de inicio, esquemas y dosis inadecuados, así como el uso de antibióticos restringidos. Es necesario reforzar la aplicación de estrategias dirigidas a mejorar la conducta prescriptiva de antibióticos en profilaxis quirúrgica.
The indiscriminate use of antibiotics is a common problem worldwide. It has increased antimicrobial resistance, treatment costs and the need for new antibiotics that are often more costly and with higher toxicity.1,2 In a cross-sectional study performed over 3 two-year periods (1991–1992; 1994–1995; 1998–1999) an increased use of broad spectrum antibiotics was observed on comparing the first and the final period studied, from 24% to 48% in adults, and from 23% to 40% in children.3,4 Antimicrobial resistance is a serious global problem for the World Health Organisation. Its primary cause is the inappropriate use of antibiotics, which results in longer hospital stays leading to increased costs to health systems, families and society.5
Surgical site infections are the second cause of nosocomial infection in several series.6–9 It is calculated that between 2% and 5% of cases undergoing clean extra-abdominal surgery and 20% of abdominal surgery cases are complicated by this type of infection. Patients with surgical site infection also have a 60% greater risk of requiring admission to an intensive care unit, a 5 times’ greater risk of readmission to hospital, and are twice at risk of dying, compared to patients who do not suffer these types of infection-based complications. All of this culminates in substantially increased treatment costs.9–11
Because the incidence of surgical site infection is one of the most significant indicators of surgical quality of care and a factor with the greatest impact on its final outcome, the correct use of prophylactic antibiotics is a fundament element in surgery.7 The basic purpose of prophylaxis is to achieve and maintain appropriate levels of antibiotic in the tissues and plasma at the time of incision and during the entire surgical procedure.6,8,12,13 Administration of prophylactic antibiotics is based on the risk of wound infection, and therefore it is essential to be aware of the wound classification.14–16 In 2002 the U.S. Center for Medicare and Medicaid Services and Center for Disease Control and Prevention started the National Surgical Infection Prevention project. According to this project, prophylactic antibiotics should be given 60min before the surgical incision, duration of administration should not exceed 24h after the end of surgery and an additional dosage of the antibiotic should be given intraoperatively only if the duration of the operation exceeds 2 half-lives of the antibiotic.8,9,16,17 Furthermore, it has been demonstrated that the postoperative administration of antibiotics does not reduce the risk of infection, and that prolonged administration is associated with a risk of antibacterial resistance.16–18
The aim of this study was to assess the impact of an intervention designed to improve surgical prophylaxis prescribing behaviour in 6 hospitals of the metropolitan area of Monterrey, in the north east of Mexico.
Material and methodsDesignProlective multicentre survey, and before and after intervention study.
Drawing up of the prophylaxis guide and measuring instrumentBased on a thorough review of the international literature, 2 infectologists (Palacios-Saucedo and Briones-Lara) designed a “Surgical antibiotic prophylaxis guide”, which was submitted for assessment by 2 other infectologists (Solórzano-Santos and Díaz-Ramos, coordinated by Palacios-Saucedo) until they reached a consensus. The surgical antibiotic prophylaxis guide drawn up for this study is very similar to other international guidelines.6,13,15–17 The guide classifies surgical procedures according to wound type (clean, clean-contaminated, contaminated or dirty). For each of the different operations the first-choice antibiotic is shown with its dosage, the second-choice antibiotic with its dosage, time of administration, conditions when the dosage should be repeated, length of administration, and the recommended antibiotic in the event of allergy. Two of the participating infectologists (Palacios-Saucedo and Briones-Lara) designed the instrument for collecting data on antibiotic use, which contained 4 sections: 1) General patient data, including name, age, sex, weight and date of admission. 2) Information on the surgical procedure, which included the diagnosis leading to surgery, name of the operation, date, start time, duration and site of surgery, and wound type (clean, clean-contaminated, contaminated or dirty). Only surgical patients were included in the final analysis. Surgical patients were considered to be all those who had undergone any non-ambulatory surgery in the 6 previous days or who were to undergo such surgery the day following implementation of the survey. 3) Information on antibiotic use, which included whether or not they were taking antibiotics and whether they were prophylactic or therapeutic, number of antibiotics, prescribing department, and the start and end date of the antibiotic. 4) An assessment of the nature of the prescription of the antibiotic/s, which included information on the date and start time, whether or not the use of prophylaxis was appropriate, whether or not the regimen, dosage and duration were appropriate, and whether restricted antibiotics were being used. The instrument clearly indicated the specific pre-established sites of the clinical records from which the information would be collected, with a maximum of 3 sites as the origin of the information for each variable. These sites included: admission note and instructions, note of outcome or external consultation where surgery was indicated, preanaesthetic note, postanaesthetic note, postsurgical note and instructions, the final outcome note, and others.
When the regimen was prescribed to prevent surgical site infection according to the infection risk as per the wound classification, it was considered prophylactic. It was considered therapeutic when it was prescribed to treat existing disease. The regimen was considered appropriate when antibiotics were prescribed for prophylactic purposes according to the “surgical antibiotic prophylaxis guide”. Treatment commencement was considered appropriate when the first dosage of the antibiotic was given between 30 and 60min before the surgical incision in clean or clean-contaminated wounds, or if it was started in the time indicated in the “surgical antibiotic prophylaxis guide”. The regimen was considered appropriate when it corresponded with said guide. The dosage was considered appropriate if the amount administered in mg (or in mg/kg or mg/m2 of body surface for children) corresponded to the dosage indicated in the guide. The length of the regimen was considered to be from the time it commenced until it was discontinued, and was considered appropriate if it corresponded to the timing indicated in the guide, which in most of the clean and clean-contaminated operations was a single dosage. The following were considered restricted antibiotics: meropenem, imipenem-cilastatin, cefepime, vancomycin, linezolid, ciprofloxacin, gatifloxacin, teicoplanin, ticarcillin/clavulanic acid and piperacillin/tazobactam.
