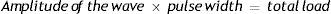Programming an implant in a patient follows a standardized and individualized protocol. Although this process uses common parameters in most cases, in a percentage of cochlear implant users, for some reason, it is not possible to establish appropriate levels of stimulation. In these patients, the audiologist has to make adjustments in some parameters, such as a change in the strategy, stimulation rate, pulse width, in order to obtain the expected performance.
La programación de un paciente implantado resulta un proceso inicialmente estandarizado, pero que debe individualizarse para cada caso. En la mayoría de las ocasiones, dicho proceso implica el establecimiento de parámetros comunes; sin embargo, existe un porcentaje de usuarios en los que por alguna circunstancia no es posible establecer niveles adecuados de estimulación, por lo que el médico audiólogo tiene que realizar ajustes en parámetros especiales como son: cambio de estrategia, tasa de estimulación, ancho de pulso, con el fin de obtener undesempeño auditivo de acuerdo a lo esperado.
A cochlear implant (CI) is an electronic device that provides a hearing sensation and replaces the function of the damaged or absent ciliated cells in the inner ear; it provides a previously processed electrical stimulus, which is distributed in a tonotopic way to the nerve fibers that emerge from the cochlea.1
Currently, after an experience of more than 60,000 cochlear implants worldwide, this technique can be considered as non-experimental since its efficacy in the treatment of profound hearing loss has been demonstrated.2
In the Mexican health care system, the cochlear implant has become an efficient and available treatment for the population. When placed in the earlier stages of life, it enables the hearing sensation and language in pediatric patients. For this reason, it is of utmost importance that the health personnel are familiar with this device and its operation.
The cochlear implant provides functional hearing and improves the levels of understanding of the language in the majority of patients with postlingual hearing loss, and allows the acquisition of spoken language in children with pre-lingual hearing loss, an improvement in the quality of life of patients with severe and profound hearing loss.
Programming an implant in a patient follows a standardized protocol. Moreover, it must be performed in an individualized manner for each patient, in order to establish the range in which it is possible to detect and discriminate the sounds of the human voice regarding the spoken language. However, in a percentage of cochlear implant users, for some reason, it is not possible to establish adequate levels of stimulation, thus remaining at sub-optimal levels of hearing. In these patients, the audiologist has to make adjustments in some parameters, such as change of strategy, stimulation rate, and the width of the pulse, among others.
2EpidemiologyThe World Health Organization (WHO) defines a disabling hearing loss as the one consisting of average audiometric thresholds greater than 40 dBHL, because of its impact on the development of spoken language, cognition, behavior and academic performance. In 2012, the WHO reported that there are nearly 275 million people around the world with moderate or profound hearing impairment, of which 32 million are children under 15 years of age. Two-thirds of these patients live in developing countries and 25% of them suffer from hearing loss since childhood.3
