The role of prostate cancer (PC) screening is currently being questioned. The objective of the European Randomized Study of Screening for Prostate Cancer (ERSPC) was to demonstrate whether PC screening reduced mortality from this disease. The results from the Spanish branch of this study are presented: all-cause and cancer-specific mortality, the characteristics of the detected tumors, primary treatments and progression to advanced disease.
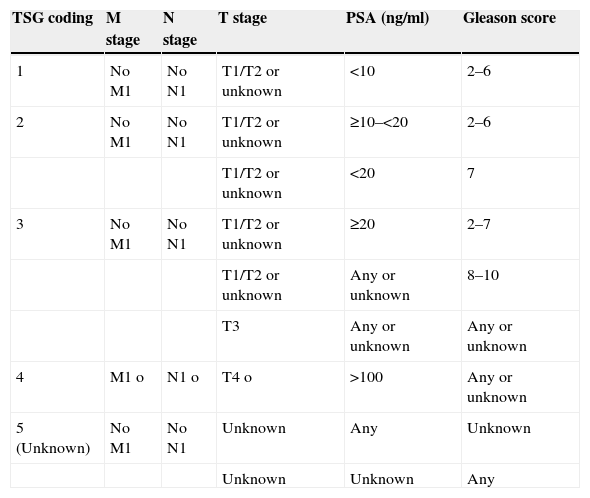
Materials and methodsA total of 18,612 men, between the ages of 45 and 70, were invited to participate in the study, excluding those with a life expectancy of less than 