Vitamin D deficiency is associated with increased susceptibility to infections and wheezing. We aimed to evaluate the relation between vitamin D levels, viral infections and severity of attacks in children with recurrent wheezing.
Materials and methodsA total of 52 patients who applied with wheezing, at the ages of 12–60 months with a history of three or more wheezing attacks in the last year and 54 healthy children were included. Sociodemographic data, risk factors for recurrent wheezing, and the severity of the wheezing attacks were recorded. 25(OH)D3, calcium, phosphor, alkaline phosphatase and parathormone levels of all children were measured. Nasopharyngeal samples of the patients for viruses were studied by multiplex polymerase chain reaction.
ResultsFor the patient group, being breastfed for six months or less, history of cesarean section, cigarette exposure, humid home environment, and family history of allergic disease were significantly higher compared with the control group. Serum vitamin D levels in the patient group were significantly lower compared to the control group. There was no significant relationship between vitamin D levels and hospitalization, oxygen or steroid therapy. Virus was detected in 38 patients (73%). Rhinovirus (63.2%) was the most frequently detected virus. Coinfection was found in 14 (36.8%) patients. There was no statistically significant difference between detection of virus and vitamin D levels.
ConclusionsCigarette exposure, being breastfed six months or less, humid home environment, history of cesarean section, family history of allergic disease and vitamin D deficiency might be risk factors for recurrent wheezing.
The incidence of asthma and allergic diseases is increasing, especially in childhood. There are different opinions about the relationship of vitamin D to asthma and wheezing. In some studies, vitamin D inadequacy was held responsible for the increase in asthma.1 In one study, low vitamin D intake during pregnancy has been shown to increase recurrent wheezing around five years of age.2 In another study, patients who received daily 2000 IU vitamin D regularly in their first year were found to have a higher risk of developing asthma compared to the group without vitamin D when they were 31 years of age.3 As seen in different studies, different results were found between the intake of vitamin D and the development of asthma. Larger and randomized studies are needed to clarify this issue.
It has been recently determined that rhinovirus is a significant contributor to wheezing illnesses in young children and in the context of asthma.4 Birth cohort studies have shown that atopic children have an increased tendency to develop asthma after rinovirus induced bronchiolitis.5,6 Some authors also speculated that pre-existing airway inflammation in children susceptible to asthma predisposes to rhinovirus infection.7 Lukkarinen et al.8 found that the first wheezing episode with rhinovirus predicted atopic asthma at school age. In a recent meta-analysis rhinovirus induced wheezing in the first three years of life was found to be related with subsequent development of wheezing/asthma.9
Although there are studies about vitamin D levels in children and adults with asthma, the number of studies in children with recurrent wheezing before the age of five is quite low.10 There are some studies investigating the relationship between wheezing and vitamin D,11 wheezing and viral infections,12 and viral infections and vitamin D.13 There are also a few studies evaluating viral infections, vitamin D and acute wheezing,14 but no study evaluating the three altogether was found in recurrent wheezing.
The aim of this study was to evaluate the relationship between serum vitamin D levels, viral infection and wheezing attack severity in children with recurrent wheezing.
Materials and methodsBetween June 2015 and December 2016 all patients who attended Adnan Menderes University Hospital Child Emergency, Child Immunology and Allergic Diseases and General Pediatric Polyclinics with wheezing attacks, at the ages of 12–60 months with a history of three or more wheezing attacks in the last year were included.
Patients with congenital structural anomalies in their lungs, cardiac and other systemic diseases, primary immunodeficiency, growth retardation and neurological disorders causing coordination disorder were not included in the study.
In the control group, 54 healthy children with similar age and gender who attended Child Health and Diseases Polyclinics whose families agreed to participate in the study were included.
Sociodemographic information, environmental conditions, wheezing attack severity, treatments given during the wheezing attack of the patient group; sociodemographic information and environmental conditions of the control group were also recorded after signing the written consent form for the guardians of all participants. Physical examination of all cases was performed. The wheezing attack severity of the patient group was performed according to Global Initiative for Asthma (GINA) criteria.15
In both patient and control groups two milliliter blood samples were acquired for biochemical analysis. The spectrophotometric method was used for determinations of serum calcium (Ca), phosphor (P), alkaline phosphatase (ALP) levels (ARCHITECT c8000, Abbott, Abbott Park, Illinois, USA). Serum 25(OH)D3 and parathormone (PTH) levels were analyzed with chemiluminescentmicroparticle immunoassay (CMIA) (ARCHITECT c8000, Abbott, Abbott Park, Illinois, USA).
