In Lebanon, asthma is underdiagnosed due to low access to healthcare, particularly in rural areas, although asthma diagnosis in children is based mainly on clinical symptoms. Thus, wheezing might be more suggestive of undiagnosed respiratory diseases including asthma in Lebanese children. This study aimed to determine the factors associated with wheezing in Lebanese children without asthma diagnosis.
MethodsThis cross-sectional study was conducted between December 2015 and April 2016, enrolling a total of 1203 schoolchildren.
ResultsOut of 1500 prepared questionnaires, 1380 questionnaires were distributed in schools, and 1203 (87.17%) were collected back from the parents of children aged between 4–17 years old. The sample included 42 (3.5%) [95% CI 0.025–0.045] children with reported chronic wheezing. A multivariable analysis was performed taking the presence versus absence of wheezing in children as the dependent variable. The results showed that spraying pesticides at home (aOR=1.91), presence of humidity at home (aOR=2.21) and child reflux (aOR=2.60) were significantly associated with the presence of wheezing in children.
ConclusionThe findings of the study suggest that certain environmental factors, such as pesticides, humidity at home and reflux disease, might be associated with wheezing episodes in children. Those factors can be prevented through raising awareness by health care professionals.
Wheezing is a symptom of hyperactive airway diseases that arises initially in early childhood, mainly in the first few years of life, as this age group is highly prone to infections leading to long-term pulmonary complications.1 It usually manifests as continuous whistling during breathing, resulting from airway narrowing or obstruction of the respiratory tract.2 The majority of episodes are usually mild, easily managed, and self-treated.3 However, different wheezing phenotypes have been described at early age (incident, persistent, intermittent),4 which can lead to complications,5 particularly at a young age, when it can be significantly associated with high morbidity and mortality rates.6 Children who experience wheezing episodes also complain from diminished airway function, which substantially impacts their quality of life and aggravates the financial burden of the family.7 Since it is a sensitive non-specific manifestation, wheezing represents a high expenditure on medical costs, including consultations, emergency department visits, utilization of healthcare resources, access to medical devices, consumption of prescription medications, and high rates of hospitalization.8,9
Wheezing is also one of the main manifestations of asthma; it affects around 30 to 50% of children and is thought to progress to asthma in almost half of the cases.10 During the first months of life, the impaired development of the immune system would increase the risk of wheezing: around 45% of infants have at least one episode of wheezing, and 23% have recurrent wheezing.11 However, the reported prevalence varies by country, depending on risk and protective factors, genetics, and residence areas.11,12 A substantial number of studies have been conducted to investigate the factors associated with wheezing episodes during the first years of life in Latin America (Brazil13), Europe (Spain14) and Arizona.1
Multiple diseases and factors are associated with the pathogenesis of wheezing, including recurrent acute viral respiratory infections in preschool-age children,3 gastroesophageal reflux disease (GERD),15 colds at young age,16 family history of asthma and rhinitis,11 male gender,17 and low maternal educational level.13
Wheezing is not limited only to preschool children; it also occurs in older children, which clearly depicts that other factors such as genetic history and immunological model, along with environmental factors such as maternal smoking, play a critical role in the occurrence of wheezing. The triggers for the progression and exacerbation of wheezing in childhood are the smaller airways in children,1 and exposure to allergens.18 Other risk factors for wheezing include pets at home,19 utilization of a stove as a heating method,20 and humidity on the bedroom walls.21,22 Some additional risk factors are specific to recurrent wheezing, such as pre-existing cardiopulmonary disorders, defective immune system, premature birth, and early infancy.20
The real extent of wheezing is challenging since its actual impact in infants has not been extensively assessed previously, and it has been demonstrated that it affects the quality of life and aggravates the financial burden on the family.23 In Lebanon, hyperactive airway diseases are underdiagnosed due to low access to healthcare, particularly in rural areas, and the diagnosis in children is usually based on clinical symptoms and after ruling out other diseases.24,25 Thus, wheezing might be more suggestive of undiagnosed respiratory diseases in Lebanese children. Therefore, this study aimed to determine the factors associated with wheezing in Lebanese children.
