Chronic spontaneous urticaria (CSU) is a disease that seriously disturbs the quality of life of the patient. Currently there is no prognostic marker of the disease. Our aim in this study was to determine possible prognostic markers of CSU in children.
MethodAll patients younger than 18 years of age who were followed by the Pediatric Immunology and Allergy Clinic of Diyarbakir Children’s Hospital with a diagnosis of CSU between June 2017 and February 2019 were included in the study. Clinical and laboratory findings were compared between the symptomatic patient group and the patient group that remained in remission for at least three months without use of medication.
ResultsOf the 52 cases included in the study, 32 (61.5%) were male. Mean age at time of diagnosis was 9.4 ± 4.4 years. Twenty-four cases (46.2%) went into remission. Young age at the time of diagnosis and being in the initial months of the disease were found to be associated with good prognosis (p < 0.05). Among laboratory results, elevation in absolute neutrophil count and neutrophil/lymphocyte ratio (NLR) were found to be associated with poor prognosis (p < 0.05). NLR was positively correlated with disease duration (p < 0.05).
ConclusionFor childhood CSU, younger age and initial months of the disease are good prognostic indicators, while only neutrophil/lymphocyte count can be used as a remission marker.
Chronic spontaneous urticaria (CSU) is defined as urticaria lasting for more than six weeks with either known or unknown etiology, characterized by almost daily recurring itching, wheals, and/or angioedema.1 Studies in the pediatric age group show that 16.5–37% of children with CSU go into remission within the first year and 36–54% do so within the first three years.2 The disease seriously disturbs the quality of life of patients.3 Currently, there is no marker available that can predict disease prognosis. The limited studies available on this subject are mostly confined to the adult patient population.4 Few studies including adult subjects have shown that the presence of angioedema, positive autologous serum skin test result, and the presence of thyroid antibodies were indicators that chronic urticaria (CU) was unlikely to enter remission in the near future.5 Additionally, remission was shown to be more frequent among children with CSU who were under control with treatment and had low urticaria activity score.6
Our aim with the present study was to investigate the relationship between the natural course of the disease and clinical and laboratory findings in children with CSU, and thus to determine the possible prognostic markers of the disease in children.
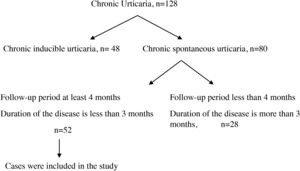
Materials and methodsFifty-two patients younger than 18 years of age who were followed by the Pediatric Immunology and Allergy Clinic of Diyarbakir Children’s Hospital with a diagnosis of CSU between June 2017 and February 2019 were included in the study. Inclusion criteria were as follows: (I) disease duration of fewer than three months when included in the study; and (II) follow-up period of at least four months. This study was conducted in accordance with the guidelines of the Ethics Committee of the University of Health Sciences, Diyarbakir Gazi Yasargil Training and Research Hospital (decision dated December 14, 2018, protocol number 2018/187, decision approval 2018/14-12).
Detailed anamnesis and physical examination were performed in all patients. All patients were investigated in terms of history of atopic disease, medication use, signs of infection, signs of soft tissue and autoinflammatory diseases, and induced urticaria. If respiratory or food allergy was suspected, respiratory and skin tests were performed. Provocation tests were applied to patients who had findings suggesting physical urticaria. In order to investigate the known causes of CSU, (I) complete blood count, hepatic and renal function tests, erythrocyte sedimentation rate, (II) infection parameters (hepatitis B and C serology, urine culture, Helicobacter pylori IgG in stool and stool microscopy for parasites), (III) autoimmunity panel [antinuclear antibody (ANA), anti-thyroglobulin antibody (anti-TG), anti-thyroperoxidase (anti-TPO) antibody, free T4 and thyroid stimulating hormone (TSH)] were investigated.