A serum 25(OH)D3 level of >20 ng/mL is indicative of vitamin D sufficiency, a level of 15–20 ng/mL as vitamin D insufficiency, a level of <15 ng/mL as vitamin D deficiency, and a level of ≤5 ng/mL as severe deficiency for children.16
The detection of respiratory viruses in nasopharyngeal swab samples from patients was performed by FTD respiratory pathogens 21 kit (Fast-track Diagnostics Ltd., Malta) according to the manufacturer’s instructions. Each sample was simultaneously tested for human adenovirus (HAdV), influenza viruses A, B and H1N1 (FluA, FluB, H1N1), human coronaviruses (HCoV-229E, HCoV-HKU, HCoV-NL63, HCoV-OC43), human metapneumovirus (HMPV), human parainfluenza viruses (HPIV-1, HPIV-2, HPIV-3 and HPIV-4), human respiratory sinsitial virus (HRSV A/B), human enterovirus (HEV) and rhinovirus (RV). The isolation of nucleic acid from nasopharyngeal swabs was performed using an EZ1 virus mini-kit V2.0 (Qiagen, Germany) in the Molecular Microbiology Laboratory. Fast-track Respiratory Pathogens 21 kit detects respiratory tract pathogens in the RotorGene Q platform (Qiagen®, Germany). The thermal profile of multiplex real time polymerase chain reaction (mRT-PCR) for the FTD kit was as follows: 15 min at 50 °C, 10 min at 95 °C and 40 cycles of 8 s at 95 °C and 34 s at 60 °C.
The study was supported by Adnan Menderes University Faculty of Medicine ADUBAPTPF-14007 numbered project and approved by Non-interventional Clinical Research Ethics Committee (Protocol number: 2014/405). Written informed consent was obtained from all parents.
The Kolmogorov–Smirnov test was used to assess the normality of numerical variables. For the numerical variables that were normally distributed, comparison between two groups was made by independent sample t test and descriptive statistics were presented as mean ± standard deviation. For the numerical variables that were not normally distributed, comparison between two groups was made by Mann–Whitney U test and descriptive statistics were presented as median (25–75 percentiles). To analyze the categorical data, a chi-square test was used and descriptive statistics were presented as frequency (%). The Spearman’s rho correlation analysis was used to determine the correlation between the numerical variable. Forward stepwise logistic regression analysis was used for predicting risk factors for wheezing. The p values below 0.05 were considered statistically significant.
ResultsA total of 52 patients who attended with wheezing attack, at the ages of 12–60 months with a history of three or more wheezing attacks in the last year, and 54 healthy children were included in the study. Vitamin D level could not be studied due to insufficient blood sample taken of a child in the patient group and therefore was excluded from the evaluations related to vitamin D.
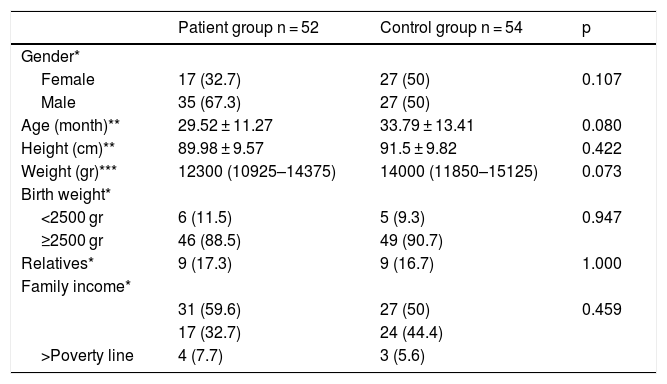
Table 1 shows the sociodemographic characteristics of patient and control groups. There was no significant difference in sociodemographic data of the patient and control groups (p > 0.05).