Study logisticsBefore the first survey was conducted, a pilot study was carried out in each participating hospital, which administered the questionnaire using 5–7 clinical records to make the necessary adjustments regarding the sites of origin of the information for each variable, and to add other types of surgery that had not been included in the guide. In addition, the time required for completing each questionnaire was assessed – 7–18min – in order to plan the time required for the survey to be conducted per hospital. The study was then administered in 3 stages. In the first stage, a baseline evaluation was made of the antibiotic used, the second stage implemented the intervention intended to improve surgical antibiotic prophylaxis prescribing behaviour, and in the third stage the survey was readministered to assess the impact of said intervention, which we describe below.
Stage 1 or preintervention stage (1st prolective multicentre survey)The survey was conducted over 2 or 3 consecutive days, using clinical records from all the patients hospitalised in the 6 largest hospitals of the metropolitan area of Monterrey. The hospitals were classified as follows: 2 public social security hospitals (HPSS), 2 private hospitals (HPRIV) and 2 public non-social security hospitals (HPNoSS). When the “surgical antibiotic prophylaxis guide” had been designed and the measuring instrument was ready, the survey was administered in 2 or 3 consecutive days for each hospital and then 2 of the infectologists (Palacios-Saucedo and Briones-Lara) independently evaluated the nature of antibiotic prescribing in each questionnaire. In the cases where there was no consensus, an agreement was reached to allocate the prescribing a score.
Stage 2 or intervention stageBased on the results from the first survey, an intervention intended to improve surgical antibiotic prophylaxis prescribing behaviour was implemented. It included the following strategies:
- 1)
Presentation of general results. The general results obtained from the 6 hospitals during the first survey were presented in one or 2 audiovisual presentations per hospital. General practitioners, department chairs and medical residents were included.
- 2)
Presentation of local results. The local results were presented in the same sessions for each corresponding hospital.
- 3)
Distribution of the guide for antibiotic use. Each participant was given a copy of the “surgical antibiotic prophylaxis guide”.
- 4)
Making copies of the guide available. Copies of the guide were left in areas of each hospital where prescribing took place, such as operating theatres, surgical and gynaecological departments etc.
- 5)
Distribution of a pocket mini-guide. A small, specially-designed, plastic-covered, pocket guide was distributed that contained the prophylaxis recommendations for the 7 most common operations in each hospital.
- 6)
Creation of a database with the participating doctors’ emails. At each session a list was passed around to collect the name, specialism, position and email of each participant.
- 7)
Delivery of emails concerning appropriate surgical prophylaxis. The above list was used to send all the participating doctors emails about surgical prophylaxis.
- 8)
Preparation of a presurgical prophylaxis format. The authorities of each participating hospital were recommended to prepare a “surgical antibiotic prophylaxis report sheet”, in a simple, rapid and easy-to-complete format, and it was suggested that this should be officially incorporated in the clinical records. This sheet should be filled in by every doctor when prescribing surgical prophylaxis.
- 9)
Creating and placing information posters in strategic areas. It was suggested to each participating hospital that information signs and posters on the most important aspects of the issue should be designed and placed in strategic areas, to remind the relevant personnel of its importance and of the guidelines for surgical prophylaxis.
In this stage, the impact of the intervention on surgical antibiotic prophylaxis prescribing behaviour was assessed, using the same instrument as in stage 1. The clinical records were reviewed again of all the patients hospitalised over 2 consecutive days in the 6 participating hospitals.
Statistical analysisAbsolute frequencies and percentages, means and standard deviations were used to analyse the results, and the χ2 test. SPSS version 15.0 was used for the analysis. A p value of <0.05 was considered significant.
Ethical aspectsThis study complies with the ethical principles of the Declaration of Helsinki and its later developments and the principles of good clinical practice. Similarly, it is in line with the standards set in the General Health Law and the institutional regulations on health research. The protocol was authorised by the Research Committee of the Hospital de Especialidades N.° 25. Informed consent was not required since only the patients’ clinical records were reviewed.
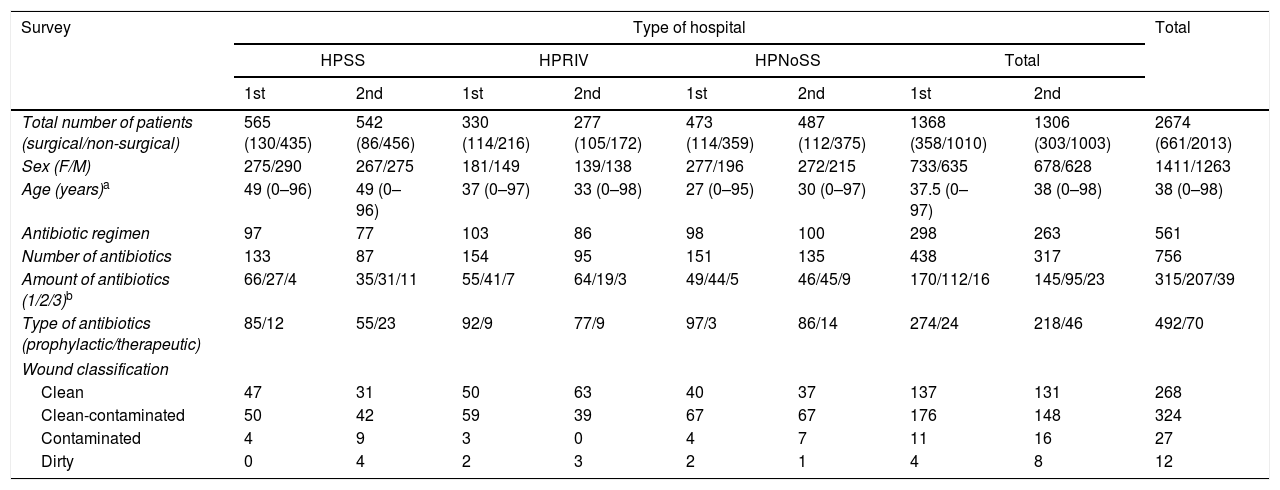
ResultsStage 1 or preintervention stageThe first survey was conducted to assess surgical antibiotic prophylaxis use in the 6 participating hospitals of the metropolitan area of Monterrey between 11 May and 7 July 2006. The survey was conducted over 2 or 3 consecutive days in each hospital. One thousand, three hundred and sixty-eight patients were assessed through their clinical records, of whom 358 were surgical patients. The latter were prescribed 298 antimicrobial regimens, of which 274 were for prophylaxis, with a total of 426 dosages of antibiotics. The median age of these patients was 37.5 years (from newborn to 97 years of age) and clean (137/328, 41.7%) and clean-contaminated (176/328, 53.6%) wounds predominated (Table 1). The most frequently used antibiotics were cefalotin in 113/298 (37.9%), cefotaxime in 68/298 (22.8%), ceftriaxone in 29/298 (9.7%), and ciprofloxacin in 28/298 (9.4%). The most frequently performed operations were caesarean section in 13.7% (49/357), vaginal delivery in 6.7% (24/357), cholecystectomy in 6.1% (22/357), and craniotomy in 5.6% (20/357).