Every year, 5,000 children with bilateral permanent hearing loss are born in the United States. An incidence between 1/900 and 1/2,500 cases of permanent bilateral congenital hearing loss is estimated in its different grades: moderate, severe or profound.4
In Mexico, a study by the National Institute of Statistics and Geography (INEGI) in 2000 revealed that 2.8/1000 people have disabling hearing alterations; thus, 200,000 total hearing impaired patients are estimated, of whom 10% may be candidates for cochlear implantation.1
According to data from the annual survey of the European Association of Cochlear Implant users, in 2009 there were 7500 users of this device in Spain, of whom 4.412 were children and 3.888 were adults.5
In Mexico, during the last five years, the program of cochlear implant has had a major boost. In 2004, 520 patients were implanted. This number increased to 1500 patients in 2006.1
3Cochlear implantEvery CI has the following common features:
- •
A microphone to capture sound and transform it into electric signals
- •
A sound processor that encodes the electric signals with a battery
- •
A transmission system or coil that communicates the processor with the implanted internal components
- •
A receptor-stimulator antenna
- •
A flexible electrode array that is surgically implanted in the cochlea
- •
The microphone, the sound processor, the batteries unit, and the transmission coil form the external parts of the CI system; the receptor/stimulator device and the electrode array constitute the internal parts2,6
The main objective of the programming is the calibration of the CI to restore the hearing within the range of the human voice. Therefore, it is necessary to establish the parameters through which the device will transform the acoustic signals into electric signals, which will be sent to the auditory system for their processing. To achieve the latter, it is essential to select the mode and the strategy of stimulation, activation of microphones, rate of stimulation, number of maximum volumes, sensitivity and activation of the different electrodes, among other parameters.2
A strategy of stimulation is defined as a set of rules that delimit the form in which the sound processor analyzes the acoustic signals and encodes them for their delivery to the CI. There are large differences in the processing of the signal used by the strategies of stimulation of the latest systems of CI; some clinical trials show that the performance is similar between the existing brands. After the selection of the strategy, the creation of the auditory map, which is basically determined by the electrical stimulation threshold and the comfort threshold, should be followed.2
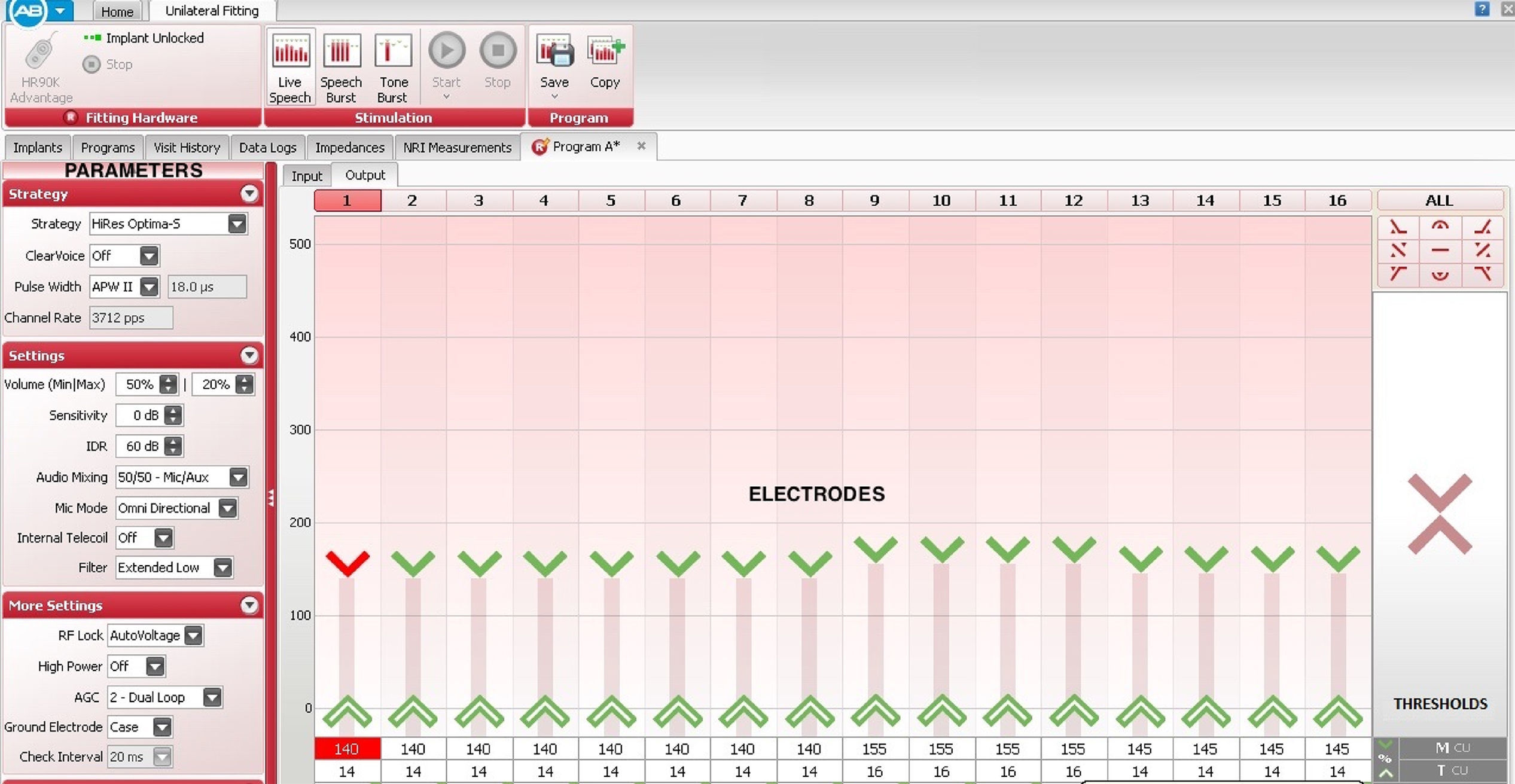
An auditory map is an individualized hearing program that is created and stored in the sound processor of the implant; the software consists of a graphical representation of the electrodes in the implant displayed on a computer screen in progressive order. The electrodes correspond to the sequential order in which they are placed inside the cochlea, but not necessarily in the order in which they are stimulated, since it can be modified1,7–9(Figure 1).