Sociodemographic characteristics of patient and control groups.
| Patient group n = 52 | Control group n = 54 | p | |
|---|---|---|---|
| Gender* | |||
| Female | 17 (32.7) | 27 (50) | 0.107 |
| Male | 35 (67.3) | 27 (50) | |
| Age (month)** | 29.52 ± 11.27 | 33.79 ± 13.41 | 0.080 |
| Height (cm)** | 89.98 ± 9.57 | 91.5 ± 9.82 | 0.422 |
| Weight (gr)*** | 12300 (10925–14375) | 14000 (11850–15125) | 0.073 |
| Birth weight* | |||
| <2500 gr | 6 (11.5) | 5 (9.3) | 0.947 |
| ≥2500 gr | 46 (88.5) | 49 (90.7) | |
| Relatives* | 9 (17.3) | 9 (16.7) | 1.000 |
| Family income* | |||
| 31 (59.6) | 27 (50) | 0.459 | |
| 17 (32.7) | 24 (44.4) | ||
| >Poverty line | 4 (7.7) | 3 (5.6) |
Descriptive statistics are given as; *: n (%), **: Mean ± SD, ***: Median (25th-75th percentiles).
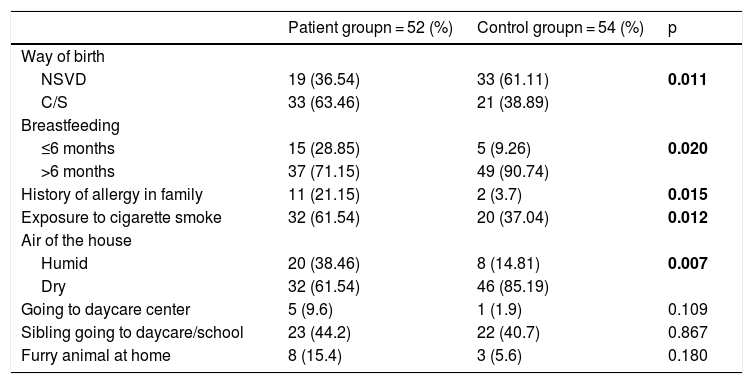
Table 2 shows the comparison of patient and control group in terms of wheezing risk factors. The cesarean delivery rate was significantly higher in the patient group than in the control group (p = 0.011). The rate of breastfeeding for six months or less was found to be significantly higher in the patient group than in the control group (p = 0.020). The history of allergic disease in first-degree relatives was significantly higher in the patient group compared to the controls (p = 0.015). Exposure to cigarette smoke was significantly higher in the patient group than the control group (p = 0.012). The humidity of the air of the house in the patient group was significantly higher than the controls (p = 0.007).
Comparison of patient and control group for wheezing risk factors.
| Patient groupn = 52 (%) | Control groupn = 54 (%) | p | |
|---|---|---|---|
| Way of birth | |||
| NSVD | 19 (36.54) | 33 (61.11) | 0.011 |
| C/S | 33 (63.46) | 21 (38.89) | |
| Breastfeeding | |||
| ≤6 months | 15 (28.85) | 5 (9.26) | 0.020 |
| >6 months | 37 (71.15) | 49 (90.74) | |
| History of allergy in family | 11 (21.15) | 2 (3.7) | 0.015 |
| Exposure to cigarette smoke | 32 (61.54) | 20 (37.04) | 0.012 |
| Air of the house | |||
| Humid | 20 (38.46) | 8 (14.81) | 0.007 |
| Dry | 32 (61.54) | 46 (85.19) | |
| Going to daycare center | 5 (9.6) | 1 (1.9) | 0.109 |
| Sibling going to daycare/school | 23 (44.2) | 22 (40.7) | 0.867 |
| Furry animal at home | 8 (15.4) | 3 (5.6) | 0.180 |
NSVD = normal spontaneous vaginal delivery; C / S = cesarean.
Bold values mean statistical significance.
