The SARS-CoV-2 pandemic has changed the urological practice around the world. Our objective is to describe the outcomes presented by patients undergoing surgery in the urology department of a tertiary hospital, across the pandemic phases.
MethodsObservational, cohort study including all patients undergoing surgery from March 1 to May 14. According to the hospital organization, we identified three periods: there were no changes during the first two weeks (1st period), the following seven weeks, when only urgent interventions were carried out after performance of nasopharyngeal swab test (2nd period), and finally, elective surgery was resumed on May 4, after the implementation of a multidisciplinary screening protocol (3rd period). Demographic, baseline, surgical and perioperative variables, as well as postoperative outcomes, were obtained in a retrospective (periods 1 and 2) and prospective (period 3) manner. Telephone follow-up was initiated at least 3 weeks after hospital discharge.
Results103 urological surgeries were performed, and 11 patients were diagnosed with COVID-19, 8 of them within the 1st period. The diagnosis was already known in 1 patient, while the other 10 developed the disease in an average of 25 days after the intervention and 16.6 days after discharge. Of seven transplant patients, four got the infection. Three deaths were recorded due to the disease: a 69-year-old woman transplanted and two men over 80 with comorbidities and high anesthetic risk who underwent drainage of retroperitoneal abscess and retrograde intrarenal surgery, respectively.
ConclusionsSARS-CoV-2 infection mainly affected renal transplant recipients or elderly patients with high anesthetic risk, during the first 2 weeks of the pandemic. After implementing preoperative PCR tests and a comprehensive screening protocol, cases were substantially reduced, and safe surgical procedures were achieved.
La pandemia por SARS-CoV-2 ha cambiado la práctica urológica a nivel mundial. Nuestro objetivo es describir los resultados en salud observados en los pacientes intervenidos en el Servicio de Urología de un hospital terciario, a lo largo de diferentes fases epidemiológicas.
MétodosEstudio de cohortes observacional que incluye todos los pacientes intervenidos entre el 1 de marzo y el 14 de mayo. Según la organización hospitalaria, distinguimos 3 periodos: durante las primeras 2 semanas no hubo cambios (1.er periodo), en las 7 semanas siguientes solo se realizaron intervenciones urgentes previa extracción de exudado nasofaríngeo (2.o periodo), y tras el 4 de mayo se reanudó la cirugía electiva aplicando un protocolo de cribado multidisciplinar (3.er periodo).
Las variables demográficas y basales, las quirúrgicas y perioperatorias, así como los resultados postoperatorios, se obtuvieron de forma retrospectiva (periodos 1 y 2) y prospectiva (periodo 3). El seguimiento telefónico se realizó al menos 3 semanas tras el alta hospitalaria.
ResultadosSe realizaron 103 cirugías urológicas y fueron diagnosticados de COVID-19 11 pacientes, 8 de ellos en el 1.er periodo. El diagnóstico era conocido en un paciente, mientras que los otros 10 desarrollaron la enfermedad en una media de 25 días tras la intervención y 16,6 días tras el alta. Cuatro de 7 pacientes trasplantados resultaron afectados. Se registraron 3 muertes por la enfermedad: una mujer de 69 años trasplantada y 2 varones mayores de 80 años con comorbilidades y alto riesgo anestésico a los que se realizó drenaje de absceso retroperitoneal y cirugía retrógrada intrarrenal, respectivamente.
ConclusionesLa infección por SARS-CoV-2 afectó principalmente a trasplantados renales o pacientes añosos con alto riesgo anestésico, durante las 2 primeras semanas de la pandemia. Tras implantar la PCR preoperatoria y un protocolo completo de cribado, los casos se redujeron de manera sustancial y se pudo operar con seguridad.
Spain is among the European countries most severely affected by the current SARS-CoV-2 coronavirus pandemic. According to official figures published on June 17, 2020, Spain accounts for 244,683 cases and 27,136 deaths. The Community of Madrid accumulates 28.93% of these cases and 32.03% of the deceased.1
Given the high proportion of patients who can develop the severe form of the disease requiring admission to an intensive care unit (ICU), health systems have had to adapt their health policies and resources and reorient them to the management of COVID-19 patients.