Thresholds vary in each implanted patient since the amount of electric current required to trigger an auditory sensation is different for each patient and depends on many circumstances, which require an individualized programming performed by the audiologist.
4.1Adjustment of threshold stimulationThe electric stimulation threshold refers to the lower amount of stimulation that the user of the CI can detect when the electrical signals (typically biphasic electrical pulses) are released to the electrodes. The precise, accurate definition, the name of the electrical stimulation threshold and the units in which it is measured vary depending on the manufacturer.
The stimulation threshold must be set accurately to provide better access to low-level sounds; it is possible to determine it for the majority of children through the use of appropriate methods of conditioning and observation in accordance with the age of the patient. These techniques include behavioral observation audiometry (BOA), visual reinforcement audiometry (VRA), conditioned play audiometry (CPA), and standard audiometric techniques for adults.8
4.2Setting the comfort thresholdIt refers to the level of energy power that is perceived as a sound of high intensity but comfortable to the user.1
The establishment of a comfort threshold is one of the most difficult aspects of the programming process. The sub-stimulation can sacrifice the recognition of the voice, hinder the quality of the sound, and limit the skills of self-control of the patient's voice; the over-stimulation can cause discomfort, hamper the recognition of voice, affect the quality of the sound, and foster an aversive reaction to the implant.2
The programming of the CI is not only based on the parameters described above. It is widely known that the perception of sound is significantly influenced by other variables, including pulse amplitude, the duration of the phase or pulse-width, the stimulation rate, the number of electrodes stimulated at the same time, the electrode coupling mode, and the configuration of the electrodes, among others. It is crucial for the audiologists to know the definition and management of the different programming parameters of the CI that affect the intensity of the coded signal.10
5Parameters that affect the signal strength5.1Dynamic rangeThe electrical dynamic range is defined as the difference between the thresholds of perception and the most comfortable level of electrical stimulation for a CI user.
In current CI, the dynamic range is between 20-30dB SPL and 65-85dB SPL depending on the manufacturer.
The lower threshold of the dynamic range determines the level of acoustic input assigned near the stimulation threshold, while the upper threshold determines the level of acoustic input assigned near the comfort threshold.
5.2SensitivityThis parameter controls the gain provided by the microphone of the sound processor. Subsequently, the most silent sound picked up by the microphone is determined.11
Sensitivity determines the input of the minimum signal required for the stimulation. To greater sensitivity, less acoustic energy (SPL levels) are required to cause stimulation. On the contrary, with a lower sensitivity, it is necessary to apply more acoustic energy (higher SPL levels) for the stimulation.7 The sensitivity control makes adjustments to the input signal before the analysis of the frequency, so its effect is similar across the range of the processor frequency. The numerical values of this parameter vary depending on each manufacturer.11
5.3VolumeIt refers to the parameter that controls the perception of loudness of the signal. For the three available brands of CI, a change in the volume directly produces a change in the high levels of stimulation.