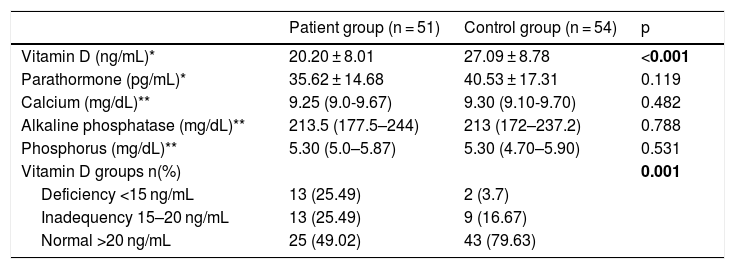
Comparison of vitamin D, Ca, P, ALP and PTH levels of the patient and control groups are given in Table 3. Vitamin D levels were significantly lower in the patient group compared to the control group (p < 0.001). There was no significant difference between the patient and control groups in terms of Ca, P, ALP and PTH levels (p > 0.05).
Biochemical data of the patient and the control group.
| Patient group (n = 51) | Control group (n = 54) | p | |
|---|---|---|---|
| Vitamin D (ng/mL)* | 20.20 ± 8.01 | 27.09 ± 8.78 | <0.001 |
| Parathormone (pg/mL)* | 35.62 ± 14.68 | 40.53 ± 17.31 | 0.119 |
| Calcium (mg/dL)** | 9.25 (9.0-9.67) | 9.30 (9.10-9.70) | 0.482 |
| Alkaline phosphatase (mg/dL)** | 213.5 (177.5–244) | 213 (172–237.2) | 0.788 |
| Phosphorus (mg/dL)** | 5.30 (5.0–5.87) | 5.30 (4.70–5.90) | 0.531 |
| Vitamin D groups n(%) | 0.001 | ||
| Deficiency <15 ng/mL | 13 (25.49) | 2 (3.7) | |
| Inadequency 15–20 ng/mL | 13 (25.49) | 9 (16.67) | |
| Normal >20 ng/mL | 25 (49.02) | 43 (79.63) |
*Descriptive statistics are given as; *: mean ± SD **: Median (25th–75th percentiles).
Bold values mean statistical significance.
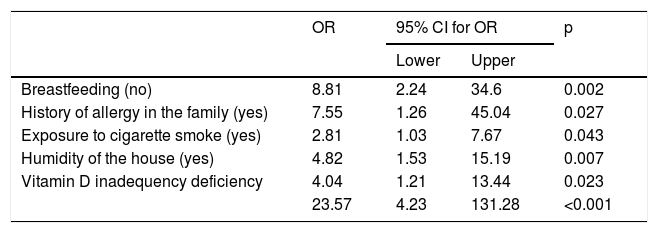
Results of forward stepwise logistic regression analysis for predicting risk of wheezing are given in Table 4.
Results of forward stepwise logistic regression analysis for predicting risk of wheezing.
| OR | 95% CI for OR | p | ||
|---|---|---|---|---|
| Lower | Upper | |||
| Breastfeeding (no) | 8.81 | 2.24 | 34.6 | 0.002 |
| History of allergy in the family (yes) | 7.55 | 1.26 | 45.04 | 0.027 |
| Exposure to cigarette smoke (yes) | 2.81 | 1.03 | 7.67 | 0.043 |
| Humidity of the house (yes) | 4.82 | 1.53 | 15.19 | 0.007 |
| Vitamin D inadequency deficiency | 4.04 | 1.21 | 13.44 | 0.023 |
| 23.57 | 4.23 | 131.28 | <0.001 | |
When the patient group was evaluated by means of attack severity, two patients (3.8%) had severe attacks and 50 patients (96.2%) had mild attacks. Due to the low number of patients with a severe attack, no statistical data could be studied between the attack severity and vitamin D. Table 5 shows the comparison of the levels of vitamin D with the factors that may indicate the severity of the wheezing attack. There was no significant difference in vitamin D levels between hospitalized and not hospitalized patients and who received and did not receive oxygen and steroid treatment during the wheezing attack (p > 0.05). There was no statistically significant correlation between oxygen saturation and vitamin D levels in wheezing attack (r = 0,062; p = 0.665).
