Tuberculosis (TB) remains a challenge because severe forms occur most frequently in children under five years old and the diagnosis is complex. The objective of this paper was to describe the clinical presentation, frequency, methods used to make the diagnosis and response to treatment in children with TB treated at a tertiary level hospital.
MethodsThe study was retrospective and descriptive of a cohort of consecutive cases treated from January 2010 to December 2013. Ninety-three medical records of children diagnosed with TB according to the definition of the NOM-006-SSA2-2013 were reviewed. The analysis was performed using descriptive statistics.
ResultsFrom 93 children, 58% were male (mean age of 7 years), 97% with a history of BCG vaccination, and 6% had contact with a TB case. The most frequent clinical forms were pulmonary (30.1%), lymph node (24.7%), miliary/disseminated (16.1%), meningeal (13%), and osteal TB (7.5%). The most common symptoms were fever and weight loss (50% and 40%, respectively). AFB stain and culture were positive in 26% and 7% of all cases, respectively. The histopathological study was conclusive in 90% of the cases. The treatment was successful in 94.6%, with not associated mortality.
ConclusionsThe association of clinical symptoms with alterations in chest radiography and positive PPD is useful in establishing the presumptive diagnosis and an early and appropriate treatment.
La tuberculosis (TB) continúa siendo un reto ya que las formas graves se presentan con mayor frecuencia en los menores de 5 años y el diagnóstico es complejo. El objetivo del presente trabajo fue describir las formas de presentación clínica, frecuencia, métodos de diagnóstico empleados y respuesta al tratamiento en niños con TB atendidos en un hospital de tercer nivel.
MétodosSe diseñó un estudio retrospectivo, descriptivo, de una cohorte de casos consecutivos atendidos desde enero de 2010 hasta diciembre de 2013. Se revisaron 93 expedientes clínicos de niños con diagnóstico de TB de acuerdo con la definición de la NOM-006-SSA2-2013. Se utilizó estadística descriptiva para el análisis.
ResultadosEl 58% de 93 niños fueron pacientes de sexo masculino con una media de edad de 7 años. El 97% contaba con antecedente de vacunación BCG; el 6% tuvo contacto con algún caso de TB. Las formas clínicas más frecuentes fueron la TB pulmonar (30.1%), ganglionar (24.7%), miliar/diseminada (16.1%), meníngea (13%) y ósea (7.5%). Los síntomas más comunes fueron fiebre y pérdida de peso (50% y 40%, respectivamente). El BAAR y el cultivo fueron positivos en el 26% y el 7% de todos los casos, respectivamente. El estudio histopatológico fue concluyente en el 90%. El tratamiento fue exitoso en el 94.6%, sin mortalidad asociada.
ConclusionesLa asociación del cuadro clínico con las alteraciones en la radiografía de tórax y PPD positivo son útiles para establecer el diagnóstico presuntivo e iniciar el manejo oportuno.