Patients and methodsStudy designThis cross-sectional study was conducted between December 2015 and April 2016 and enrolled a total of 1203 schoolchildren aged four to 17. School principals (two in Beirut, two in Mount Lebanon, one in North Lebanon, one in South Lebanon, and one in Bekaa) were contacted to obtain authorization to enter the classrooms to distribute the questionnaires. Of the seven schools approached, six (85.71%) agreed to distribute the questionnaires to their students, while one school (in Bekaa) refused to participate. The questionnaire was distributed to all children aged four to 17. Parents who agreed to participate completed the questionnaire and returned it along with signed written consent. From the parents’ responses, children were grouped into healthy children (no diagnosis of wheezing and no respiratory symptoms reported by the parents, such as wheezing, cough, and dyspnea), and the case group (wheezing).26 Children with a diagnosed chronic respiratory condition, such as asthma and cystic fibrosis were excluded, as were those whose parents refused to fill out the questionnaire. The methods used in this study have been described in previous research.10,26–33
Ethical aspectsThe Institutional Review Board of Faculty of Pharmacy of the Lebanese University waived the need for approval based on the fact that it was an observational study that respected the autonomy and confidentiality of the participants and caused them no harm. Additionally, data was anonymous and could not be traced back.
Questionnaire and variablesThe questionnaire used was self-administered and taken from two standardized questionnaires: the ATS-DLD-78C of the American Thoracic Society,34 and the International Study of Asthma and Allergies in Childhood (ISAAC).35 It was adapted to the local Arabic language, the native language of Lebanon, through a forward and back translation process to ensure the accuracy of questions. The same questionnaire was administered to all participants to evaluate the diagnosis of asthma and respiratory symptoms, particularly wheezing. One question assessed wheezing in children: “Has your child been diagnosed of having wheezing by a physician?”
The socio-demographic and socioeconomic characteristics included age (further subdivided into three categories: pre-school children (aged 3–5),36 school children (aged 6–12), and adolescents (aged 13–17)), gender, region, household crowding (number of rooms and the number of persons living in the house), education level of both parents (quantified by the number of years of schooling), and family history of asthma/wheezing.
The remaining evaluated factors associated with wheezing included: smoking or drinking alcohol or the use of any prescribed medication during pregnancy and breastfeeding, number of smokers at home, and the exposure to pesticides (occupational, regional, local, and domestic) and cleaning products. The exposure to pesticides was recorded using the following questions: “Have you ever used pesticides in your work?”, “Have you ever used pesticides out of your work (for house or garden treatment…)?”, “Do you live in a region heavily treated by pesticides?”, “Do you live in the proximity of a heavily treated field by pesticides?” The duration of exposure at work and the number of times the house or the garden was sprayed by pesticides per week or year were also recorded. Questions about smoking or alcohol intake during pregnancy and during breastfeeding, the kind of smoking or alcohol along with the quantity were included, in addition to the use of any prescribed medication during pregnancy or lactation, occupational, regional, local, and domestic pesticides exposures and cleaning products use. Active smoking was determined by several questions (i.e. number of daily cigarettes, weekly waterpipes smoked, other nicotine/tobacco use), categorizing subjects in non-smokers or current smokers. Passive smoking was characterized by the number of smokers at home.
Detergents use was determined by questions about who uses these products at home, the type of detergents and if there is any mixture of these products or not (the use of two or more detergents simultaneously). Information about the heating system used at home, the presence of an air conditioner and a dehumidifier, the presence of humidity or mold at home as seen on walls, the child's history of recurrent otitis, tonsillectomy, cardiac problems, premature birth, kindergarten, respiratory problems forbidding daily activities, reflux, breastfeeding were also recorded.
Sample size calculationThe Epi info software was used to calculate the minimal sample size required, considering a study power of 80%, a confidence interval of 95%, and a previous prevalence of ever wheezing of 21.4% in Lebanon.32 The minimal sample size calculated was 516 children; this figure was tripled to allow adequate bivariate and multivariable analyses, and also to take into account refusal rates.
Statistical analysisData analysis was performed using SPSS software, version 25. Percentages were reported for qualitative variables, and means and standard deviations for quantitative variables. The Chi-square test was used to compare group percentages and the Student's t-test for quantitative variables of normal distribution and homogeneous variances. A multivariable backward logistic regression was also applied, taking the presence versus absence of wheezing as the dependent variable; independent variables that showed a p-value <0.2 in the bivariate analysis were entered in the multivariable model. A p-value <0.05 was considered significant.