Numerous urological associations and working groups have developed recommendations for the reorganization of urological activities and procedures, including surgical ones.2–6 Currently, these guidelines are constantly being reviewed and modified based on the new scientific evidence available on the SARS-CoV-2 coronavirus.7
Literature on patients undergoing surgery during the pandemic is still scarce. A study from Wuhan8 suggests that patients undergoing surgery during the incubation phase of the virus have a high risk of requiring ICU admission (44.1%) and death (20.5%). In the Dublin series9 including 101 urological patients undergoing urgent and elective surgery during the pandemic, who had undergone a screening protocol, 3 patients were diagnosed with COVID-19 and one of them died. The international project CovidSurg has reported a mortality rate of 23.8% at 30 days in patients who underwent surgery with confirmed COVID-19 infection in the 7 days prior to surgery or 30 days after the procedure.10 A multicenter study carried out in Paris11 on urological surgeries performed during the epidemic peak reported a slightly lower mortality of 18.7%.
Our institution is a tertiary hospital in the southern area of Madrid that has 1256 hospital beds and serves a population area of 450,000 inhabitants. The first admissions of COVID-19 patients began on February 29, and since then, there have always been patients hospitalized for COVID until today.
The objective of this study is to characterize the impact of the pandemic on our service across the pandemic phases, especially in terms of surgical outcomes and risk factors that may have influenced the diagnosis and severity of COVID-19 infection.
Material and methodsAn observational study was designed including all patients undergoing surgery in the urology department from March 1 to May 14, 2020. Data were collected retrospectively between March 1 and May 3 and prospectively between May 4 and 14.
Data analysis has been divided into 3 periods according to the country's epidemiological situation and to the organization and policies of our center:
- –
First period (March 1–15): the number of COVID-19 admissions was slowly rising. Hospital and ICU beds were available. Elective surgery activity did not change.
- –
Second period (March 16 to May 3): the high bed occupancy due to COVID-19 forced the suspension of elective surgery activity and kidney transplant program. Urgent surgeries were only performed after carrying out a polymerase chain reaction (PCR) on nasopharyngeal exudates, except for those cases in which we could not wait for the result due to the clinical status of the patient. Protective measures for patients and professionals were maximized.12
- –
Third period (May 4–14): resumption of elective surgery as COVID-19 bed requirements were lower. A preoperative protocol was developed consisting of a telephone consultation carried out 7 days before surgery to rule out the presence of symptoms; if the patient was asymptomatic, a blood test and PCR on nasopharyngeal exudates were then performed within 1–3 days before the intervention.13 In the event of a negative PCR and abnormal findings suggestive of COVID-19 in the blood test, a chest computed tomography (CT) scan was performed1.4–16
After discharge, a follow-up telephone consultation was carried out with all the patients, to ask them about symptoms related to COVID-19 and about their epidemiological situation (number of cohabitants at home and their infection status). This follow-up was carried out at least 3 weeks after discharge to exceed the incubation period as much as possible.
The demographic characteristics of the patients and risk factors for COVID-1917 have been collected: age, sex, Charlson index, arterial hypertension (HT), diabetes mellitus (DM), obesity (body mass index [BMI]>30kg/m2), chronic kidney disease (CKD), acute myocardial infarction, pulmonary pathology, immunosuppression or neoplasia. The preoperative status of COVID-19 was categorized as not evaluated (1st period patients), pending positive and negative results.
In addition, other variables such as time of admission until surgery, length of stay, follow-up time after discharge were considered. Likewise, variables related to the operation and postoperative period were collected: preoperative urological diagnosis, surgical approach, operative time, type of surgery (urgent or elective), time of admission to recovery room, anesthetic risk (ASA), alternatives to surgery, postoperative complications (Clavien-Dindo classification), postoperative ancillary tests (blood test, chest X-ray, chest CT).
In the case of COVID-19 positive, additional variables were collected: presence of fever, cough, asthenia, secretions, dyspnea, anosmia/hyposmia, myalgia/arthralgia, headache, nausea/vomiting, diarrhea; time until the appearance of first symptom, evolution time from first symptom to ICU admission or death, type and duration of treatment, need for oxygen therapy, and patient's status after surgery.