For the treatment of CSU, second-generation antihistamines were administered as the first level of therapy. Two weeks after treatment, antihistamine doses were increased in patients who could not control the disease with a standard dose of antihistamine. In the treatment of patients whose symptoms persisted despite high antihistamine doses for four weeks, either antihistaminic agent was modified, or antileukotrienes were added. In patients whose symptoms were not controlled after 1–4 weeks with this treatment, H2 antihistamines or omalizumab was administered.7,8 Patients were invited for follow-up visits once every four weeks. Those who did not have urticarial symptoms for at least three months without use of medication were accepted as being in remission. The patient group in remission and the symptomatic patient group were compared in terms of clinical and laboratory properties.
Statistical analysesIBM SPSS version 22.0 (Armonk, NY, USA) was used for all statistical analyses. The Kolmogorov–Smirnov test was used to test the normality of variables. Parametric methods were used for analysis of variables with a normal distribution, whereas non-parametric methods were used for analysis of variables that were not normally distributed. The Pearson’s chi-square and linear-by-linear association tests were used with an exact test for the comparison of categorical data. The categorical data are expressed as a percentage of the number (n) of children evaluated. The level of significance for the analyses was p < 0.05. Correlation of variables was analyzed with Spearman’s rho test.
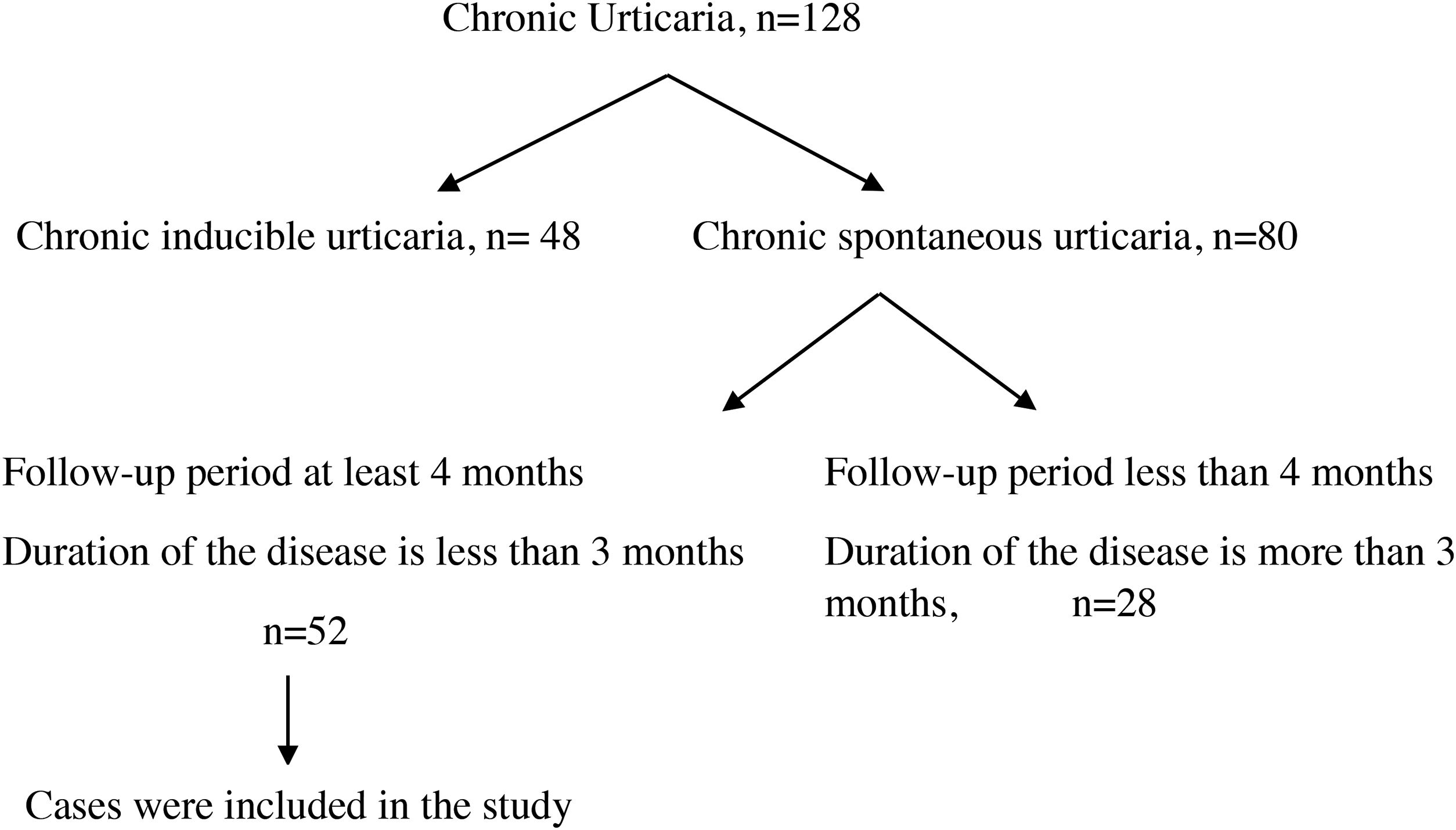