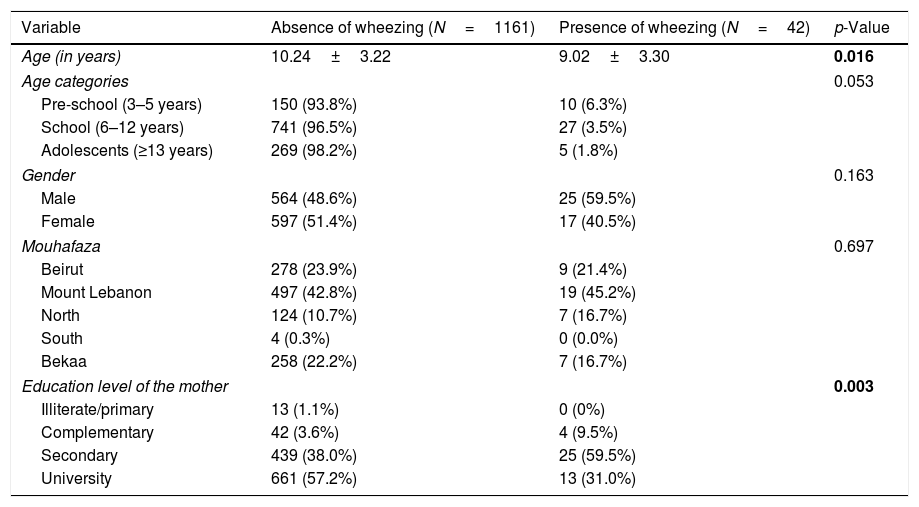
ResultsSocio-demographic and socioeconomic characteristicsOut of 1500 prepared questionnaires, 1380 were distributed in schools, and 1203 (87.17%) were collected back from parents of the children aged between 4–17 years old. No missing values were replaced in the database. The sample included 42 (3.5%) [95% CI 0.025–0.045] children with reported chronic wheezing. The socio-demographic and socioeconomic factors are summarized in Table 1. A significantly lower mean age (9.02 vs. 10.24) and a higher percentage of children whose mothers have a secondary level of education were found in wheezing compared to non-wheezing children. After applying the age categories, a higher percentage of pre-schoolers had wheezing compared to school children and adolescents, this association tending to be significant (p=0.053).
Bivariate analysis of sociodemographic and socioeconomic factors associated with wheezing in children.
| Variable | Absence of wheezing (N=1161) | Presence of wheezing (N=42) | p-Value |
|---|---|---|---|
| Age (in years) | 10.24±3.22 | 9.02±3.30 | 0.016 |
| Age categories | 0.053 | ||
| Pre-school (3–5 years) | 150 (93.8%) | 10 (6.3%) | |
| School (6–12 years) | 741 (96.5%) | 27 (3.5%) | |
| Adolescents (≥13 years) | 269 (98.2%) | 5 (1.8%) | |
| Gender | 0.163 | ||
| Male | 564 (48.6%) | 25 (59.5%) | |
| Female | 597 (51.4%) | 17 (40.5%) | |
| Mouhafaza | 0.697 | ||
| Beirut | 278 (23.9%) | 9 (21.4%) | |
| Mount Lebanon | 497 (42.8%) | 19 (45.2%) | |
| North | 124 (10.7%) | 7 (16.7%) | |
| South | 4 (0.3%) | 0 (0.0%) | |
| Bekaa | 258 (22.2%) | 7 (16.7%) | |
| Education level of the mother | 0.003 | ||
| Illiterate/primary | 13 (1.1%) | 0 (0%) | |
| Complementary | 42 (3.6%) | 4 (9.5%) | |
| Secondary | 439 (38.0%) | 25 (59.5%) | |
| University | 661 (57.2%) | 13 (31.0%) | |
The variables that are not present in the table did not show a significant association with wheezing in children.
Numbers in bold indicate significant p-values.