General results per type of hospital before (1st survey) and after (2nd survey) an intervention intended to improve surgical antibiotic prophylaxis in 6 hospitals of the metropolitan area of Monterrey.
| Survey | Type of hospital | Total | |||||||
|---|---|---|---|---|---|---|---|---|---|
| HPSS | HPRIV | HPNoSS | Total | ||||||
| 1st | 2nd | 1st | 2nd | 1st | 2nd | 1st | 2nd | ||
| Total number of patients (surgical/non-surgical) | 565 (130/435) | 542 (86/456) | 330 (114/216) | 277 (105/172) | 473 (114/359) | 487 (112/375) | 1368 (358/1010) | 1306 (303/1003) | 2674 (661/2013) |
| Sex (F/M) | 275/290 | 267/275 | 181/149 | 139/138 | 277/196 | 272/215 | 733/635 | 678/628 | 1411/1263 |
| Age (years)a | 49 (0–96) | 49 (0–96) | 37 (0–97) | 33 (0–98) | 27 (0–95) | 30 (0–97) | 37.5 (0–97) | 38 (0–98) | 38 (0–98) |
| Antibiotic regimen | 97 | 77 | 103 | 86 | 98 | 100 | 298 | 263 | 561 |
| Number of antibiotics | 133 | 87 | 154 | 95 | 151 | 135 | 438 | 317 | 756 |
| Amount of antibiotics (1/2/3)b | 66/27/4 | 35/31/11 | 55/41/7 | 64/19/3 | 49/44/5 | 46/45/9 | 170/112/16 | 145/95/23 | 315/207/39 |
| Type of antibiotics (prophylactic/therapeutic) | 85/12 | 55/23 | 92/9 | 77/9 | 97/3 | 86/14 | 274/24 | 218/46 | 492/70 |
| Wound classification | |||||||||
| Clean | 47 | 31 | 50 | 63 | 40 | 37 | 137 | 131 | 268 |
| Clean-contaminated | 50 | 42 | 59 | 39 | 67 | 67 | 176 | 148 | 324 |
| Contaminated | 4 | 9 | 3 | 0 | 4 | 7 | 11 | 16 | 27 |
| Dirty | 0 | 4 | 2 | 3 | 2 | 1 | 4 | 8 | 12 |
F: female; HPNoSS: public non-social security hospitals; HPRIV: private hospitals; HPSS: public social security hospitals; M: male.
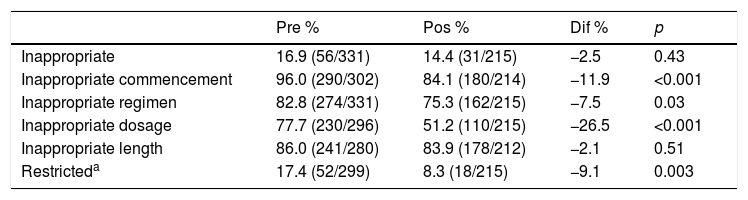
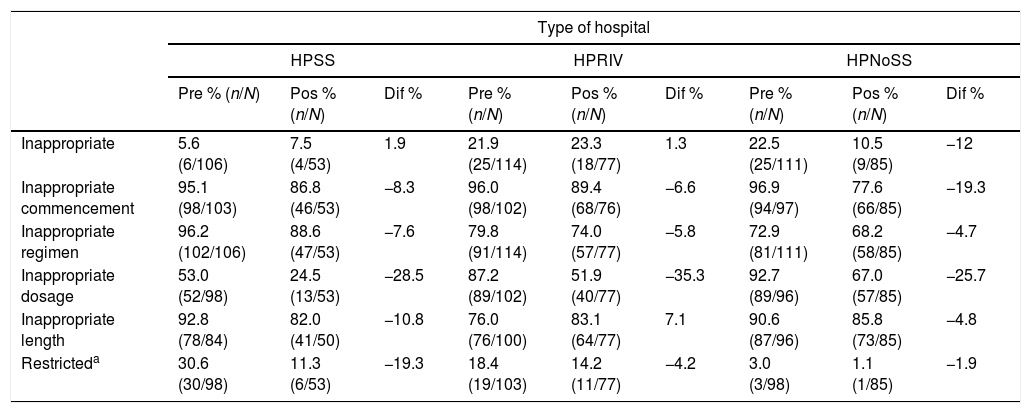
Assessment of the nature of antibiotic prescription, over the entire sample studied, showed that the regimen was in appropriate in 16.9% (56/331), began with inappropriate timing in 96% (290/302), inappropriate regimen in 82.8% (274/331), inappropriate dosage in 77.7% (230/296), of inappropriate length in 86% (241/280) and involved restricted antibiotics in 17.4% (52/299) (Table 2). In the analysis per type of hospital, the antibiotic prophylaxis was inappropriate in 5.6% (6/106) of the HPSS patients, in 21.9% (25/114) of the HPRIV patients and in 22.5% (25/111) of the HPNoSS patients (p=0.001). Prophylaxis began with inappropriate timing in 95.1% (98/103) of the HPSS patients, in 96% (98/102) of the HPRIV patients, and in 96.9% (94/97) of the HPNoSS patients (p=0.816). In most cases it was started after surgery in 66.3% (65/98) of the HPSS patients, in 74.4% (73/98) of the HPRIV patients, and in 69.1% (65/94) of the HPNoSS patients (p=0.449). Inappropriate prophylactic regimens were used in 96.2% (102/106) of the HPSS patients, in 79.8% (91/114) of the HPRIV patients, and in 72.9% (81/111) of the HPNoSS patients (p<0.001). Inappropriate dosages were prescribed in 53% (52/98) of the HPSS patients, in 87.2% (89/102) of the HPRIV patients and in 92.7% (89/96) of the HPNoSS patients (p<0.001). The length of prophylaxis was inappropriate in 92.8% (78/84) of the HPSS patients, in 76% (76/100) of the HPRIV patients and in 90.6% (87/96) of the HPNoSS patients (p=0.001). Restricted antibiotics were used in 30.6% (30/98) of the HPSS patients, in 18.4% (19/103) of the HPRIV patients, and in 3% (3/98) of the HPNoSS patients (p<0.001) (Table 3).