ResultsOne hundred and twenty-eight cases with CU were followed. The diagnosis of chronic inducible urticaria in 48 cases and CSU in 80 cases was confirmed. A total of 52 cases were included in the study (Fig. 1), and 32 of them (61.5%) were male. Mean age at the time of diagnosis was 9.4 ± 4.4 years, and disease duration was 5.0 ± 3.1 months. Six patients (11.5%) had additional atopic disease (two patients had allergic rhinitis, two had asthma, and two had both). Twelve patients (23%) were detected to have autoimmunity. Among these patients, two patients had hashimoto thyroiditis, and one patient had type 1 diabetes mellitus disease. In the other nine patients only autoantibodies were detected. Helicobacter pylori IgG antibody was detected in six (11.5%) patients. Two of these patients developed remission with treatment. Mean erythrocyte sedimentation rate (ESR) was 15.4 ± 10.3 mm/h; mean serum total IgE antibody level was 76.8 ± 107.0 IU/L; mean absolute neutrophil count was 4.7 ± 2.0 × 103/µl; mean absolute lymphocyte count was 3.2 ± 1.5 × 103/µl; mean neutrophil/lymphocyte ratio (NLR) was 1.8 ± 1.6; mean absolute eosinophil count was 0.2 ± 0.2 × 103/µl; mean absolute basophil count was 0.03 ± 0.02 × 103/µl; and mean platelet count was 315.2 ± 90.8 × 103/µl (Table 1). Thirty-one cases with CSU were controlled by a standard dose of second-generation antihistamines, and 14 cases were controlled by increased dosage of antihistamines. Five cases with CSU were controlled by modification of second-generation antihistamines, and two cases were controlled by the addition of antileukotrienes to antihistamines.
General characteristics of 52 patients.
