Cytokines, including interleukin-2 (IL-2) and interferon-gamma (IFN-γ), seem to play a role in the pathogenesis of juvenile idiopathic arthritis (JIA). The aim of this study was to investigate the associations of IL-2 and IFN-γ single nucleotide polymorphisms (SNPs) with susceptibility to JIA in an Iranian population.
MethodsGenomic DNA of 54 Iranian patients with JIA and 139 healthy unrelated controls were typed for IL-2 (G/T at −330 and +166) as well as IFN-γ gene (A/T at +874), using polymerase chain reaction with sequence-specific primers method, and compared between patients and controls.
ResultsA significantly higher frequency of the IL-2 −330 GG genotype (p<0.01) was found in the JIA patients compared to the controls. However, the GT genotype at the same position was notably lower than in controls (p<0.01). Moreover, IL-2 (−330, +166) GT haplotype was more frequent in patients with JIA in comparison with controls. No significant differences was observed between the two groups of case and control for IL-2 (G/T at +166) and IFN-γ (A/T at +874) SNPs.
ConclusionThe results of the current study suggest that certain SNPs of IL-2 gene have association with individuals’ susceptibility to JIA. However, further investigations are required to confirm the results of this study.
Juvenile idiopathic arthritis (JIA) is the most prevalent chronic arthritic condition of childhood with its prevalence varying worldwide among different ethnicities,14 which comprises seven distinct categories as demarcated by the International League of Associations for Rheumatology (ILAR) classification criteria.16 JIA encompasses all forms of arthritis of unknown aetiology that commence before the age of 16 years and persists for more than 6 weeks.16 Notwithstanding the heterogeneity between the JIA subgroups, they do appear to have genetic risk factors in common.7 The common feature of all JIA subtypes is the presence of chronic inflammation within synovial joints, in which cytokines play pivotal roles. It has been previously suggested that certain single nucleotide polymorphisms (SNPs) within the promoter and coding sequences of various cytokines’ genes, which affect their level of production, could be associated with the vulnerability to a group of autoimmune disorders.4,6,10,12,21
In the current study, we have selected two of the cytokines, including interferon-gamma (IFN-γ) and interleukin-2 (IL-2), that have been reported previously to have a role in JIA aetiopathogenesis,3,19 to determine whether certain SNPs in these cytokines’ genes are associated with susceptibility to JIA.
It has been postulated that these genes may also confer susceptibility to JIA. Therefore, the aim of this study was to assess whether IL-2 (G/T at −330 and +166) as well as IFN-γ (A/T at +874) gene polymorphisms are also associated with susceptibility to JIA in Iranian patients.
Patients and methodsStudy populationA total of 54 consecutive JIA patients, recruited from the Rheumatology Clinic of the Children's Medical Center Hospital, the Paediatrics Center of Excellence in Iran, were included in the case group and compared to 139 healthy unrelated individuals who were randomly selected from blood donors at Iranian blood transfusion organisations.1 Diagnosis of JIA was based on the ILAR classification criteria for JIA.17 Our patients’ group comprised 24 individuals with oligoarticular JIA, 19 with polyarticular JIA, and 11 with systemic disease subtype.
Written informed consents were obtained from all entrants to this investigation, and the study protocol was approved by the Ethical Committee of Tehran University of Medical Sciences.
Sampling and genotypingTo obtain genomic DNA, 5mL of peripheral blood was collected from each patient and stored at −20°C in EDTA tubes until DNA extraction using the “salting out” technique.13 Polymerase chain reaction, with sequence-specific primers method (PCR-SSP assay kit; Heidelberg University, Germany) was employed for cytokine gene typing according to the manufacturer's instructions.1 The three single nucleotide polymorphisms (SNPs) studied were IL-2 (G/T at −330 and +166) and IFN-γ (A/T at +874).
Statistical analysisThe allele, genotype and haplotype frequencies were evaluated by direct gene counting and compared with the healthy controls using the chi square test. The odds ratio (OR) and 95% confidence interval (CI) were estimated for each allele, genotype, and haplotype in the patient and control groups. Adherence to the Hardy–Weinberg equilibrium for each genotype was assessed for both case and control groups using the chi square test. P values of less than 0.05 were considered statistically significant.