Tuberculosis (TB) is a chronic and infectious disease caused by a group of bacteria of the Actinomycetales order of the Mycobacteriaceae family. The Mycobacterium tuberculosis complex (M. tuberculosis, M. bovis, M. microti, M. africanum y M. canetti) is transmitted from the bacillifer host to the healthy subject by inhalation of infecting material, contaminated milk ingestion; less frequently, by contact of skin lesions with bacillifer subjects or sick bovines; and lastly, by vertical transmission.1 In 2014, the World Health Organization estimated that 9.6 million people were infected with tuberculosis and 1.5 million died of this disease. Around one million children got infected and 140 000 of them died.2
In countries with low incidence, TB in the pediatric age represents less than 5% of all TB cases.3 Meanwhile, in high incidence areas it represents 20-40%. In a prospective study in South Africa, it was observed that children younger than 13 years old represented 13.7% of the total regional burden of TB cases, with a calculated incidence of 407/100 000 people/year (half of the cases in adults).4
In TB endemic areas, children present advanced stages of the disease and TB represents a big percentage of morbidity and mortality in patients younger than five years old. A study in Zambia proved that TB was the major cause of death secondary to respiratory diseases in children with or without human immunodeficiency virus (HIV) infection.5,6
In 2015, 20 561 new TB cases were registered in Mexico; cases in people younger than 14 years old represented 3.7% (774 cases), of which 53.3% were pulmonary, 2.5% meningeal and 44% had another form of presentation.7,8 In 2009, another study in Mexican children found a similar proportion of boys and girls (1.1: 1) although TB reactivation was more frequent in teenage girls. Of the total of notified cases, 11.1% was associated with malnutrition, 0.9% with HIV infection, 0.7% with alcoholism and 0.6% with diabetes mellitus; in 77% of the cases, there were no concomitant diseases. In infants younger than one-year-old, malnutrition was the condition most associated with TB. It was estimated a national rate of 3.8/100 000 cases in patients younger than 18 years old. The states in the country with the highest TB rates were Baja California (18.5), Tamaulipas (7.9), Baja California Sur (7.5), Colima (7.5), Sonora (7.2), Guerrero (6.9), Chiapas (6.8) and Nayarit (5.7).7,8
Pulmonary parenchymal disease and adenopathy are the main manifestations of TB in the pediatric age in 60-80% of the cases.9 Among extrapulmonary manifestations, lymphadenopathy is the most common (67%), followed by central nervous system affections (13%) (meningitis, tuberculomas, abscesses), pleural (6%) miliary or disseminated (5%) and osteal (4%).10,11 In a case-series of 102 pediatric patients with extrapulmonary forms of TB, Maltezou et al. observed the following manifestations: lymphadenitis 47%, pleural effusion 26.5%, and meningitis 15.5%, osteal 5%, miliary 3%, abdominal 2% and pericarditis 1%.12 Pulmonary and extrapulmonary atypical presentations have been reported in adults as well as in children, although the clinical presentation is even less specific in malnourished and immunocompromised patients. Exclusive pulmonary affection presents in less than 50% of the cases and, in the rest, it is associated with exclusive or mixed extrapulmonary compromise. Ganglionic TB represents almost 40% of all extrapulmonary forms, while meningeal and disseminated (miliary) TB are more frequent in younger patients (< 3 years old) and in patients with HIV.13,14
These forms of clinical presentation vary in frequency depending on the studied population and other concomitant clinical phenomena such as malnutrition, HIV infection and other causes of immunosuppression in the population.
The south and southwest of the country are the areas with more influence of the disease.
The objective of this study was to describe different forms of clinical presentation, frequency, diagnostic resources used and therapeutic response to M. tuberculosis in pediatric patients. This study was conducted at a High Specialty Medical Unit, to where patients who require in-hospital medical care are referred.
2MethodsThis study carried out at a third level of attention medical unit that receives patients referred from the states of Morelos, Guerrero, Chiapas, Querétaro and the south region of Mexico City.
A retrospective cohort from January 2010 to December 2013 was followed. The study was descriptive and observational. We included consecutive cases from newborns to adolescents aged sixteen years 11 months who had the diagnosis of “disease by M. tuberculosis” according to the definitions from the NOM-006-SSA-2013 and whose clinical file information was complete. We created a database with demographic, clinical, diagnostic and therapeutic information. No informed consent letter was issued. We performed a descriptive statistical analysis with frequency measures for qualitative variables and mean or median (according to its distribution) for quantitative variables. We used SPSS® Statistics 20 software for the analysis.
The project was approved by the local Committee for Health Research 3603 of the Hospital de Pediatría, Centro Médico Nacional Siglo XXI, Mexico City, with the number R-2012-3603-12.