The SAS software 9.4 (© 2013 SAS Institute Inc, Cary, NC, USA) was used for data analysis. Mean and standard deviation were used to characterize the quantitative variables, and frequency expressed in percentages was used for the qualitative variables. The comparison of independent continuous quantitative variables was carried out using non-parametric tests, Kruskal–Wallis or Mann–Whitney U tests. Chi-squared test with Yates correction or Fisher's exact test was used for frequencies. The Pearson correlation coefficient was used to analyze the correlation between quantitative variables.
ResultsDuring the 75 days of study, 103 patients were submitted to surgical procedures: 55 in the 1st period, 28 in the 2nd and 20 in the 3rd. Table 1 shows the comparison of the baseline characteristics of each period, as well as the most frequent diagnoses and surgeries of each one.
Comparison of the baseline characteristics across the different periods. Most frequent diagnoses and surgeries.
| First period (n=55) | Second period (n=28) | Third period (n=20) | p-value | |
|---|---|---|---|---|
| Age (years) | 62.1 (16.48) | 60.21 (19.61) | 63.35 (14.6) | 0.138 |
| Sex (female) | 15 (27.3%) | 10 (35.7%) | 4 (20%) | 0.480 |
| Charlson index (p50) | 3 | 3 | 5 | 0.229 |
| # 1 (diagnosis/surgery) | Bladder neoplasm/TURB (11, 20%) | RUC and sepsis/DJ (10, 35.7%) | Bladder neoplasm/TURB (11, 55%) | N/A |
| # 2 (diagnosis/surgery) | BPH/TURP (8, 14.5%) | Hematuria/TURB-h (5, 17.9%) | RUC and sepsis/DJ (3, 15%) | N/A |
| # 3 (diagnosis/surgery) | CKD/Kidney transplant (7, 12.7%) | RUC and AKF/URS (2, 7.1%) | Prostate Cancer/LRP (2, 10%) | N/A |
# 1, 2, 3 (diagnosis/surgery): these represent the first, second and third preoperative diagnosis and most frequent surgical intervention in this period.
RUC: renoureteral colic; DJ: double J catheter; CKD: chronic kidney disease; AKF: acute kidney failure; BPH: benign prostatic hyperplasia; N/A: not applicable; LRP: laparoscopic radical prostatectomy; TURB: transurethral resection of the bladder; TURP: transurethral resection of the prostate; TUR-h: transurethral resection of the bladder-hemostatic; URS: ureterorenoscopy.
The mean follow-up was 48.8 days, and 11 patients were diagnosed with COVID-19; 8 from period 1 and 3 from period 2. One of them underwent surgery with confirmed diagnosis and the other 10 developed the disease in a mean of 25 days after surgery and 16.6 days after discharge.
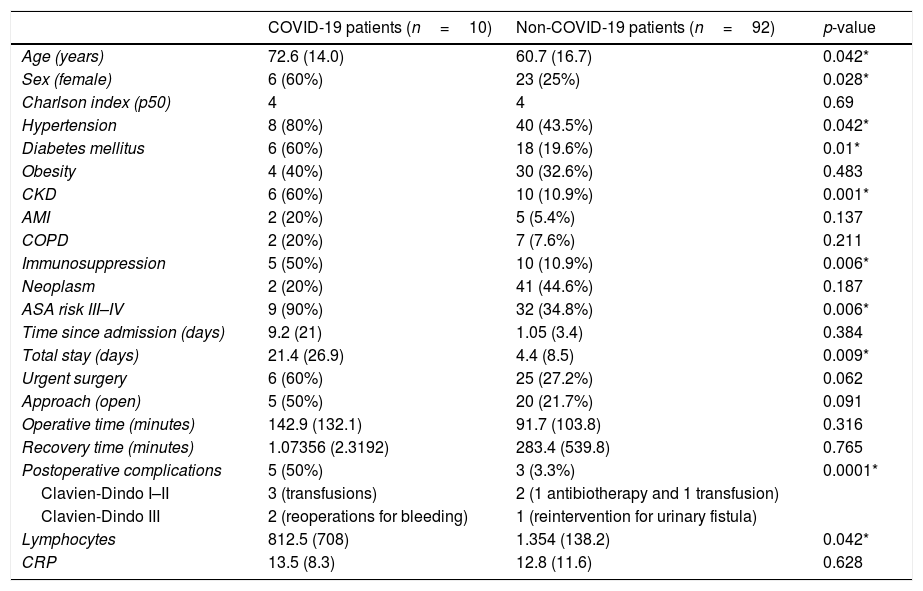
The surgical procedure with highest COVID-19 incidence was kidney transplantation with 57.1% (4 positive patients out of 7 transplant procedures). Older age, female sex, HT, DM, CKD, immunosuppression, and high anesthetic risk have been common characteristics among COVID-19 patients. These patients had a longer hospital stay and more postoperative complications. The results and the comparison of data between COVID-19 and non-COVID-19 patients are shown in Table 2.