5.4Rate of stimulationAlso called rate of repetition, it refers to the number of biphasic electrical pulses received at each electrode per second (pps).7,9
The first multi channel CI had relatively low rates of stimulation (250pps) However, current systems allow very high stimulation rates of up to 5000pps11. The total rate of stimulation of the CI is obtained from the product of the stimulation by electrode rate by the number of stimulated or active electrodes.
The optimum rate of stimulation may vary depending on the hardware of each CI. To optimize the delivery of electric pulses to a high rate, the current sources of the internal device of the CI should be capable of generating narrow pulse-widths with high current amplitudes. There is an inverse relationship between pulse-width and stimulation rate: high stimulation rates require narrower pulse-widths. The optimum stimulation rate is set depending on the user and the CI.11–13 The audiologist may use default values or alternatively select a stimulation rate.7,9
Theoretically, high rates of stimulation provide benefits in terms of speech discrimination. It has been observed that rapid changes in the pulse amplitude, coupled with an equal rate of pulses per second by the electrode, provide sufficient information to represent the spectral envelope of speech and hearing, and provide fine temporal and structural information in order to discriminate between different phonemes.
5.5Pulse-widthIt is the duration of the phase of the biphasic wave of each pulse, expressed in microseconds (μs), which translates as the amount of time that the stimulator provides electric current in every phase of an impulse.9
The pulse-width is one of the parameters that determine the load that the CI will receive. The total magnitude of a biphasic electric impulse is determined by the amplitude of the current and the pulse width with the following formula:
It is expressed in units of charge.
There are many publications about the effects of the modification of the length of the pulse phase, also called pulse-width. In 1985, R. Shannon concluded that as the pulse duration increases, the load threshold increases, first to a maximum of 400μs/phase, and then decreases as the length of the pulse increases even more. Shannon also noted that for short pulses (100-400μs / phase) with low stimulation rates (less than 100 pps), threshold waves are almost flat. In other words, the threshold is not affected by the frequency of the pulse for rates below 100 pulses / s. However, in pulse frequencies over 100 pps, the threshold level decreases approximately to 3dB/octave.14
It is plausible that by altering some of the properties of the stimuli, such as the rate of stimulation, the amplitude or the pulse width, the properties of temporary integration can be improved and promote decreases in the behavioral response thresholds. Davids et al., in 2008, altered the pulse duration and achieved a decrease in behavioral thresholds but not in the electrophysiological thresholds in the studied patients.15
6Clinical changes of pulse-width applicationsModifications in the pulse-width constitute a tool for improving the clinical hearing threshold without modifying the current units; they can be used in patients in which the desired level of current is hampered by the CI limitations of compliance. Therefore, if a patient requires high levels of stimulation, the pulse-width can be increased and, as a result, the stimulation rates will decrease. The exact way in which the stimulation rate and the pulse-width are managed slightly vary in each CI system.7,11
A small percentage of cases presenting stimulation of the facial nerve secondary to the use of the CI (incidence of 0.31–14%) has been reported. In these patients, it is not possible to establish the desired power level, and in most cases, it is not necessary to perform a surgical procedure, but it is possible to benefit the patient by reprogramming the processor.16
One of the purposes of health professionals is to correct a pathology or minimize its consequences. Regarding disabling hearing loss, many technologic, electronic and biomaterial advances have been developed. The hearing rehabilitation with the CI has provided impressive results in less than 30 years of evolution, making possible for a patient with profound hearing loss to perceive sounds and to discriminate voices and spoken language, rehabilitating or even enabling cognitive and language skills in every treated patient.
Since the use of the CI has increased exponentially in the last 10 years in our country, it is adequate to sensitize and involve every health professional to achieve an efficient and timely multidisciplinary care and to turn every patient into a case of success.
Conflict of interestThe authors declare no conflict of interest of any nature.
Please cite this article as: Banda González RI, Castillo Castillo S, Roque Lee G. Parámetros de programación del implante coclear. Bol Med Hosp Infant Mex. 2017;74:65–69.