Comparison of vitamin D levels according to the factors that may indicate the severity of wheezing attack.
| Level of Vitamin D (ng/ml) | P | ||
|---|---|---|---|
| Yes | No | ||
| Hospitalization during attack | 21.76 ± 8.20(n = 13) | 19.67 ± 7.99(n = 38) | 0.420 |
| Receiving steroid treatment during attack | 21.86 ± 7.96(n = 13) | 19.63 ± 8.06(n = 38) | 0.393 |
| Receiving oxygen treatment during attack | 20.25 ± 8.80(n = 26) | 20.15 ± 7.30(n = 25) | 0.963 |
Descriptive statistics are given as mean ± SD.
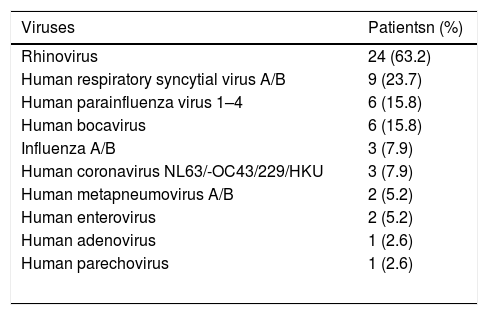
Thirty-eight (73%) of the children were infected by viruses. Rhinovirus was detected in 24 (63.2%) children, HRSV A/B was found in nine (23.7%), and human bocavirus was found in six (15.8%). One virus was detected in 24 (63.2%) of the children infected, 14 (36.8%) of them were coinfected. Four patients were coinfected with three and 10 with two viruses. Of the viral coinfections, rhinovirus and then HRSV A/B were the most frequently detected ones. The distribution of viruses detected is given in Table 6.
The distribution of respiratory viruses in patients.
| Viruses | Patientsn (%) |
|---|---|
| Rhinovirus | 24 (63.2) |
| Human respiratory syncytial virus A/B | 9 (23.7) |
| Human parainfluenza virus 1–4 | 6 (15.8) |
| Human bocavirus | 6 (15.8) |
| Influenza A/B | 3 (7.9) |
| Human coronavirus NL63/-OC43/229/HKU | 3 (7.9) |
| Human metapneumovirus A/B | 2 (5.2) |
| Human enterovirus | 2 (5.2) |
| Human adenovirus | 1 (2.6) |
| Human parechovirus | 1 (2.6) |
When the relationship between the presence of virus and vitamin D level was observed, 10 of 13 patients with vitamin D deficiency, eight of 13 patients with inadequacy and 19 of 25 patients with normal vitamin D levels were infected with at least one virus. No significant difference was found between vitamin D levels and the presence of a virus (p > 0.05).
The average vitamin D level in patients infected with one virus was 19.65 ± 8.18 ng/ml and in patients with multiple viruses was 20.22 ± 7.89 ng/mL. No significant difference was found in vitamin D levels according to the numbers of the viruses infected (p = 0.84).
DiscussionThe incidence of asthma and allergic diseases is increasing. In developed countries, the prevalence of wheezing was found to be 15–32% in children under five years of age.17 The factors responsible for this increase are; viral infections, air pollution, family history of atopy, low duration of breastfeeding, duration of pregnancy, passive smoking exposure, low socioeconomic status and exposure to allergens.18
Kolokotroni et al.19 showed that cesarean delivery was a risk factor for wheezing and asthma in a study of 2216 children. In a meta-analysis on this subject, cesarean delivery was found to increase the risk of asthma.20 In our study, the cesarean delivery rate was significantly higher in children with recurrent wheezing.
The results of studies investigating the relationship between breastfeeding and, recurrent wheezing and asthma are controversial. In a study by Taveras et al.,21 it was found that breastfeeding for more than nine months did not reduce recurrent wheezing attacks. However, in many studies it was reported that recurrent wheezing attacks decreased with the increase in breastfeeding time.22 In our study, breastfeeding for fewer than six months was significantly higher in the recurrent wheezing group.
Family history of allergic disease is an important factor for recurrent wheezing. In many studies in the literature, the presence of a history of atopy and asthma in first-degree relatives has been shown to be an important risk factor for recurrent wheezing and development of asthma in early childhood.23 In our study, the history of allergic disease in first-degree relatives was significantly higher in the recurrent wheezing group than the controls.