Comparison of the postoperative period between COVID-19 and non-COVID-19 patients.
| COVID-19 patients (n=10) | Non-COVID-19 patients (n=92) | p-value | |
|---|---|---|---|
| Age (years) | 72.6 (14.0) | 60.7 (16.7) | 0.042* |
| Sex (female) | 6 (60%) | 23 (25%) | 0.028* |
| Charlson index (p50) | 4 | 4 | 0.69 |
| Hypertension | 8 (80%) | 40 (43.5%) | 0.042* |
| Diabetes mellitus | 6 (60%) | 18 (19.6%) | 0.01* |
| Obesity | 4 (40%) | 30 (32.6%) | 0.483 |
| CKD | 6 (60%) | 10 (10.9%) | 0.001* |
| AMI | 2 (20%) | 5 (5.4%) | 0.137 |
| COPD | 2 (20%) | 7 (7.6%) | 0.211 |
| Immunosuppression | 5 (50%) | 10 (10.9%) | 0.006* |
| Neoplasm | 2 (20%) | 41 (44.6%) | 0.187 |
| ASA risk III–IV | 9 (90%) | 32 (34.8%) | 0.006* |
| Time since admission (days) | 9.2 (21) | 1.05 (3.4) | 0.384 |
| Total stay (days) | 21.4 (26.9) | 4.4 (8.5) | 0.009* |
| Urgent surgery | 6 (60%) | 25 (27.2%) | 0.062 |
| Approach (open) | 5 (50%) | 20 (21.7%) | 0.091 |
| Operative time (minutes) | 142.9 (132.1) | 91.7 (103.8) | 0.316 |
| Recovery time (minutes) | 1.07356 (2.3192) | 283.4 (539.8) | 0.765 |
| Postoperative complications | 5 (50%) | 3 (3.3%) | 0.0001* |
| Clavien-Dindo I–II | 3 (transfusions) | 2 (1 antibiotherapy and 1 transfusion) | |
| Clavien-Dindo III | 2 (reoperations for bleeding) | 1 (reintervention for urinary fistula) | |
| Lymphocytes | 812.5 (708) | 1.354 (138.2) | 0.042* |
| CRP | 13.5 (8.3) | 12.8 (11.6) | 0.628 |
ASA: American Society of Anesthesiologists; COPD: chronic obstructive pulmonary disease; CKD: chronic kidney disease; AMI: acute myocardial infarction; Lymphocytes: lymphocytes in postoperative lab test (per μl); CRP: postoperative C-reactive protein (mg/dL).
Two patients presented symptoms during admission: a 69-year-old woman who was admitted for kidney transplantation and was reoperated for bleeding, and an 85-year-old man, ASA IV, who presented a giant retroperitoneal abscess after 67 days of admission to Internal Medicine. Both patients had a poor evolution: they developed acute respiratory distress syndrome (ARDS) and death.
Another 3 patients developed the infection in the 2 weeks following surgery (9–10 days), requiring readmission at 6, 19 and 37 days, respectively.
The remaining COVID-19 positive (5 patients) cases presented the first symptom between 28 and 42 days after surgery (21 to 28 days after discharge), 2 of them were institutionalized patients (one died) and the other 3 lived with family members who had recently contracted the infection. Therefore, we believe these 5 patients were infected outside the hospital setting.