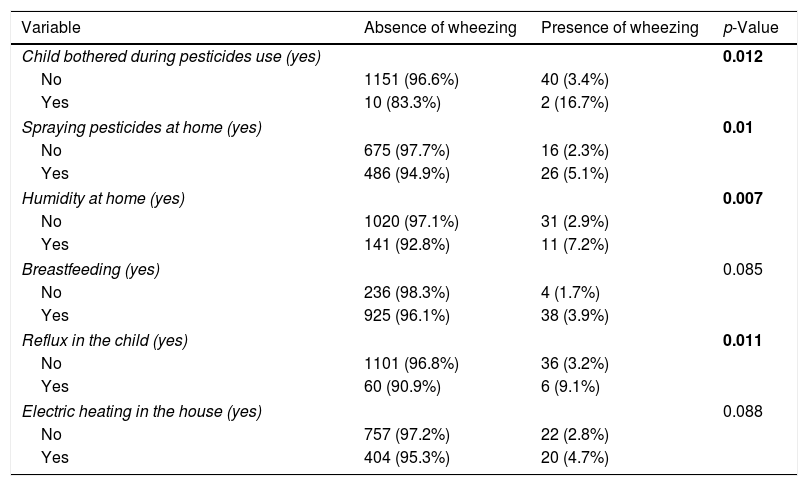
A significantly higher percentage of children had wheezing among those bothered when using pesticides (16.7% vs. 3.4%), whose parents spray pesticides at home (5.1% vs. 2.3%), who live in a house with humidity on the walls (7.2% vs. 2.9%), and those with GERD (9.1% vs. 3.2%), compared to children who did not have these factors (Table 2). All other variables did not show a significant association with wheezing.
Bivariate analysis of other factors associated with wheezing in children.
| Variable | Absence of wheezing | Presence of wheezing | p-Value |
|---|---|---|---|
| Child bothered during pesticides use (yes) | 0.012 | ||
| No | 1151 (96.6%) | 40 (3.4%) | |
| Yes | 10 (83.3%) | 2 (16.7%) | |
| Spraying pesticides at home (yes) | 0.01 | ||
| No | 675 (97.7%) | 16 (2.3%) | |
| Yes | 486 (94.9%) | 26 (5.1%) | |
| Humidity at home (yes) | 0.007 | ||
| No | 1020 (97.1%) | 31 (2.9%) | |
| Yes | 141 (92.8%) | 11 (7.2%) | |
| Breastfeeding (yes) | 0.085 | ||
| No | 236 (98.3%) | 4 (1.7%) | |
| Yes | 925 (96.1%) | 38 (3.9%) | |
| Reflux in the child (yes) | 0.011 | ||
| No | 1101 (96.8%) | 36 (3.2%) | |
| Yes | 60 (90.9%) | 6 (9.1%) | |
| Electric heating in the house (yes) | 0.088 | ||
| No | 757 (97.2%) | 22 (2.8%) | |
| Yes | 404 (95.3%) | 20 (4.7%) | |
The variables that are not present in the table did not show a significant association with wheezing in children.
Numbers in bold indicate significant p-values.
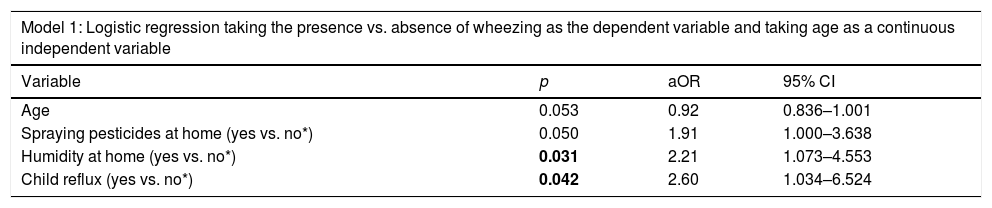
A first logistic regression model was performed taking the presence versus absence of wheezing in children as the dependent variable, and age as a continuous independent variable. Wheezing in children was significantly associated with spraying pesticides at home (aOR=1.91), humidity on the walls at home (aOR=2.21), and children with GERD (aOR=2.60) (Table 3, Model 1).