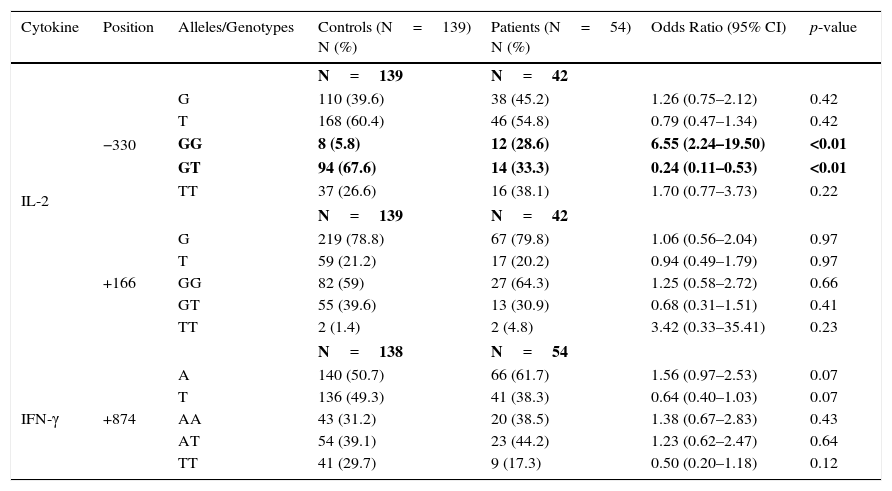
ResultsAllele, genotype and haplotype frequenciesThe allelic and genotype frequencies in patients with JIA and healthy controls are demonstrated in Table 1.
IL-2 and IFN-γ allele and genotype polymorphisms in patients with JIA and controls.
| Cytokine | Position | Alleles/Genotypes | Controls (N=139) N (%) | Patients (N=54) N (%) | Odds Ratio (95% CI) | p-value |
|---|---|---|---|---|---|---|
| IL-2 | N=139 | N=42 | ||||
| −330 | G | 110 (39.6) | 38 (45.2) | 1.26 (0.75–2.12) | 0.42 | |
| T | 168 (60.4) | 46 (54.8) | 0.79 (0.47–1.34) | 0.42 | ||
| GG | 8 (5.8) | 12 (28.6) | 6.55 (2.24–19.50) | <0.01 | ||
| GT | 94 (67.6) | 14 (33.3) | 0.24 (0.11–0.53) | <0.01 | ||
| TT | 37 (26.6) | 16 (38.1) | 1.70 (0.77–3.73) | 0.22 | ||
| N=139 | N=42 | |||||
| +166 | G | 219 (78.8) | 67 (79.8) | 1.06 (0.56–2.04) | 0.97 | |
| T | 59 (21.2) | 17 (20.2) | 0.94 (0.49–1.79) | 0.97 | ||
| GG | 82 (59) | 27 (64.3) | 1.25 (0.58–2.72) | 0.66 | ||
| GT | 55 (39.6) | 13 (30.9) | 0.68 (0.31–1.51) | 0.41 | ||
| TT | 2 (1.4) | 2 (4.8) | 3.42 (0.33–35.41) | 0.23 | ||
| N=138 | N=54 | |||||
| IFN-γ | +874 | A | 140 (50.7) | 66 (61.7) | 1.56 (0.97–2.53) | 0.07 |
| T | 136 (49.3) | 41 (38.3) | 0.64 (0.40–1.03) | 0.07 | ||
| AA | 43 (31.2) | 20 (38.5) | 1.38 (0.67–2.83) | 0.43 | ||
| AT | 54 (39.1) | 23 (44.2) | 1.23 (0.62–2.47) | 0.64 | ||
| TT | 41 (29.7) | 9 (17.3) | 0.50 (0.20–1.18) | 0.12 | ||
A positive genotypic association was detected for IL-2 −330/GG with JIA susceptibility (28.8% vs. 5.8%, p<0.01). Meanwhile, the GT genotype frequency at IL-2 −330 in JIA patient was notably lower than in healthy individuals (33.3% vs. 67.6%, p<0.01). No significant difference was discovered between the two groups of case and control for IL-2 (G/T at +166) and IFN-γ (A/T at +874) SNPs.
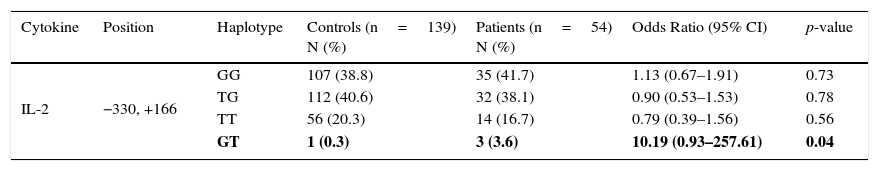
At the haplotypic level, IL-2 (−330, +166) GT haplotype was found to be more frequent in patients with JIA in comparison with healthy controls (3.6% vs. 0.3%, p=0.04). We observed no significant differences between the two groups for GG, TG and TT haplotypes at the same positions (Table 2).