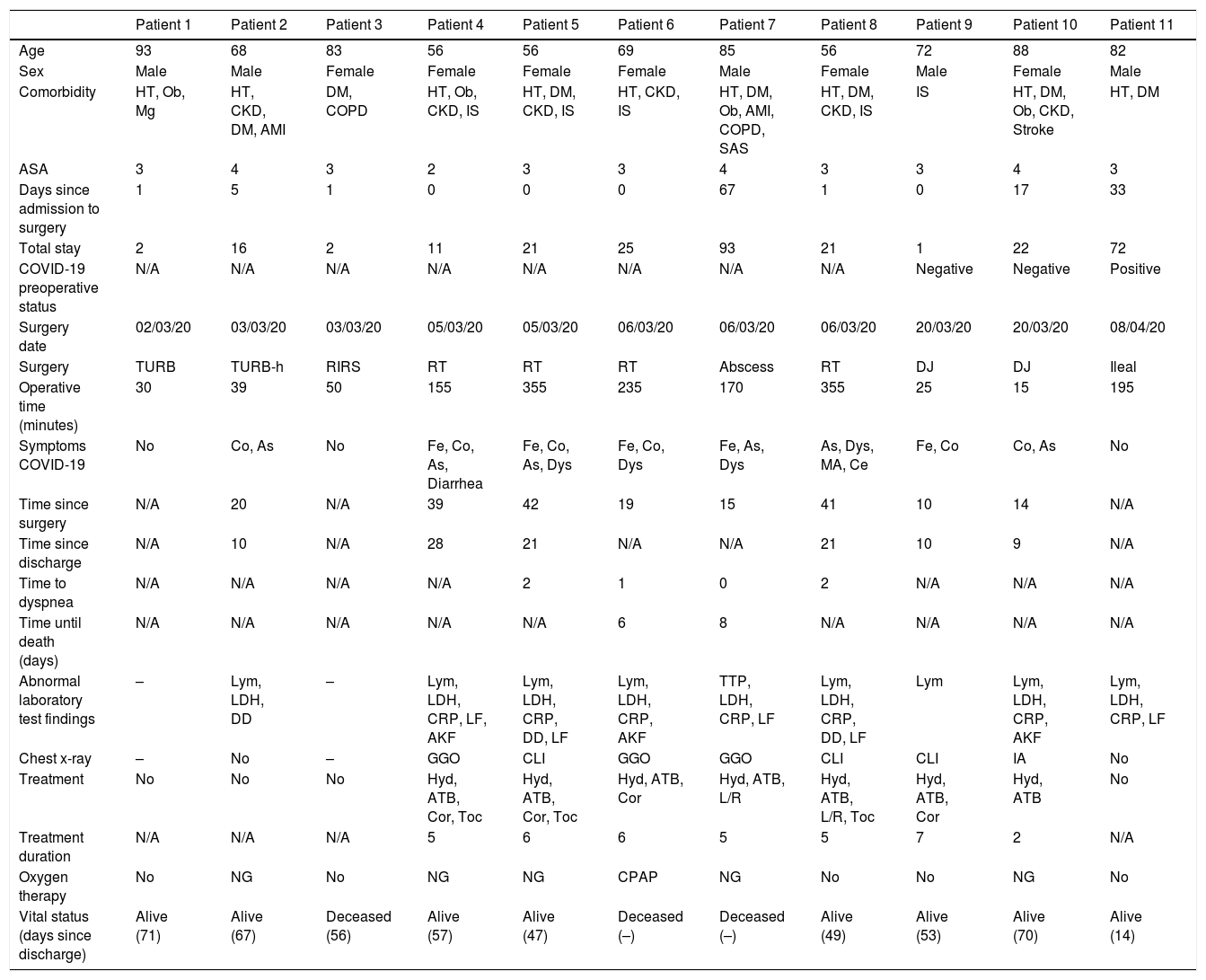
The most frequent symptoms were fever (5 patients), cough (6 patients) and asthenia (6 patients). Six patients required non-invasive ventilation, one of them with continuous positive airway pressure (CPAP). Hydroxychloroquine was the most widely used treatment, with a mean duration of 5 days. The complete description of the characteristics of COVID-19 positive patients is shown in Table 3.