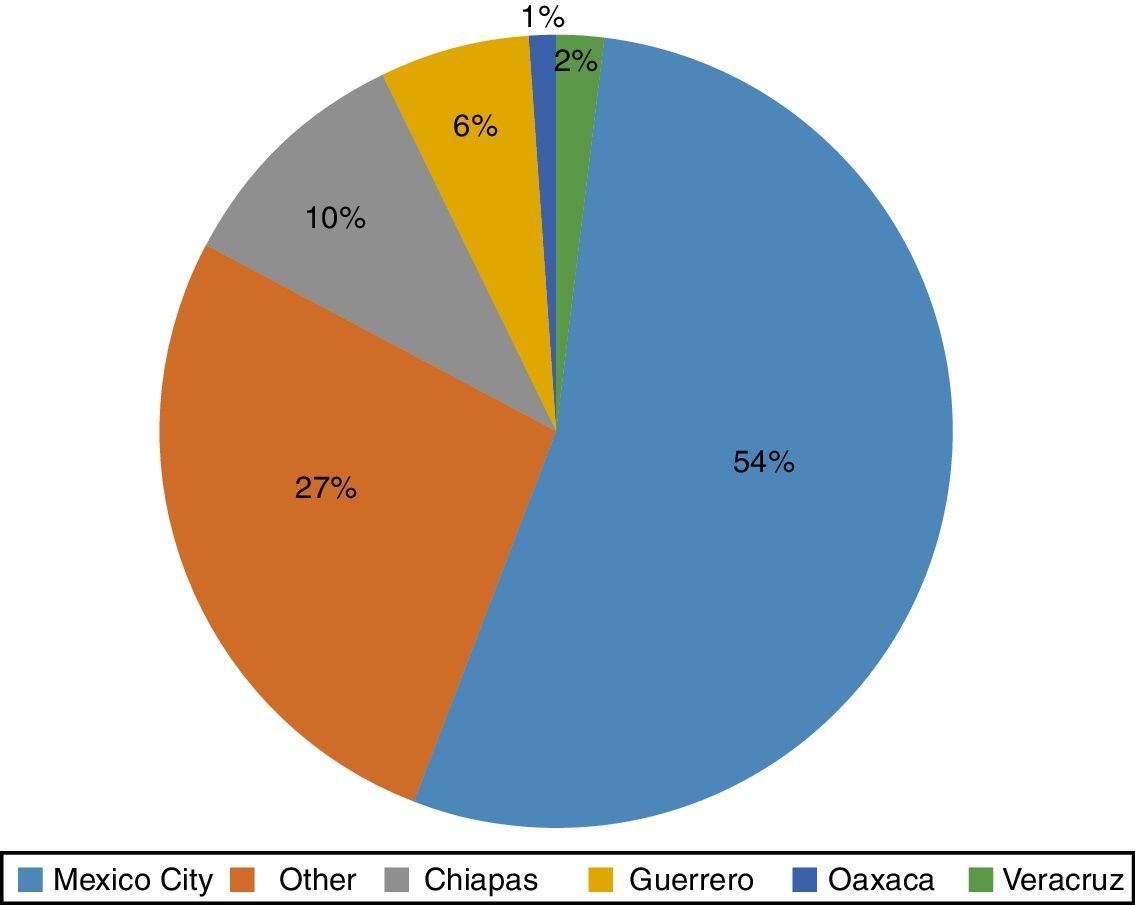
3Results3.1Demographic characteristicsWe included 93 patients with the diagnosis of TB according to the NOM-006-SSA2-2013 definitions, of which 58% were male. The age range was between 9 months and 16 years three months of age, with a median of 6 years 11 months. The place of origin is described in Figure 1.
Only three (3.2%) patients had not received the Bacillus Calmette-Guérin (BCG) vaccine. Epidemiological TB exposure was confirmed in only 6.4% of the cases. From 93 patients, 74.2% (69) were previously healthy; the rest presented comorbidities. In average, the patients were referred to the third level of attention three weeks after the beginning of their symptoms (range between 2-10 weeks).