Multivariable analysis.
| Model 1: Logistic regression taking the presence vs. absence of wheezing as the dependent variable and taking age as a continuous independent variable | |||
|---|---|---|---|
| Variable | p | aOR | 95% CI |
| Age | 0.053 | 0.92 | 0.836–1.001 |
| Spraying pesticides at home (yes vs. no*) | 0.050 | 1.91 | 1.000–3.638 |
| Humidity at home (yes vs. no*) | 0.031 | 2.21 | 1.073–4.553 |
| Child reflux (yes vs. no*) | 0.042 | 2.60 | 1.034–6.524 |
| Model 2: Logistic regression taking the presence vs. absence of wheezing as the dependent variable and taking age as a categorical independent variable | |||
|---|---|---|---|
| Variable | p | aOR | 95% CI |
| Child bothered during pesticides use (yes vs. no*) | 0.048 | 4.93 | 1.01–23.97 |
| Spraying pesticides at home (yes vs. no*) | 0.028 | 2.06 | 1.08–3.91 |
| Humidity at home (yes vs. no*) | 0.052 | 2.05 | 0.99–4.25 |
| Child reflux (yes vs. no*) | 0.021 | 2.96 | 1.18–7.44 |
aOR: Adjusted odds ration; CI: 95% Confidence Interval; Numbers in bold indicate significant p-values.
When taking age as a categorical variable in the model, significantly higher odds of wheezing were associated with children bothered when using pesticides (aOR=4.93), spraying pesticides at home (aOR=2.06), and children with GERD (aOR=2.96) (Table 3, Model 2).
DiscussionThis cross-sectional study highlights the major findings on the contributing factors associated with wheezing diagnosis in a sample of Lebanese schoolchildren. Some factors related to higher odds of wheezing were highlighted, such as spraying pesticides at home, humidity on walls at home, and child with GERD.
The use of pesticides at home was significantly associated with wheezing in children, in agreement with previous findings.37,38 Spraying pesticides by a lay person whether at home, in the yard, or both, might increase the effects of pesticides on respiratory symptoms and might expose children to higher doses of pesticides, since a non-professional might not follow the instructions on the product's label.38
Humidity on the walls at home was significantly associated with wheezing in children, in agreement with multiple studies.39–41 Indeed, home dampness exacerbates asthma symptoms as it enables the growth and infestation of dust mites and molds that were shown to be highly associated with allergic respiratory symptoms in previous studies.38,41 Moreover, humidity and high temperature provide a suitable medium and environment for the development of molds and increase the infectivity of certain respiratory viruses, such as respiratory syncytial virus.42
Our results also showed that GERD was significantly associated with wheezing in children. These findings are consistent with those in the literature.4,43 The assumed mechanism is that esophageal acid stimulates vagally-mediated tracheobronchial responses and increases the bronchial hyper-responsiveness to other stimuli. In addition, GERD irritates the sensitive airways after the aspiration of tiny refluxed material from the tracheobronchial tree, thereby triggering airway defensive reflexes, such as cough, bronchospasm and mucus secretion, and contributing to wheezing.43
Advanced age in children was another factor that tended to be significantly associated with a lower risk of wheezing, consistent with previous findings showing that the incidence of wheezing declined with age.1,44 One explanation would be that as children grow, the lungs mature, and the immune system develops, thereby decreasing the risk of wheezing.45,46 Another explanation would be that respiratory syncytial virus, an important cause of early childhood wheezing, affects young age groups only.47
Clinical implicationsThis research highlights the fact that wheezing is triggered by different factors, whether pathological or environmental (indoor and outdoor). In view of these findings, preventive measures should be adequately implemented by the relevant stakeholders to curb and decrease the progression of hyperactive airway symptoms in children.
Limitations and strengthsThe study has some limitations. Although the sample size was decent, the number of wheezing children was small. Since it is a cross-sectional study, it might be subject to selection bias and recall bias by the parents, since the use of questionnaires may not always be accurate and limited by the difficulty in remembering the severity of symptoms. Many, but not all, of the variables that could affect the development of wheezing were taken into account. Additionally, not all wheezing causes were excluded, which might have caused a selection bias. Consequently, prospective studies taking into account these limitations are warranted, primarily to obtain unbiased information on the use of medications and exposure to triggers.
Despite those limitations, the study has strengths, such as the large number of participants recruited from different centers in different Lebanese geographic areas and the questionnaire which was similar to those used in other cross-sectional studies, thus allowing international comparisons.
ConclusionEnvironmental factors seem to be associated with wheezing in children, some of which (pesticide use, reflux, humidity) can be prevented by raising awareness among healthcare professionals. Further studies are needed to confirm our results.
Conflicts of interestThe authors have no conflicts of interest.