Exposure to smoking is an important risk factor for wheezing. In one study, 1456 children were followed-up for 10 years and cigarette smoke exposure between 0–4 years of age was found to be a risk factor for recurrent wheezing attacks.24 A meta-analysis showed that exposure of the mother to smoking during the prenatal period and smoking in the postnatal period increased the risk of developing wheeze and asthma in the child.25 In our study it was found that the children with recurrent wheezing are more likely to be exposed to smoking in the postpartum period.
A humid home environment may be a risk factor for recurrent wheezing due to the increase of home mites, fungal spores, and viruses in humid air causing recurrent wheezing. It was found in a study of 1105 infants that the humid environment at home was an important risk factor for recurrent wheezing attacks.26 Garcia-Marcos et al.22 showed in their study of 28,687 infants that living in a humid home is a risk factor for recurrent wheezing. In our study, children with recurrent wheezing attacks were found to be in more humid home environments.
Vitamin D is recognised as an important modulator of both the innate and adaptive immune system, and deficiency is associated with increased susceptibility to infections,13 and development of asthma.27 In one study, vitamin D levels were found to be lower in wheezy children triggered by viral infection compared to the control group.14 Demirel et al.11 also detected lower vitamin D levels in children with recurrent wheezing than the controls. Similarly, in many studies it has been reported that low levels of vitamin D may be a risk factor for recurrent wheezing.10,28 We found significantly lower vitamin D levels in children with recurrent wheezing compared to the control group. Vitamin D deficiency increased the risk of recurrent wheezing by 23.57 times.
In a study by Turkeli et al.,29 102 patients with asthma and 102 healthy control group aged between one and four years were compared by means of asthma control, asthma severity and vitamin D levels. It was determined that asthma severity was higher and asthma control was more difficult in patients with asthma with low vitamin D levels. It is stated that while there is strong evidence that high vitamin D levels reduce the risk of asthma exacerbation, there is weaker evidence of asthma incidence, prevalence and severity of asthma.30 In another study, no relation was found between the severity of asthma and vitamin D levels.31 In our study, due to the low number of patients with a severe attack, no statistical data could be studied between the severity of attack and vitamin D. There was no significant relationship between vitamin D levels and hospitalization, oxygen or steroid therapy. However, due to the small number of patients, more comprehensive studies are needed to reach a consensus on this issue.
There is definitive evidence that HRSV and rhinovirus can damage the airways to promote airway obstruction and recurrent wheezing.4 In the study of Jarti et al.14 with 284 children hospitalized with wheezing attack, rhinovirus and then HRSV were found to be the most common agents. More than one virus was found in 112 (39%) patients and of the viral coinfections, 51% involved rhinovirus and 35% involved HRSV. In our study, viral infection was detected in 38 (73%) of 52 children applying with recurrent wheezing attack. The most common viral agents were rhinovirus in 24 (63.2%) and HRSV in nine (23.7%). Coinfection was found in 14 (36.8%) of them. The data we obtained were consistent with the literature. Jarti et al.14 found that the child’s serum 25(OH)D3 level was inversely associated with HRSV, rhinovirus and multiple viral cause. They concluded that vitamin D might play a role in antiviral defense and might be important in infants exposed to RSV and in high asthma risk children exposed to rhinovirus. We found no significant difference between vitamin D levels and the presence of a virus in our study.
The low number of subjects in the study groups, small sample size of the patients with severe attacks and the lack of data about vitamin D supplementations of the children were the limitations of the study.
As a result, it was found that smoking exposure, being breastfed for ≤6 months, humid nature of the home, history of cesarean delivery and allergic disease in family, and vitamin D deficiency and inadequacy may be risk factors for recurrent wheezing. Rhinovirus and HRSV were the most common viruses detected. Due to the low number of patients, we could not evaluate the relationship between the severity of attacks and vitamin D levels. Also, there was no significant relationship between vitamin D levels and the presence of viruses. Because of the frequent detection of vitamin D deficiency and inadequacy in children with recurrent wheezing, we think there is a need for prospective randomized controlled studies for vitamin D supplementation in children with low vitamin D levels and recurrent wheezing.
Conflict of interestThe authors have no conflict of interest to declare.