3.2Clinical characteristicsThe most frequent clinical presentations of TB were pulmonary, in 28 patients (30.1%); ganglionic, in 23 patients (24.7%); miliary (disseminated), in 15 patients (16.1%); meningeal, in 12 patients (13%); osteal, in seven patients (7.5%); peritoneal/intestinal, in five patients (5.3%); cutaneous, in two patients (2.1%); and renal, in one patient (1%).
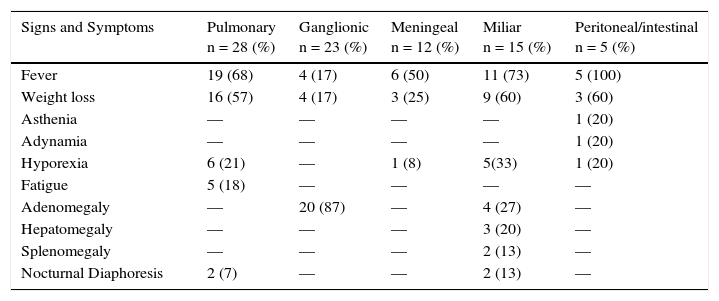
Fever was present in 50% of the patients and predominated in pulmonary (68%), miliary (73%) and meningeal (50%) forms; in the ganglionic form, the percentage of patients who presented fever was low (18%). Equally, weight loss predominated in the pulmonary and miliary forms (605 in both). However, hyporexia was reported only in 21% and 33%, respectively (Table 1).
Signs and general symptoms according to the clinical presentation of tuberculosis in pediatric patients.
| Signs and Symptoms | Pulmonary n = 28 (%) | Ganglionic n = 23 (%) | Meningeal n = 12 (%) | Miliar n = 15 (%) | Peritoneal/intestinal n = 5 (%) |
|---|---|---|---|---|---|
| Fever | 19 (68) | 4 (17) | 6 (50) | 11 (73) | 5 (100) |
| Weight loss | 16 (57) | 4 (17) | 3 (25) | 9 (60) | 3 (60) |
| Asthenia | — | — | — | — | 1 (20) |
| Adynamia | — | — | — | — | 1 (20) |
| Hyporexia | 6 (21) | — | 1 (8) | 5(33) | 1 (20) |
| Fatigue | 5 (18) | — | — | — | — |
| Adenomegaly | — | 20 (87) | — | 4 (27) | — |
| Hepatomegaly | — | — | — | 3 (20) | — |
| Splenomegaly | — | — | — | 2 (13) | — |
| Nocturnal Diaphoresis | 2 (7) | — | — | 2 (13) | — |
Respiratory affection was expressed by cough in almost 70% of the cases, accompanied by respiratory distress in 18% of the patients with pulmonary clinical manifestations, diaphoresis in 7%, and general fatigue in 18% of the patients.
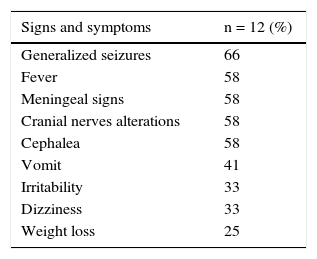
In the meningeal form, almost half of the patients presented endocranial hypertension manifested by cephalea (58%), vomit (41%), and an altered mental status (33%). In 58% of the patients (5/12), there were objective signs of meningeal irritability (Table 2).
On the contrary, in the ganglionic form, general symptoms such as fever and weight loss were infrequent (both with 17%). In the majority of the cases, adenopathy was the only sign, predominantly in the cervical region (70%), followed by the submandibular (45%) and axillary (13%) regions. In the miliary form, adenomegaly was also present in 26% of the patients; additionally, it was associated with hepatomegaly (20%) and splenomegaly (13%) besides general symptoms in most patients.
Observed sequelae were moderate neurologic damage in four patients and severe in one patient for the meningeal cases and short bowel syndrome in one case of the abdominal form.