Impact of an intervention intended to improve surgical antibiotic prophylaxis in 6 hospitals of the metropolitan area of Monterrey.
| Pre % | Pos % | Dif % | p | |
|---|---|---|---|---|
| Inappropriate | 16.9 (56/331) | 14.4 (31/215) | −2.5 | 0.43 |
| Inappropriate commencement | 96.0 (290/302) | 84.1 (180/214) | −11.9 | <0.001 |
| Inappropriate regimen | 82.8 (274/331) | 75.3 (162/215) | −7.5 | 0.03 |
| Inappropriate dosage | 77.7 (230/296) | 51.2 (110/215) | −26.5 | <0.001 |
| Inappropriate length | 86.0 (241/280) | 83.9 (178/212) | −2.1 | 0.51 |
| Restricteda | 17.4 (52/299) | 8.3 (18/215) | −9.1 | 0.003 |
“Pre”, “Pos” and “Dif” refer to the results of each study before the intervention, after the intervention and the difference between both, respectively.
Assessment of antibiotic use per type of hospital before (Pre) and after (Pos) an intervention intended to improve surgical antibiotic prophylaxis in 6 hospitals of the metropolitan area of Monterrey.
| Type of hospital | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| HPSS | HPRIV | HPNoSS | |||||||
| Pre % (n/N) | Pos % (n/N) | Dif % | Pre % (n/N) | Pos % (n/N) | Dif % | Pre % (n/N) | Pos % (n/N) | Dif % | |
| Inappropriate | 5.6 (6/106) | 7.5 (4/53) | 1.9 | 21.9 (25/114) | 23.3 (18/77) | 1.3 | 22.5 (25/111) | 10.5 (9/85) | −12 |
| Inappropriate commencement | 95.1 (98/103) | 86.8 (46/53) | −8.3 | 96.0 (98/102) | 89.4 (68/76) | −6.6 | 96.9 (94/97) | 77.6 (66/85) | −19.3 |
| Inappropriate regimen | 96.2 (102/106) | 88.6 (47/53) | −7.6 | 79.8 (91/114) | 74.0 (57/77) | −5.8 | 72.9 (81/111) | 68.2 (58/85) | −4.7 |
| Inappropriate dosage | 53.0 (52/98) | 24.5 (13/53) | −28.5 | 87.2 (89/102) | 51.9 (40/77) | −35.3 | 92.7 (89/96) | 67.0 (57/85) | −25.7 |
| Inappropriate length | 92.8 (78/84) | 82.0 (41/50) | −10.8 | 76.0 (76/100) | 83.1 (64/77) | 7.1 | 90.6 (87/96) | 85.8 (73/85) | −4.8 |
| Restricteda | 30.6 (30/98) | 11.3 (6/53) | −19.3 | 18.4 (19/103) | 14.2 (11/77) | −4.2 | 3.0 (3/98) | 1.1 (1/85) | −1.9 |
HPNoSS: public non-social security hospitals; HPRIV: private hospitals; HPSS: public social security hospitals.
“Pre”, “Pos” and “Dif” refer to the results of each study before the intervention, after the intervention and the difference between both, respectively.
The intervention intended to improve surgical prophylaxis prescribing behaviour was implemented between 16 and 27 October 2006. Nine sessions were held in the 6 participating hospitals with a total of 189 doctors. The intervention included the audiovisual presentation of the total and local results for each hospital: 4 sessions in the HPSS, 2 sessions in the HPRIV and 3 in the HPNoSS. Of the 189 doctors that participated, 14 were department chairs, 58 were basic general practitioners (surgeons, interns, anaesthetists, gynaecologists, otolaryngologists, ophthalmologists, gastroenterologists, orthopaedic surgeons, urologists, dermatologists, etc.) and 117 were residents in various specialities. Most of the participants were from the HPSS, with 72 physicians (6 department chairs, 39 general and 27 residents). Sixty three doctors from the HPRIV participated (4 department chairs, 16 general and 43 resident physicians) and 54 doctors from the HPNoSS participated (4 department chairs, 3 general and 47 resident physicians). All the doctors who participated in the sessions were given the “surgical antibiotic prophylaxis guide” and the pocket mini-guide with the prophylaxis recommendations for the most common operations of each hospital. Copies of the guide were placed in strategic areas in all the hospitals. A “surgical antibiotic prophylaxis report sheet” was included in the clinical records of 2 of the participating hospitals, and information signs or posters were designed and distributed in 4 hospitals. The participating doctors’ database was used to send them reminder emails on the most important aspects of appropriate surgical prophylaxis.