Description of all COVID-19 patients.
| Patient 1 | Patient 2 | Patient 3 | Patient 4 | Patient 5 | Patient 6 | Patient 7 | Patient 8 | Patient 9 | Patient 10 | Patient 11 | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Age | 93 | 68 | 83 | 56 | 56 | 69 | 85 | 56 | 72 | 88 | 82 |
| Sex | Male | Male | Female | Female | Female | Female | Male | Female | Male | Female | Male |
| Comorbidity | HT, Ob, Mg | HT, CKD, DM, AMI | DM, COPD | HT, Ob, CKD, IS | HT, DM, CKD, IS | HT, CKD, IS | HT, DM, Ob, AMI, COPD, SAS | HT, DM, CKD, IS | IS | HT, DM, Ob, CKD, Stroke | HT, DM |
| ASA | 3 | 4 | 3 | 2 | 3 | 3 | 4 | 3 | 3 | 4 | 3 |
| Days since admission to surgery | 1 | 5 | 1 | 0 | 0 | 0 | 67 | 1 | 0 | 17 | 33 |
| Total stay | 2 | 16 | 2 | 11 | 21 | 25 | 93 | 21 | 1 | 22 | 72 |
| COVID-19 preoperative status | N/A | N/A | N/A | N/A | N/A | N/A | N/A | N/A | Negative | Negative | Positive |
| Surgery date | 02/03/20 | 03/03/20 | 03/03/20 | 05/03/20 | 05/03/20 | 06/03/20 | 06/03/20 | 06/03/20 | 20/03/20 | 20/03/20 | 08/04/20 |
| Surgery | TURB | TURB-h | RIRS | RT | RT | RT | Abscess | RT | DJ | DJ | Ileal |
| Operative time (minutes) | 30 | 39 | 50 | 155 | 355 | 235 | 170 | 355 | 25 | 15 | 195 |
| Symptoms COVID-19 | No | Co, As | No | Fe, Co, As, Diarrhea | Fe, Co, As, Dys | Fe, Co, Dys | Fe, As, Dys | As, Dys, MA, Ce | Fe, Co | Co, As | No |
| Time since surgery | N/A | 20 | N/A | 39 | 42 | 19 | 15 | 41 | 10 | 14 | N/A |
| Time since discharge | N/A | 10 | N/A | 28 | 21 | N/A | N/A | 21 | 10 | 9 | N/A |
| Time to dyspnea | N/A | N/A | N/A | N/A | 2 | 1 | 0 | 2 | N/A | N/A | N/A |
| Time until death (days) | N/A | N/A | N/A | N/A | N/A | 6 | 8 | N/A | N/A | N/A | N/A |
| Abnormal laboratory test findings | – | Lym, LDH, DD | – | Lym, LDH, CRP, LF, AKF | Lym, LDH, CRP, DD, LF | Lym, LDH, CRP, AKF | TTP, LDH, CRP, LF | Lym, LDH, CRP, DD, LF | Lym | Lym, LDH, CRP, AKF | Lym, LDH, CRP, LF |
| Chest x-ray | – | No | – | GGO | CLI | GGO | GGO | CLI | CLI | IA | No |
| Treatment | No | No | No | Hyd, ATB, Cor, Toc | Hyd, ATB, Cor, Toc | Hyd, ATB, Cor | Hyd, ATB, L/R | Hyd, ATB, L/R, Toc | Hyd, ATB, Cor | Hyd, ATB | No |
| Treatment duration | N/A | N/A | N/A | 5 | 6 | 6 | 5 | 5 | 7 | 2 | N/A |
| Oxygen therapy | No | NG | No | NG | NG | CPAP | NG | No | No | NG | No |
| Vital status (days since discharge) | Alive (71) | Alive (67) | Deceased (56) | Alive (57) | Alive (47) | Deceased (–) | Deceased (–) | Alive (49) | Alive (53) | Alive (70) | Alive (14) |
The symbol “–” indicates “not performed or unknown”, depending on the context. N/A: not applicable.
ASA: American Society of Anesthesiologists.
Comorbidities, DM: diabetes mellitus; COPD: chronic obstructive pulmonary disease; CKD: chronic kidney disease; HT: hypertension; AMI: acute myocardial infarction; IS: immunosuppression; Mg: malignancy; Ob: obesity; SAS: sleep apnea syndrome.