3.3Medical diagnosisPurified protein derivate (PPD) was applied in 86/93 patients, of which 43 (50%) had an induration >10mm, five patients (6.3%) had an induration between 5-9mm and, 44.1% had no reaction at all. In patients with comorbidities (18) 27.7% had an induration >5mm; the median of the PPD reaction diameter in this group was 16mm. A booster dose was given to six patients previously classified as anergic but with suggestive clinical signs of TB (four were previously healthy). The PPD reaction diameter was 18mm, which was considered as positive.
Regarding conventional microbiologic documentation, acid-fast bacilli stain (AFB) was performed in 104 biological samples or body fluids (sputum, urine and pleural effusion). In some cases, more than one sample was obtained per anatomical site. Bacilloscopy was positive in only 27 samples: nine from gastric lavage, five from urine, four from sputum, three from pleural effusion, two from bronchioalveolar lavage, two from cerebrospinal fluid and two from pulmonary tissue.
Also, 85 biological samples from 60 patients were cultured. M. tuberculosis was isolated in six samples (7%) distributed as follows: one from sputum, one from gastric lavage, one from cerebrospinal fluid, one from pleural effusion and another source (pulmonary biopsy and abscess). In two out of six positive cultures, the initial AFB stain was negative (one from a pleural effusion and one from an abscess).
Polymerase chain reaction (PCR) was used for the analysis of twenty-two samples with amplification of M. tuberculosis nucleic acids in different fluids and tissues. Results were positive in 82% of the samples. In 94% of the positive samples, of which more than 50% were from cerebrospinal fluid, the Ziehl-Neelsen stain was negative.
In tuberculous meningoencephalitis cases, PCR was positive in 8 out of 12 cases.
As part of the non-microbiological diagnostic techniques, adenosine deaminase (ADA) was determined in five samples from different body fluids, of which only three from pleural effusions were positive (>40 U/l). Finally, 57 samples from 48 patients underwent histopathology analysis, of which 51 (89.4%) were reported with a TB compatible granulomatous reaction (Table 3).
Positivity of diagnostic tests in children with TB.
| TEST | PPD n = 86 | AFB stain n = 93 | Culture n = 60 | PCR n = 22 | Biopsy n = 48 | ADA n = 5 |
|---|---|---|---|---|---|---|
| Positive (frequency) | 43 | 27 | 6 | 18 | 43 | 3 |
| Percentage | 50% | 29% | 10% | 82% | 90% | 60% |
PPD, purified protein derivate; AFB, acid fast bacilli stain; PCR, polymerase chain reaction; ADA, adenosine deaminase.
All patients underwent chest X-rays. The most common radiographic pattern of pulmonary TB (n=20) was macronodular in 48%, micronodular in 17% of patients and consolidated pneumonia, pleural effusion and mediastinal widening in 13% of patients. Pachypleuritis and pulmonary nodules were found in 7% of the cases and caverns in only one case.
Sixteen children had a pulmonary CT scan. Pneumonia was observed in 39% of the patients, a micronodular pattern in 39%, perihilar lymphadenopathy in 26%, granulomas in 17%, caverns, and calcifications in 9% and pachypleuritis and pleural effusion in 4% of the cases.
In miliary TB cases, abnormalities in chest X-rays were found in eight patients; in the pulmonary region, a macronodular pattern in 40%, a micronodular pattern in 33%, nodules, pneumonia with pleural effusion and pachypleuritis in 7%, respectively. The CT scan showed pneumonia and micronodular pattern in 40%, ascites in 47%, retroperitoneal ganglia in 20%, granulomas in 13% and hepatomegaly in 7% of the cases.
In meningeal TB cases, hydrocephaly (5/10), basal arachnoiditis (4/10), infarction (3/10) and tuberculomas (2/10) were identified by a CT scan.
Based on clinical manifestations and epidemiological background, each patient with TB suspicion was given antifimic treatment before corroborating the diagnosis.