Stage 3 or postintervention stageIn order to assess the impact of the intervention on surgical prophylaxis prescribing behaviour, the survey was repeated between 14 and 22 November 2006. During this stage, 1306 patients were evaluated through their clinical records, 303 of whom were surgical patients. Two hundred and sixty-three antibiotic regimens were used in these patients, of which 218 were prophylactic, with a total 404 dosages of antibiotics (Table 1). The median age of the patients in this stage was 38 years (from newborn to 98 years of age) and clean (131/303, 43.2%) and clean-contaminated (148/303, 48.8%) wounds predominated again (Table 1). The most frequently used antibiotics were cefalotin in 43.3% (94/217), cefotaxime in 22.1% (48/217), metronidazole in 13.3% (29/217) and ceftriaxone in 9.6% (21/217). The most frequent operations were caesarean section in 14.5% (44/303), vaginal delivery in 3.9% (12/303), cholecystectomy in 4.9% (15/303) and craniotomy in 5.9% (18/303).
Assessment of the nature of prescribing by the entire sample in this stage of the study found that the antibiotic regimen was inappropriate in 14.4% (31/215), began with inappropriate timing in 84.1% (180/214), the regimen was inappropriate in 75.3% (162/215), appropriate dosage in 51.2% (110/215) and of inappropriate length in 83.9% (178/212), and restricted antibiotics were used in 8.3% (18/215) (Table 2). In the analysis per type of hospital, antibiotic prophylaxis was inappropriate in 7.5% (4/53) of the HPSS patients, in 23.3% (18/77) of the HPRIV patients, and in 10.5% (9/85) of the HPNoSS patients (p=0.018). Prophylaxis began with inappropriate timing in 86.8% (46/53) of the HPSS patients, in 89.4% (68/76) of the HPRIV patients, and in 77.6% (66/85) of the HPNoSS patients (p=0.101). In most cases, it was started after surgery in 65.2% (30/46) of the HPSS patients, in 83.8% (57/68) of the HPRIV patients, and in 80.9% (51/63) of the HPNoSS patients (p=0.049). Inappropriate prophylactic regimens were used in 88.6% (47/53) of the HPSS patients, in 74% (57/77) of the HPRIV patients and in 68.2% (58/85) of the HPNoSS patients (p=0.024). Inappropriate dosages were prescribed in 24.5% (13/53) in the HPSS patients, in 51.9% (40/77) of the HPRIV patients, and in 67% (57/85) of the HPNoSS patients (p<0.001). The length of prophylaxis was inappropriate in 82% (41/50) of the HPSS patients, in 83.1% (64/77) of the HPRIV patients, and in 85.8% (73/85) of the HPNoSS patients (p=0.812). Use of restricted antibiotics was 11.3% (6/53) in the HPSS patients, 14.2% (11/77) in the HPRIV patients and 1.1% (1/85) in the HPNoSS patients (p=0.007) (Table 3).
When comparing the nature of prescribing before (1st survey) and after (2nd survey) the intervention, a reduction of 2.5% in the inappropriate use of prophylaxis was observed (16.9% vs 14.4%, p=0.436), of 11.9% in the frequency of inappropriate timing (96% vs. 84.1% before and after, respectively, p<0.001), and of 7.5% in the use of inappropriate regimens (82.8% vs 75.3%, p=0.034), of 26.5% in inappropriate dosages (77.7% vs 51.2%, p<0.001), of 2.1% in inappropriate length of prophylaxis (86% vs. 83.9%, p=0.515) and of 9.1% in prescribing restricted antibiotics (17.4% vs 8.3%, p=0.003) (Table 2).
Evaluation of the impact of the intervention per type of hospital found a 12% reduction in the inappropriate prescription of antibiotic prophylaxis in the HPNoSS (22.5% vs 10.5%, p=0.029). An 8.3% reduction in inappropriate treatment commencement was observed in the HPSS (95.1% vs 86.8%, p=0.064), 6.6% in the HPRIV (96% vs 89.4%, p=0.082), and 19.3% in the HPNoSS (96.9% vs 77.6%, p<0.001). A 28.5% reduction in the prescription of inappropriate dosages was observed in the HPSS (53% vs 24.5%, p=0.001), 35.3% in the HPRIV (87.2% vs. 51.9%, p<0.001), and 25.7% in the HPNoSS (92.7% vs 67%, p<0.001). A 19.3% reduction was observed in the use of restricted antibiotics in the HPSS (30.6% vs 11.3%, p=0.008), 4.2% in the HPRIV (18.4% vs. 14.2%, p=0.459) and 1.9% in the HPNoSS (3% vs. 1.1%, p=0.384) (Table 3).
DiscussionSpecific guidelines for surgical antibiotic prophylaxis have been created in the past, however, several studies reveal that inappropriate use and length of antibiotic regimens in this area are a worldwide problem.19 Not only does this increase the risk of surgical site infection with the consequent higher morbidity and treatment costs, but it also contributes to increased antimicrobial resistance.15–18 In the case of surgical antibiotic prophylaxis, in addition to creating guidelines for antibiotic use that are acceptable to surgeons, most studies have sought to develop targeted interventions to improve physicians’ adherence to these guidelines.19–23 This study assessed the use of antibiotics and the impact of a targeted intervention to change surgical prophylaxis prescribing behaviour in 6 of the largest hospitals of the metropolitan area of Monterrey, in north east Mexico, the country's third largest metropolitan area, which to the knowledge of the authors is the first study of this kind performed in Mexico.
Unlike other international studies,9,11,13,19,22–27 3 different types of institutions offering surgical services were included and compared in this study: HPSS, HPRIV and HPNoSS. In the initial survey inappropriate prescribing of antibiotic prophylaxis and at inappropriate dosages was less frequent in the HPSS. By contrast, restricted antibiotics were prescribed more frequently in these types of hospital. A large proportion of the rest of the prescribing characteristics assessed were inappropriate in all 3 hospital types. The lower frequency of inappropriate prophylaxis prescribing and at inappropriate dosages in the HPSS might be because they are training hospitals, where the resident doctors’ clinical activity is supervised by a specialist most of the time. Although the 2 NPNoSS were also teaching hospitals, it is possible that supervision was shorter during the day. Furthermore, the more frequent prescription of restricted antibiotics in the HPSS might be because the patients do not have to buy the drug directly.