| Gender (male), n (%) | 32 (61.5) |
| Age, yearsa | 9.4 ± 4.4 |
| Disease duration, montha | 5.0 ± 3.1 |
| Atopic disease, n (%) | 6 (11.5) |
| Remission, n (%) | 24 (46.2) |
| Autoimmunity, n (%) | 12 (23) |
| Helicobacter pylori IgG, n (%) | 6 (11.5) |
| Sedimentation (mm/h)a | 15.4 ± 10.3 |
| Serum total IgE level (IU/L)a | 76.8 ± 107.0 |
| Absolute neutrophil count (×103/µl)a | 4.7 ± 2.0 |
| Absolute lymphocyte count (×103/µl)a | 3.2 ± 1.5 |
| Neutrophil-lymphocyte ratioa | 1.8 ± 1.6 |
| Absolute eosinophil count (×103/µl)a | 0.2 ± 0.2 |
| Absolute basophil count (×103/µl)a | 0.03 ± 0.02 |
| Platelet count (×103/µl)a | 315.2 ± 90.8 |
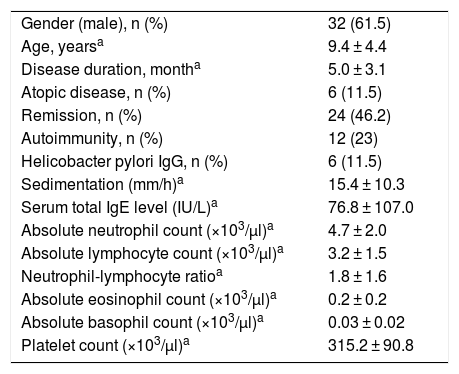
Twenty-four (46.2%) patients went into remission. Upon comparing the remission group and the symptomatic group in terms of demographic properties including sex, age at diagnosis, duration of urticaria and additional atopic disease, it was found that younger age at diagnosis and short duration of urticaria were good prognostic indicators. With regard to laboratory findings including autoimmunity, ESR, Helicobacter pylori IgG antibody, serum total IgE level, absolute neutrophil count, absolute lymphocyte count, NLR, absolute eosinophil count, absolute basophil count and platelet count, it was found that those with higher neutrophil count and NLR had lower remission rate (p < 0.05) (Table 2). While there was no correlation between absolute neutrophil count and disease duration (p > 0.05), a positive correlation was found between NLR and disease duration (p < 0.05).
Comparison of remission group and symptomatic group in patients with chronic spontaneous urticaria.
| Remission group, n = 24 | Symptomatic group, n = 28 | p | |
|---|---|---|---|
| Gender (male), n (%) | 17 (62.5) | 15 (60.7) | 0.202 |
| Age, yearsa | 8.0 ± 4.2 | 10.5 ± 4.4 | 0.046 |
| Disease duration, monthsa | 3.5 ± 2.1 | 6.4 ± 3.3 | 0.001 |
| Atopic disease, n (%) | 2 (8.3) | 4 (14.2) | 0.473 |
| Autoimmunity, n (%) | 5 (20.8) | 7(19.6) | 0.785 |
| Helicobacter pylori IgG, n (%) | 1 (4.2) | 5 (17.6) | 0.475 |
| Sedimentation (mm/h)a | 15.9 ± 11.5 | 15.1 ± 9.8 | 0.806 |
| Serum total IgE level (IU/L)a | 72.5 ± 92.7 | 80.2 ± 119.0 | 0.812 |
| Absolute neutrophil count (×103/µl)a | 4.3 ± 2.0 | 5.3 ± 2.0 | 0.028 |
| Absolute lymphocyte count (×103/µl)a | 3.3 ± 1.3 | 3.2 ± 1.7 | 0.749 |
| Neutrophil-lymphocyte ratioa | 1.3 ± 0.7 | 2.26 ± 2.0 | 0.036 |
| Absolute eosinophil count (×103/µl)a | 0.2 ± 0.2 | 0.2 ± 0.2 | 0.939 |
| Absolute basophil count (×103/µl)a | 0.03 ± 0.01 | 0.03 ± 0.02 | 0.958 |
| Platelet count (×103/µl)a | 329.7 ± 115.8 | 303.3 ± 63.4 | 0.307 |
p-value of less than 0.05 are bolded.
In this study, we found that younger age at diagnosis and being in the initial months of the disease were associated with good prognosis, while elevated absolute neutrophil count and neutrophil/lymphocyte ratio were associated with poor prognosis in children with CSU. A positive correlation was found between neutrophil/lymphocyte ratio and disease duration.
The majority of patients with CSU have been reported to be aged between 20–59 years old.9 In a study of 117 patients with CSU from the adult and pediatric age populations, Hiragun et al.4 reported that those younger than 19 years old had greater remission rates compared to older patients. Similarly, we found more frequent remission among younger patients in the present study.