Surgeries (procedures), Abscess: open surgical drainage of retroperitoneal abscess; DJ: double J catheter; Ileal: intestinal diversion with ileal conduit; RIRS: retrograde intrarenal surgery; TURB: transurethral resection of the bladder; TURB-h: transurethral resection of the bladder-hemostasis; RT: renal transplantation.
Symptoms, As: asthenia; Ce: cephalea; Dys: dyspnea; Fe: fever; MA: myalgias or arthralgias; Co: cough.
Abnormal laboratory findings (postoperative), DD: d-dimer (>500ng/mL); LF: liver function; AKF: acute kidney failure; LDH: lactate dehydrogenase (>250U/L); Lym: lymphopenia (<1.200/l); CRP: C-reactive protein (>5mg/dL); TTP: thrombocytopenia (<140.000/μl).
Chest X-ray (CXR) alterations, IA: interstitial alterations; BCLI: bilateral cotton-like infiltrates; GGO: ground glass opacity.
Treatment, ATB: antibiotics; Cor: corticosteroids; Hyd: hydroxychloroquine; L/R: lopinavir/ritonavir; Toc: tocilizumab.
Oxygen therapy (O2 requirements2), CPAP: continuous positive airway pressure; NG: nasal glasses.
Our study describes the outcomes of all surgical interventions performed in a urology department located in one of the European areas most affected by the SARS-CoV-2 pandemic. The change in institutional policy, the allocation of urologists to the care of COVID-19 patients, and the temporary suspension of elective surgery are common circumstances among urology departments in Spain.18 It must be highlighted that the mean number of surgeries performed daily during periods 1 and 3 (3.67 and 1.82 respectively) is higher than the number of procedures in the second period (0.57) because only urgent surgeries were performed during this period. Baseline characteristics of patients (age, sex, Charlson) were similar across periods. Several centers in Italy19 and Portugal20 have had a lower volume of activity compared to the same periods in the previous year, but with a higher proportion of complicated cases requiring urgent intervention.
In order to schedule surgical activity during period 3, the most urgent procedures were prioritized following the recommendations of the European Association of Urology,2 either for oncological reasons or because of quality of life loss.21 In addition, during this last period, a multidisciplinary institutional protocol was followed, based on the recommendations of the health authorities and the available scientific evidence, with the intention of applying the best clinical practices in our hospital.13
In the retrospective study from Wuhan, 34 patients developed COVID-19 in a mean period of 2 days after surgery, assuming they underwent surgery already in the incubation period.8 Fifteen patients (44.1%) required ICU support and 7 of them died (20.5%). When comparing the patients that required ICU support with those who did not, ICU patients were of a more advanced age, had greater comorbidity, had been submitted to longer and more complex surgeries, and presented more abnormal laboratory findings (leukocytosis and leukopenia). Therefore, the authors suggest that these factors could predict a poor outcome. They also hypothesize that surgery could have accelerated and exacerbated COVID-19 due to the rapid appearance of the first symptom (mean of 2.6 days after surgery), dyspnea (3.5 days from first symptom) and death (8.7 days from first symptom). The possible biases of this study are that the authors do not report the total number of surgeries performed during those days, and that patients were admitted with a mean of 2.5 days before surgery, which overlaps with the incubation period of SARS-CoV-2 (2–7 days).16
Our cohort includes all patients submitted to surgery between March 1 and May 14, 2020, with a mean number of days of admission prior to surgery of 1. This reduces the risk of being infected with COVID-19 while being at the hospital awaiting intervention, in contrast to the Wuhan cohort. Ten patients (9.7%) got infected with COVID-19 after surgery, of which 3 (30%) died. The mean number of days from surgery until the first symptom was 25 days, which is longer than the period reported about Wuhan. However, the time between the first symptom and the appearance of dyspnea (5 days) and death (7 days) is similar in both cohorts. Similarly, these patients were older and with more associated comorbidities than those who did not get infected.