The number of patients and the variables assessed in the initial survey presented in this study are similar to those reported in other studies.9,11,13,19,22–27 Nevertheless, the number of patients assessed in the various international studies has been very variable: ranging from 150 in a Canadian study to 35,543 in a national study in the U.S.A.9,11,13,19,22–27 This study is the first to evaluate surgical antibiotic prophylaxis in a large patient sample in Mexico. This is important because the results obtained enable a vision of how surgical antibiotic prophylaxis is used in the north east of Mexico. The surgical antibiotic prophylaxis guide used in this study was constructed and validated with the participation of 4 infectologists based on a thorough review of the international literature, including other countries’ guidelines; therefore it is very similar to other international guidelines.6,13,15–17
The local guide designed for this study is different in that, in some series, prophylaxis for 48h after heart surgery was considered an appropriate regimen length, whereas, in this study, antimicrobial prophylaxis for 24h following these types of operations was considered appropriate.12,13,17,22,24–26 Another difference is that while the published consensuses recommend the use of cefazolin and cefuroxime, in this guide cefalotin is recommended, a first generation cephalosporin with pharmacological and antimicrobial characteristics similar to cefazolin, since the latter is not available on the Mexican market.6,9,13,16,17 Another difference from other studies is that the age range of the patients assessed here is very wide. Patients of all ages were included, from newborns to elderly adults.9,11,13,19,22–27 As in other studies, this study assessed patients who had undergone different surgical interventions.11,22–26
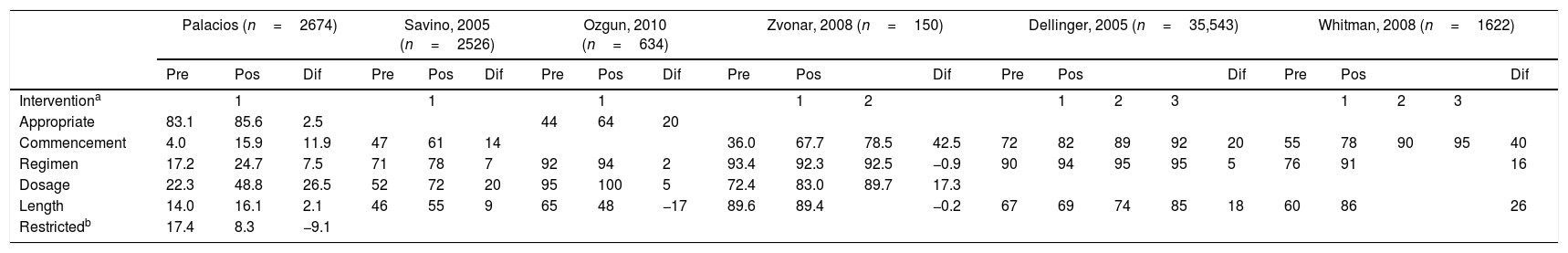
On comparing the results of this study with those reported by other international authors, it is evident that surgical antibiotic prophylaxis is a serious problem in Mexico, with inappropriate prescribing taking place in more than 90% of patients (Table 4). It is worth mentioning that this study assessed whether antibiotic use was appropriate and whether restricted antibiotics were being used, whereas other studies, although they assessed 3 or 4 different prescribing characteristics (start, regimen, dosage and length of regimen), did not look at these factors.9,11,13,19,22–27 Unlike most of the recent studies on surgical antibiotic prophylaxis in developed countries, which present their results as percentages of appropriate use, this study presents the results as percentages of inappropriate use, since these were high. The percentages of inappropriate antibiotic use in treatment commencement, regimens, dosages, and length of prophylaxis were much higher than those reported by other authors.9,11,13,19,22–27 In this study, 96% of the prescriptions were started with inappropriate timing, even after surgery in the majority of cases. This is in contrast with the 43%, 45% and 50%, reported by Bratzler et al.9 and Whitman et al.22 in the United States and van Kasteren et al.19 in Holland, respectively. It has been demonstrated that starting prophylaxis one hour before the surgical incision reduces the incidence of surgical site infection.6–9,11,12,16,17,25,28,29 Commencement at the appropriate time enables the antibiotic to achieve suitable levels in the surgical site at the time of incision. This benefit was lost in most of the patients in this study, since for the majority antibiotic prophylaxis was started after the operation had ended (Table 4).
Comparison of the impact of the interventions of 6 studies intended to improve surgical antibiotic prophylaxis.