IL-2 haplotype polymorphism in patients with JIA and controls.
| Cytokine | Position | Haplotype | Controls (n=139) N (%) | Patients (n=54) N (%) | Odds Ratio (95% CI) | p-value |
|---|---|---|---|---|---|---|
| IL-2 | −330, +166 | GG | 107 (38.8) | 35 (41.7) | 1.13 (0.67–1.91) | 0.73 |
| TG | 112 (40.6) | 32 (38.1) | 0.90 (0.53–1.53) | 0.78 | ||
| TT | 56 (20.3) | 14 (16.7) | 0.79 (0.39–1.56) | 0.56 | ||
| GT | 1 (0.3) | 3 (3.6) | 10.19 (0.93–257.61) | 0.04 |
In addition, no significant difference was detected between the aforementioned gene polymorphisms and individuals’ susceptibility to different JIA subtypes, including systemic, polyarticular, and oligoarticular JIA.
DiscussionThis study aims at evaluating the association of IL-2 (−330 G/T, +166 G/T) and IFN-γ (+874 A/T) single nucleotide polymorphisms in Iranian patients with JIA. To the best of our knowledge, ours is the first study to analyse the role of the above-said gene variants in susceptibility to JIA in an Iranian population.
Interleukin-2 (IL-2) is recognised as a typical T helper 1 (Th1) cytokine, which exerts forceful modulatory effect on various immune cells.11 It has been proposed that dysfunction of the IL-2 could culminate in functional or pathological alterations in the immune system, resulting in autoimmunity.8 It is widely accepted that IL-2 expression is reduced in peripheral blood and both the synovial fluid and the synovium of patients with rheumatoid arthritis (RA) compared to healthy individuals.11,20 Furthermore, IL-2 protein has previously been identified to be decreased in the sera of patients with systemic-onset JIA.19IL-2 SNPs are the probable underlying causes of the variations found in IL-2 concentrations between patients and controls.11 Two single nucleotide polymorphisms have been recognised at −330 G/T and +166 G/T positions from the transcription start site in IL-2 gene.9 The allele G at IL-2 −330 position is known to be associated with higher levels of IL-2 production.1 In the present study, we found overrepresentation of the GG genotype (acknowledged as a polymorphism with an elevated level of IL-2 secretion1) at IL-2 −330 position in the patients group, whilst the frequency of the GT genotype at the same position (identified as a genotype with an intermediate level of cytokine gene expression1) was found to be lower than the controls. Moreover, at the haplotype level, we detected GT haplotype at IL-2 (−330, +166) to be more frequent in patients with JIA compared to healthy individuals. Our findings are in line with the results of the association study carried out by Pawlik et al., which revealed the association of IL-2 −330 GG genotype with RA severity.15
Interferon-gamma (IFN-γ), a pleiotropic cytokine with antiviral properties, is another Th1 cytokine, which affects inflammation through promoting macrophages to secrete inflammatory and tissue-damaging mediators, including proteases, reactive oxygen species, nitric oxide and tumour necrosis factor alpha (TNF-α), as well as enhancing major histocompatibility complex (MHC) class II expression.5 This cytokine is found in the synovial tissue and in the fluid of RA patients.2 In addition, changes in the levels of IFN-γ protein have been previously reported in the sera of patients with systemic-onset JIA.11 As the IFN-γ gene polymorphisms may be the plausible candidates for individuals’ proneness to JIA, the functional SNP at IFN-γ +874 position, which maps to a putative nuclear factor-kB (NF-kB) binding site, has been analysed in the current study. The presence of T allele at the afore-mentioned position enhances the NF-kB binding efficiency resulting in elevated IFN-γ production.18 In the present study, we did not detect any association between SNPs at IFN-γ +874 position and individuals’ vulnerability to JIA.
ConclusionIn brief, our findings suggest that the IL-2 −330 GG genotype together with IL-2 (−33-, +166) GT haplotype confers susceptibility to JIA, while IL-2 −330 GT genotype acts as a protective factor against JIA in an Iranian population. Nevertheless, further multicentre studies with larger sample size could be suggested so as to confirm the associations of the aforementioned polymorphisms of the IL-2 and IFN-γ genes with the proneness to JIA. Evaluation of IL-10 production could also be recommended in subsequent investigations.
Ethical disclosuresConfidentiality of dataThe authors declare that they have followed the protocols of their work centre on the publication of patient data and that all the patients included in the study have received sufficient information and have given their informed consent in writing to participate in that study.
Right to privacy and informed consentThe authors have obtained the informed consent of the patients and/or subjects mentioned in the article. The author for correspondence is in possession of this document.
Protection of human subjects and animals in researchThe authors declare that the procedures followed were in accordance with the regulations of the responsible Clinical Research Ethics Committee and in accordance with those of the World Medical Association and the Helsinki Declaration.
FundingThis study was funded by Tehran University of Medical Sciences and Health Services (Grant Number: 92-03-30-24902).
Conflict of interestThe authors have no conflict of interest to declare.