An international multicenter study10 including 1128 patients who underwent surgery in 235 hospitals, with COVID-19 confirmed 7 days before or 30 days after surgery, between January 1 and March 31, reports a mortality of 23.8% after 30 days of follow-up. It also identifies certain groups with higher risk: men (OR 1.75), >70 years old (OR 2.3), urgent surgery (OR 1.67), ASA>III (OR 2.35), malignancy vs. benignity or obstetric cause (OR 1.55) and major surgery vs. minor surgery (OR 1.52). Those who presented ARDS had a mortality of 63%.
Our study also shows a higher risk in patients of advanced age and higher anesthetic risk. On the contrary, urgent surgery does not reach statistical significance, and we found a higher proportion of women with COVID-19, and ARDS had a mortality of 100%.
Eight of the 11 COVID-19 patients (72.7%) belonged to the 1st period (representing 14.5% of the 1st period), in which there were still no specific safety recommendations in our hospital. Once the preoperative PCR (2nd period) and the total protocol (3rd period) were established, the percentage of COVID-19 patients decreased to 10.7% (3 patients out of 28 in the 2nd period) and 0% in the 3rd period. These data are similar to those published by an Irish group9 that implemented a preoperative screening protocol and analyzed a 7-week period in which they found 3 cases of SARS-CoV-2 infection in 101 surgical procedures performed (day 4, 14 and 20 after surgery). These patients had longer hospital stay as a consequence of respiratory complications: 2 of them required CPAP/BiPAP and the third required invasive ventilation in the ICU and finally died.
The most frequent symptoms have been fever, cough and asthenia, as in the series by Guan et al.17 Our study has a higher proportion of dyspnea (36.3%). Treatment schemes have not been constant across periods due to the evolution of the available scientific evidence and the updating of hospital protocols. Hydroxychloroquine and antibiotics were administered in all patients who required treatment (7 of 11, 63.6%) and 6 patients required non-invasive ventilation (one of them CPAP).
The surgery with the highest incidence of COVID-19 was kidney transplantation with 57.1% (4 patients out of 7). Patients with CKD are at higher risk of developing infectious complications due to their basal impaired immune system and to the immunosuppressive therapy after transplantation22. We must consider the fact that, in our cohort, these patients were operated on during the 1st period with the associated problems that we have already mentioned.
A retrospective study comparing the clinical evolution between 10 kidney transplant patients with SARS-CoV-2 pneumonia and 10 patients with healthy immune system shows greater severity and duration of the disease in transplanted patients: 3 required non-invasive ventilation and one died due to acute respiratory failure.23
On the other hand, the higher proportion of postoperative complications among COVID-19 patients in our series is partially influenced by kidney transplantation, since 3 of these 5 complications were blood transfusions in these patients (Clavien-Dindo classification grade II).
Our series does not have a very high proportion of patients with a diagnosis of malignancy infected with COVID-19 (4.7%, 2 patients out of 43). However, in other series, these patients have been associated with greater susceptibility and worse results due to the immunosuppression caused by the neoplasia itself and its treatment (chemotherapy, radiotherapy or surgery).24
Our study has the intrinsic limitations of a single center retrospective analysis. Despite being a small sample, it includes all surgeries performed at the urology department of a high-volume hospital during a period of 75 days that includes the different pandemic phases of our country. For this reason, we believe that it can represent the experience of many hospitals around the world, emphasizing the importance of preoperative screening protocols and patient selection during the pandemic.
ConclusionsIn the retrospective analysis of the patients undergoing surgery in our urology department since the beginning of the SARS-CoV-2 pandemic, we have found an incidence of COVID-19 similar to other surgical series, mostly concentrated in kidney transplant patients or elderly patients with high anesthetic risk at the beginning of the pandemic. After the implementation of preoperative PCR and a multidisciplinary screening protocol, cases were drastically reduced, and surgeries could be performed in a safe manner.
FundingThe authors assure that there is no sponsor that may have influenced the results of this study.
Conflict of interestThe authors assure that there is no conflict of interest with any organization and/or funding related to the subject covered in the manuscript.
Please cite this article as: González-Díaz A, Abad-López P, Peña-Vallejo E, Caro-González MP, Calzas-Montalvo C, Gil-Moradillo J, et al. Cirugía urológica durante la pandemia por SARS-CoV-2. Análisis descriptivo de la experiencia en un Servicio de Urología durante distintas fases epidemiológicas. Actas Urol Esp. 2020;44:665–673.