| Palacios (n=2674) | Savino, 2005 (n=2526) | Ozgun, 2010 (n=634) | Zvonar, 2008 (n=150) | Dellinger, 2005 (n=35,543) | Whitman, 2008 (n=1622) | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pre | Pos | Dif | Pre | Pos | Dif | Pre | Pos | Dif | Pre | Pos | Dif | Pre | Pos | Dif | Pre | Pos | Dif | ||||||
| Interventiona | 1 | 1 | 1 | 1 | 2 | 1 | 2 | 3 | 1 | 2 | 3 | ||||||||||||
| Appropriate | 83.1 | 85.6 | 2.5 | 44 | 64 | 20 | |||||||||||||||||
| Commencement | 4.0 | 15.9 | 11.9 | 47 | 61 | 14 | 36.0 | 67.7 | 78.5 | 42.5 | 72 | 82 | 89 | 92 | 20 | 55 | 78 | 90 | 95 | 40 | |||
| Regimen | 17.2 | 24.7 | 7.5 | 71 | 78 | 7 | 92 | 94 | 2 | 93.4 | 92.3 | 92.5 | −0.9 | 90 | 94 | 95 | 95 | 5 | 76 | 91 | 16 | ||
| Dosage | 22.3 | 48.8 | 26.5 | 52 | 72 | 20 | 95 | 100 | 5 | 72.4 | 83.0 | 89.7 | 17.3 | ||||||||||
| Length | 14.0 | 16.1 | 2.1 | 46 | 55 | 9 | 65 | 48 | −17 | 89.6 | 89.4 | −0.2 | 67 | 69 | 74 | 85 | 18 | 60 | 86 | 26 | |||
| Restrictedb | 17.4 | 8.3 | −9.1 | ||||||||||||||||||||
Furthermore, antibiotic prophylaxis was given to the patients in this study for longer than indicated in the local guide and in the international guides. It has been demonstrated that in most clean and clean-contaminated operations where antibiotic prophylaxis is indicated, one single dosage is sufficient, and should the surgery time exceed 2 half-lives of the antibiotic (around 4h) a second dosage is sufficient.6,8,13,16,17,25 Prolonged, unnecessary use of antibiotics increases the risk of antimicrobial resistance and adverse effects,9–21,24,25 but prolonged administration of antibiotics together with the fact that most of the regimens prescribed were inappropriate – some included broad spectrum antibiotics – placed the patient at risk should they have incurred a surgical site infection caused by a multi-resistant organism. In this study several of the antibiotics prescribed for surgical prophylaxis were restricted (17%). These should only be prescribed by an infectious disease specialist and, of course, are not appropriate for surgical prophylaxis. Only one of the international studies on surgical antibiotic prophylaxis that the authors reviewed assessed this type of antibiotic use. This was the national survey conducted in the USA by Bratzler et al.9 in which almost 50% of the patients who were given vancomycin had no documented allergy to betalactamines: the primary indication for this antibiotic was for surgical prophylaxis.
In line with the standards for choosing an antibiotic, a first generation, and in some cases a second generation, cephalosporin is preferred for most patients. Third or fourth generations are not recommended. The published consensuses recommend cefalotin or cefuroxime.6,9,13,16,17 This contrasts with the results of this study where cefalotin, cefotaxime, ceftriaxone and ciprofloxacin were the most frequently used antibiotics. In the studies undertaken by Bratzler et al.,9 Pons-Busom et al.,13 Meeks et al.27 and Ozgun et al.,23 the most used antibiotic was cefazolin or cefuroxime. Cefazolin is not available on the Mexican market, cefalotin is used instead, a first generation cephalosporin with similar pharmacological and antimicrobial characteristics.
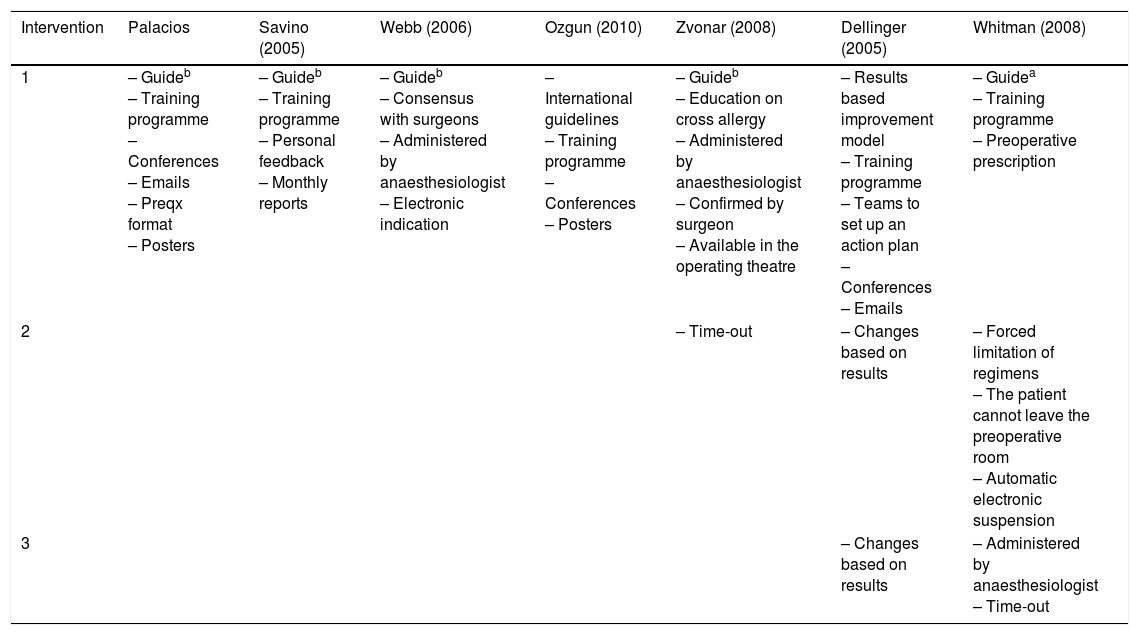
The impact of an intervention intended to improve the use of antibiotics for surgical prophylaxis was also assessed in this study. Zvonar et al.,25 in Canada, applied an intervention intended for that purpose and assessed its impact on 150 patients in 3 audits. Whitman et al.,22 in Philadelphia, by contrast, evaluated the impact of a similar intervention on 1622 patients. In a national study in the United States, Dellinger et al.26 also assessed the impact of an intervention of this nature on 35,543 patients. For the purposes of this discussion, we reviewed 6 studies published internationally that undertook an intervention intended to modify the prescribing behaviour of surgical antibiotic prophylaxis in order to compare them with this study (Table 5). Most of these studies started by creating a local surgical prophylaxis guide from a review of the literature and the consensus of infectologists and surgeons, as did this study.11,13,22–26 Although we have mentioned that the basis for a successful programme is to adopt local antimicrobial prophylaxis guides, compliance with them can still be suboptimal.13 In 4 of the studies the intervention involved learning sessions similar to those of this study.22–24,26 In 2 of the studies, the teaching process continued on an individual basis through email, as in this study.24,26 Several authors have stressed that the educational component is more effective when reinforced through informal networks and meetings, placing the emphasis on models aimed at changing behaviour.24 The policy of giving the antibiotic linked to timeout, in order to ensure that all the operating room staff are aware that prophylactic antibiotic has already been administered was included in 3 interventions.22,24,25 Unlike this study, in 3 of the studies the interventions included delegating the responsibility for administering the prophylactic antibiotic to the anaesthesiology department, which improved the commencement time in the studies performed by Zvonar et al.25 (36% vs 78.5%, pre and post intervention, respectively), Whitman et al.22 (55% vs 95%) and Webb et al.11 (51% vs 98%) (Table 4).