In 73 patients (78%), four anti-tuberculosis drugs were used in the intensive phase: isoniazid (H), rifampicin (R), pyrazinamide (Z), and ethambutol (E). In 3% of the cases, a triple antifimic treatment was used (HRZ), and HRE only in 1%. Average treatment duration in this phase was 4.4 months (range between 2 and seven months). The support phase was based on two drugs (HR) with an average duration of 7.4 months (range between four and 22 months).
Steroids were administered to 22.5% of the population, which corresponded to the meningeal and miliary forms. Full remission was present in 94.6% of the clinical forms (88 cases). However, 9.6% of the cases (9) did not remit with the first scheme (four ganglionic, two meningeal, one pulmonary, one disseminated and one cutaneous). Three patients (3.2%) had treatment failure and three relapsed after treatment. Average follow-up time was 14.8 months.
4DiscussionIn Mexico, M. tuberculosis infection is highly prevalent; even further, its prevalence has increased in the last decade: from 14 837 cases per year in 1990 to 16 907 cases in 2014, the majority of them with a pulmonary presentation mainly associated with AIDS. Most patients also have diabetes, malnutrition, and addictions. Prevalence is higher in the coasts, in the Pacific, including Sonora, Sinaloa, Guerrero, and Chiapas, and the Gulf of Mexico, including Tabasco, Veracruz, and Tamaulipas. States in the north and the south of the country have an average prevalence, and center states including the Bajío region have a lower prevalence. In this study, an important percentage of patients came from Mexico City, followed by the states of Chiapas and Guerrero. According to the Unique Platform of Information reports, the latter is the second state with the highest rate of TB in the pediatric population and Chiapas holds the sixth place in the country. These results were expected given that poverty and margination stigmas predominate in these regions of Mexico, which in turn are consequences of the prevailing socially inequality.15
In this study, the predominant age of all forms of TB was < 5 years old. This group of age corresponds to almost 50% of the cases, which is comparable with previous studies. A possible explanation for this pattern is the frequent exposure with the index case at early ages, with a higher frequency of pulmonary disease. This is also possible since BCG may confer a protective effect to the severe forms of extrapulmonary TB.
In this study, it should be noted that three cases did not receive the BCG vaccine. These cases corresponded to two extrapulmonary forms (miliary and meningeal), as well a pulmonary case. According to the 2012 National Health and Nutrition Survey, BCG vaccination coverage was higher than 90%, although some states have lower coverage levels.16 Rodrigues et al. observed that the protective effect of the BCG vaccine for miliary or meningeal TB was 86% (C.I. 95% 65-95) in controlled studies, and 75% (C.I. 95% 61-84) in case-controls studies.17 The lack of BCG efficacy to protect different forms of TB in children has been associated with a lower immunological experience in young patients, the lower protective effect of the BCG vaccine and the diversity of pathogenic mechanisms of the pulmonary disease (reinfection, reactivation, or primary progression).
It was found that more than 70% of the analyzed patients were healthy; the rest of them had at least some comorbidity, which coincided with previous reports.8 It is worth mentioning that in this study, there were none HIV-related TB cases, considering that the HIV epidemic has had a negative impact on TB control efforts.
In almost 90% of the cases, it was not possible to detect the source of infection, as referred by Winston and Collaborators.18 This denotes the delay in the search and identification of infected adults, which in turn contributes to the continuous exposure of non-infected individuals in risk of developing TB, and finally indicates that the chain of transmission is still active.