Most adult patients with CSU are reported to be women; however there are very few studies that investigated whether there is any difference between the sexes with regard to remission.10 In their study of adult CSU patients, Gregoriou et al.11 reported that female patients went into remission later than male patients. Similarly, in their study, Harris et al.12 retrospectively evaluated 52 children with CU and found a remission rate of 37% among females at the end of one year, and reported that this rate was greater compared to males. On the contrary, Chansakulporn et al.13 did not find a difference between sexes regarding remission rates in their study including children with CSU. Sahiner et al.14 studied the pediatric age group of patients with CU, and reported that prognosis with regard to remission was not good in females older than 10 years old. Consistent with the literature, we found no difference between sexes regarding remission rates.
Epidemiological studies showed that while approximately 50% of adult patients with CSU enter remission within the first three months, 80% do so within the first year, and 11% remain symptomatic even after the 5th year.15 In their study, Eser et al.16 retrospectively evaluated 52 pediatric patients with CSU and found that disease duration at time of initial presentation was longer among late-remitting cases. On the other hand, Chansakulporn et al.2 reported that disease duration was not a remission marker in their study including adult CSU patients. In line with the literature, we found that patients mostly went into remission during the initial month of the disease.
Contradicting results have been found in studies investigating the relationship between autoimmunity and remission in CSU. In their study including an adult group of patients with CU, Kulthanan et al.17 reported that there was no association of remission with either positive or negative autologous serum test result. In contrast, Staubach et al.18 reported that remission occurred later among adult cases with positive autologous serum skin test result. In their study of adult patients, Chanprapaph et al.19 reported that thyroid peroxidase antibody was a remission marker in CSU patients with autoimmune thyroiditis. Consistent with the literature, we found that there was no relationship between autoimmunity and remission.
Many studies have reported that CSU is a systemic chronic inflammatory disease.20 NLR has been used as a marker of systemic inflammation in various diseases because the physiological response of circulating leukocytes in the human body to stress is an elevation in neutrophil count and a reduction in lymphocyte count.21 However, the number of patients evaluated is limited and further studies are required to confirm the role of NLR as a remission marker. Ye et al.22 reported a correlation of uncontrollable CU with C3, TNF-α, and eosinophilic cationic protein levels. Aleem et al.23 reported that CRP level was correlated with CSU activity and could be used as a remission marker. Consistent with the literature, we found both neutrophil count and NLR among inflammation markers were elevated in the symptomatic patient group. Additionally, there was a positive correlation between NLR and disease duration.
Various basophil-associated disorders have been reported in patients with CSU. Grattan et al.24 reported a relationship between circulating basophil levels and CU disease activity score, with reduced basophil count in patients with CU. Ye et al.25 stated that reduction in basophil count could be a marker of severe CU in such patients. In their study including adult CSU patients, Berti et al.26 reported that positive histamine release test result was an indicator of whether remission will occur before 12 months. In contrast to the literature, we did not find a relationship between basophil count and remission in the present study.
Elevated serum total IgE levels have been shown in 93% of patients with CU. In their study including adult patients, Kessel et al.27 showed that serum total IgE level was frequently elevated in cases with CU and was associated with both disease severity and duration. Unlike the literature findings, we did not find a relationship between serum total IgE level and remission.
In conclusion, our results show that remission was achieved sooner when onset of CSU was at a younger age in children. We found reduced remission rates as the disease duration increased. Among inflammation markers, neutrophil count and neutrophil/lymphocyte ratio are elevated in CSU patients with poor prognosis. Neutrophil/lymphocyte ratio can be used as a remission marker in CSU.
Ethical approvalApproval by the local ethics committee was granted.
Funding sourceNo external funding was secured for this study.
Financial disclosureThe authors have no financial relationships relevant to this article to disclose.
Conflict of interestThe authors have no conflicts of interest to disclose.