Comparison of the interventions in 7 studies intended to improve surgical antibiotic prophylaxis.
| Intervention | Palacios | Savino (2005) | Webb (2006) | Ozgun (2010) | Zvonar (2008) | Dellinger (2005) | Whitman (2008) |
|---|---|---|---|---|---|---|---|
| 1 | – Guideb – Training programme – Conferences – Emails – Preqx format – Posters | – Guideb – Training programme – Personal feedback – Monthly reports | – Guideb – Consensus with surgeons – Administered by anaesthesiologist – Electronic indication | – International guidelines – Training programme – Conferences – Posters | – Guideb – Education on cross allergy – Administered by anaesthesiologist – Confirmed by surgeon – Available in the operating theatre | – Results based improvement model – Training programme – Teams to set up an action plan – Conferences – Emails | – Guidea – Training programme – Preoperative prescription |
| 2 | – Time-out | – Changes based on results | – Forced limitation of regimens – The patient cannot leave the preoperative room – Automatic electronic suspension | ||||
| 3 | – Changes based on results | – Administered by anaesthesiologist – Time-out |
On comparing the nature of prescribing pre and post intervention in all of the hospitals included, a significant reduction in 4 of the 6 areas evaluated was observed: treatment commencement, regimens, inappropriate dosages, and overuse of restricted antibiotics. The impact of the intervention with regard to the antimicrobial regimens prescribed and dosages was similar to other studies published internationally (Table 4),11,22–26 but was much lower with regard to the commencement of prophylaxis: an improvement of 11.9% in this study compared to 47%, 42% and 40% in the studies undertaken by Webb et al.,11 Zvonar et al.25 and Whitman et al.,22 respectively. The little impact it had on the length of prophylaxis administration might be due to the physicians’ fear that the patient might become infected if the antibiotic were to be discontinued early. If so, this fear is unfounded, since it has been demonstrated that most surgical site infections are acquired at the time of surgery and not afterwards, and therefore it is more important to start the antibiotic at the appropriate time to ensure that the tissues are impregnated with antibiotic when the surgical incision is made.6,8,12,13,17 Even so, Savino et al.24 only managed to improve this area by 9%, Zvonar et al.25 did not manage to improve it, and the study by Ozgun et al.23 observed a reduction of 17% in this area (Table 4).23–25 Ozgun et al.23 found a reduction of 8% in the inappropriate prescribing of antibiotics.27 It is probable that the low impact observed in that study and in this study might be due to physicians’ habit of using antibiotics even in operations that do not require them, due to the unfounded fear that the patient will develop a surgical site infection. Finally, in the study by Ozgun et al.23 overall adherence (prescribed antibiotic, appropriate antibiotic, appropriate dosage and appropriate length) rather than increasing, reduced by 5.7% after the intervention; it is also the only study to assess the financial impact and, despite the reduction of one dollar (US) in the average cost of antimicrobial prophylaxis, the total cost of the irrational use of antimicrobial prophylaxis is in excess of 26,000 dollars (USA).23
The impact that the intervention achieved on physicians’ antibiotic prophylaxis prescribing behaviour could be improved. The total participation in this intervention was more than 180 doctors in the 6 hospitals. If this number were to increase, and if the number of sessions were also to increase, prescribing behaviour could be improved still further. Although placing posters or signs and including a record sheet in the clinical record was a recommendation, only 4 of the 6 hospitals followed it. If the 6 hospitals had participated more actively in this area, the impact would also have been greater. The problem of the inappropriate prescription of surgical antibiotic prophylaxis in these hospitals requires constant intervention, which includes a good proportion of resident doctors. For 2 reasons: in the HPSS and in the HPNoSS, these are the doctors who are most frequently involved in prescribing prophylaxis and it is they who will be in charge of the health systems of the future.
ConclusionsThe results of this study show that in the metropolitan area of Monterrey:
- •
Inappropriate surgical antibiotic prophylaxis is a prevalent problem.
- •
Inappropriate regimens, started with inappropriate timing and of a longer than required duration are an issue that must be dealt with promptly.
- •
This problem must be causing not only a substantial increase in treatment costs, but must also be increasing antibacterial resistance.
- •
Intervention intended to change surgical antibiotic prophylaxis prescribing behaviour improved 4 of the variables relating to this activity. Therefore, the implementation not only of local and national guides but also targeted interventions to change this behaviour in medical personnel, as used in this study, are a real need that must realised.
- •
Periodic and ongoing application of this type of strategy must be reinforced and delivered to more of the staff involved and those in charge of health care units.
The authors declare that the procedures followed were in accordance with the regulations of the relevant clinical research ethics committee and with those of the Code of Ethics of the World Medical Association (Declaration of Helsinki).
Confidentiality of dataThe authors declare that they have followed the protocols of their work centre on the publication of patient data.
Right to privacy and informed consenThe authors have obtained the written informed consent of the patients or subjects mentioned in the article. The corresponding author is in possession of this document.
Conflict of interestThe authors have no conflict of interest to declare.
Please cite this article as: del Carmen Palacios-Saucedo G, de la Garza-Camargoa M, Briones-Larab E, Carmona-Gonzáleza S, García-Cabelloa R, Islas-Esparzac LA, et al. Evaluación del uso de antibióticos e impacto de una intervención dirigida a modificar la conducta prescriptiva en profilaxis quirúrgica en 6 hospitales del área metropolitana de Monterrey. Cir Cir. 2017;85:459–470.