In this cohort, ganglionic TB was the most frequent extrapulmonary form, corresponding to 35.4% of the cases. The suspicion for the diagnosis was made on general symptomatology, PPD reactivity, and the presence of cervical calcified ganglia detected on a CT scan.12–14 It is worth mentioning that separate image studies do not categorically distinguish between tuberculous lymphadenitis and other causes of adenomegaly, mainly at a cervical level. It is interesting to highlight that in < 50% of the patients with cervical ganglionic TB showed abnormalities in their chest radiography; in this case-series, 26% was reported without alterations. The ultrasound is an excellent initial method to the diagnosis since it facilitates fine needle aspiration biopsies and allows evaluating the presence of adenomegalies. All patients underwent lymphadenectomy, which enabled a diagnosis to be confirmed by histopathology. In histological studies, the granulomatous pattern with caseous necrosis (71%) predominated, without the demonstration of the bacilli with stain or culture isolation. Gupta et al. analyzed 102 TB ganglionic compatible surgical samples and found the presence of AFB (32.2%) o microbiological isolation (56.9%) mainly in the necrotic material, as well as in the granulomatosis pattern with caseous necrosis (30% and 40%, respectively).19
In this analysis, 27% of the patients had severe forms of TB; the majority were younger than five years old, in whom symptomatology is unspecific. In the first level of attention, this disease is frequently unsuspected, and the lack of timely diagnostic studies may be the reason for the late documentation of this condition. In this group of age, many factors may risk the vaccine application. However, it has been demonstrated that its application in developing countries has contributed to diminishing the number of meningeal tuberculosis cases.
For the pulmonary form cases, traditional diagnostic tools were used, such as image studies, bacilloscopy and dermic reactions.
PPD test limitations should always be considered. In the pulmonary form, chest X-rays were abnormal in most cases. The macronodular pattern predominated, followed by the micronodular and, in a lower proportion, mediastinal adenopathy as well as consolidations. This differs from some literature reports where mediastinal affection is observed in 70% of the cases, followed by consolidations in 40% and 60%.20 It is advisable to complement image studies with a CT scan in the cases in which a suspicion of extrapulmonary affections exists, or to evaluate the extension of the parenchymal damage.
Meningeal TB is cataloged as an emergency; given the associated mortality and risk for sequelae, it is important to make a timely diagnosis, as well as the start of a specific treatment. In this study, besides fever in over 50% of the cases of meningeal TB, endocranial hypertension signs, cranial nerve affection, as well as seizures were observed, in general, in stages II (83.3%) and III (16.6%). These results differ from Coria et al.,21 who found a greater percentage of patients at stages II and I, with consequently better prognosis.
Regarding diagnosis methods, 25% of the patients had a positive PPD. This result is highly variable given that is positive in 10-20% in some studies, and up to 50% of the cases in others. Due to this variability, image studies are very useful for diagnosis.
The need for different tests to conclude a TB diagnosis is clear, although no test considered as the gold standard exists to date.22 To suspect TB in children, epidemiological background and clinical manifestations are fundamental. Consequently, if the first-contact physician has the appropriate diagnostic tools, he will be able to act promptly and avoid treatment delays and further patient damage.
In this case series, treatment success reached 94%, probably due to the lack of HIV cases in addition to a good treatment adherence. Treatment was well tolerated in most cases; there was only one case with transient hepatotoxicity, which resolved without management changes and another case of myelotoxicity secondary to ethambutol, which remitted after its withdrawal. In 17% of the patients, intensive treatment phase was prolonged, mainly in immunocompromised patients.
This study has limitations mainly due to its retrospective nature; also, the samples came from the third level of attention hospital. However, this case-series provides a landscape of the pediatric manifestations of TB in this environment.
The reinforcement of epidemiological surveillance is necessary to ensure timely detection and treatment of infected adults to decrease the incidence of TB in the pediatric population.
Ethical disclosuresProtection of human and animal subjectsThe authors declare that no experiments were performed on humans or animals for this study.
Confidentiality of dataThe authors declare that they have followed the protocols of their work center on the publication of patient data.
Right to privacy and informed consentThe authors declare that no patient data appear in this article.
FundingThis study did not receive funding.
Conflict of interestThe authors declare no conflicts of interest of any nature.
Please cite this article as: Vázquez RG, Acosta GC, Miranda NMG, Fuentes PYC, Labra ZMD, Pacheco RO, et al. Análisis de una serie de casos de tuberculosis en pacientes pediátricos atendidos en un hospital de tercer nivel. Bol Med Hosp Infant Mex. 2017;74:27–33.