10 years. The men were randomized to the screening arm (serum prostate-specific antigen [PSA] reading) or the control arm (no diagnostic tests). Randomized transrectal ultrasound-guided sextant prostate biopsies were indicated for the men in the screening arm with PSA levels ≥3ng/ml. The detected PCs were identified (stage and primary treatment), as well as the deaths that occurred (date and cause of death).
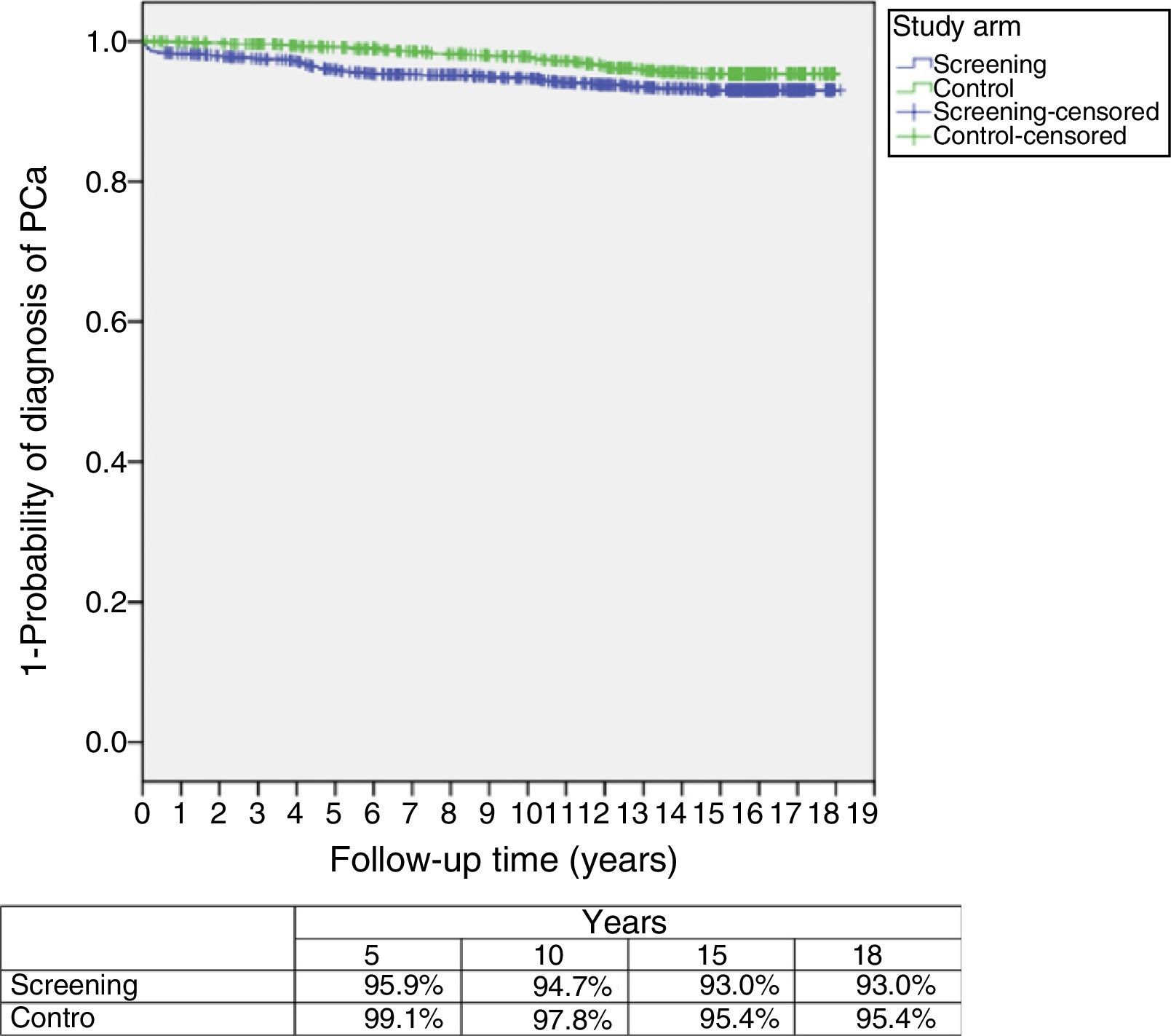
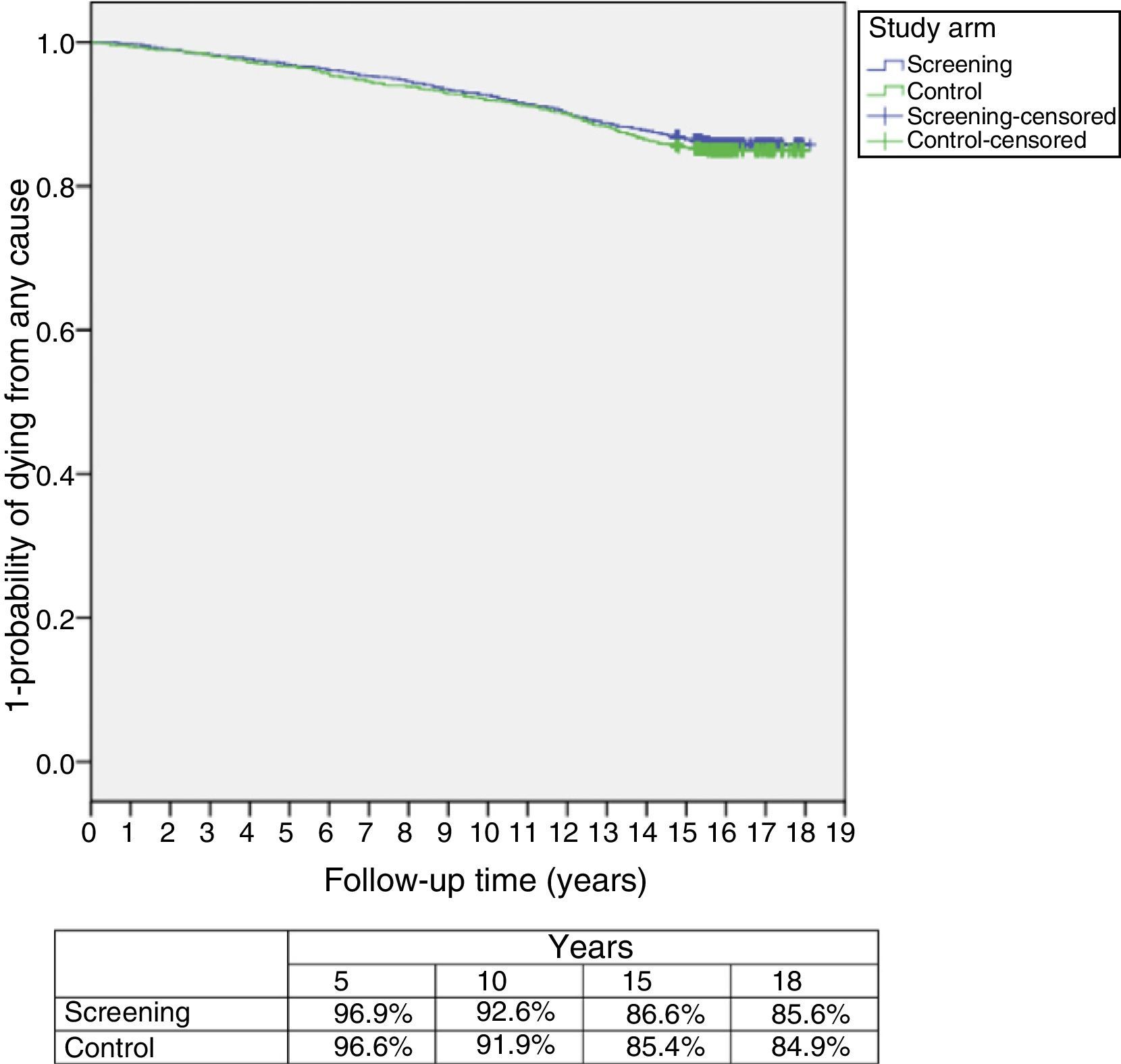
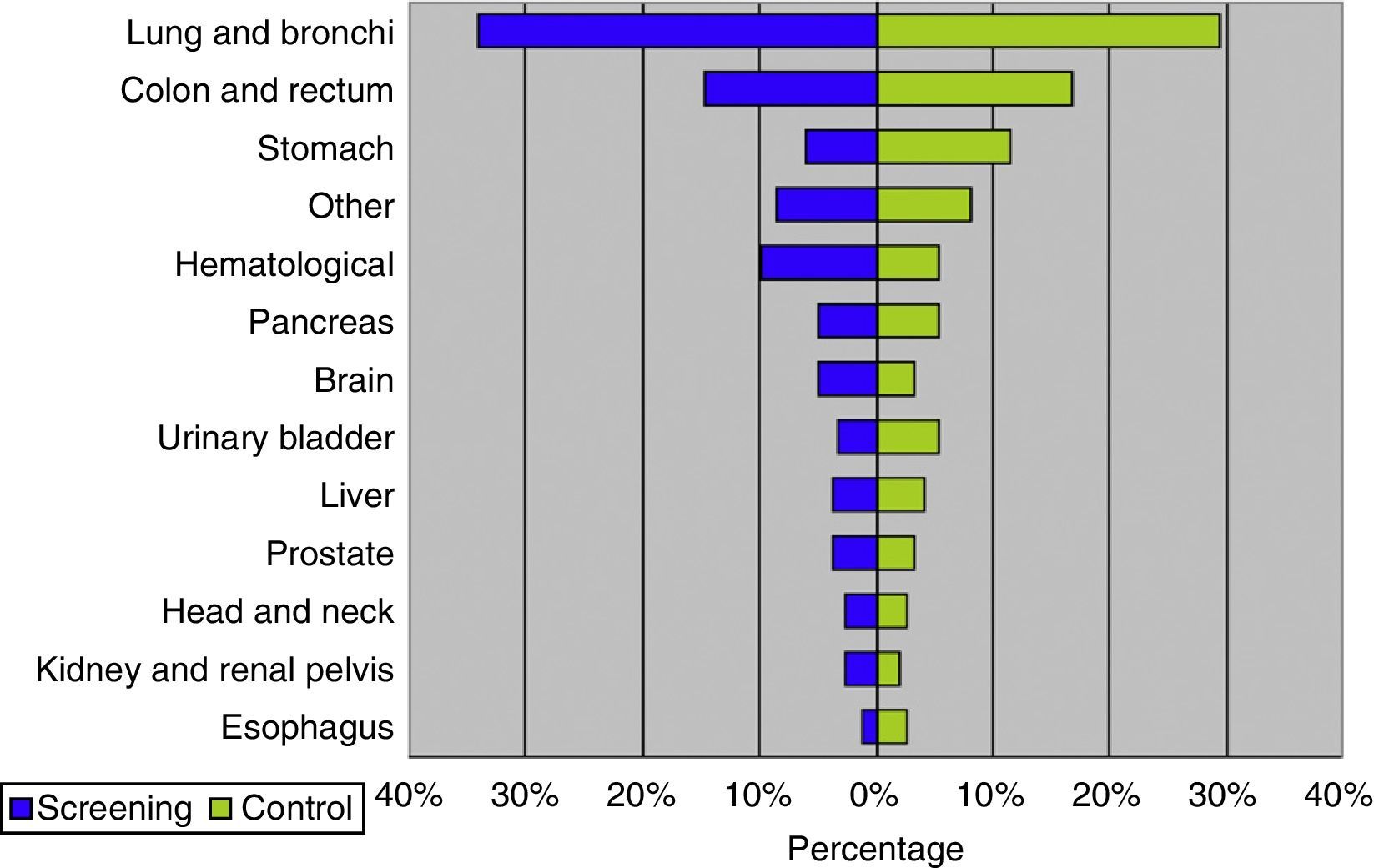
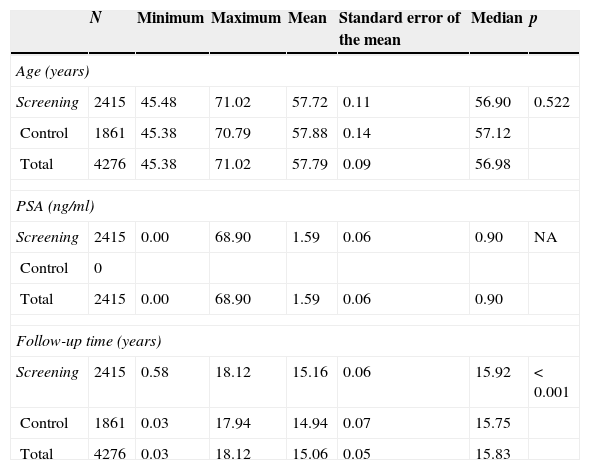
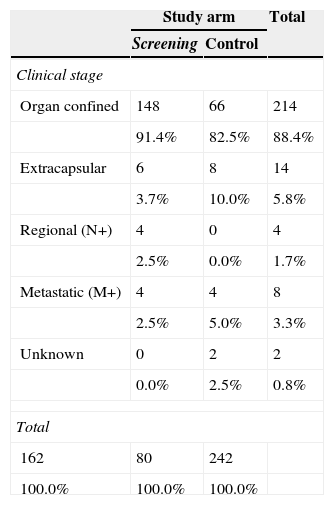
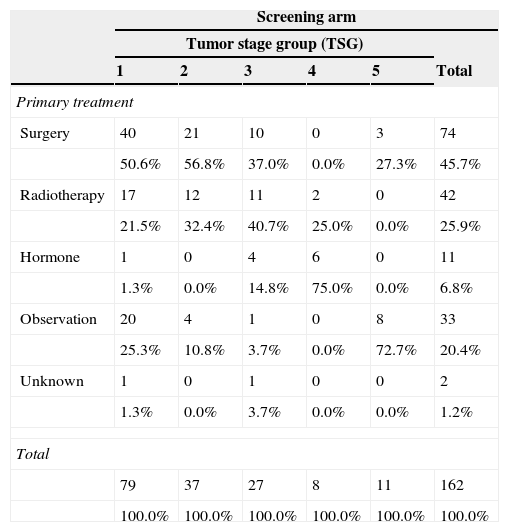
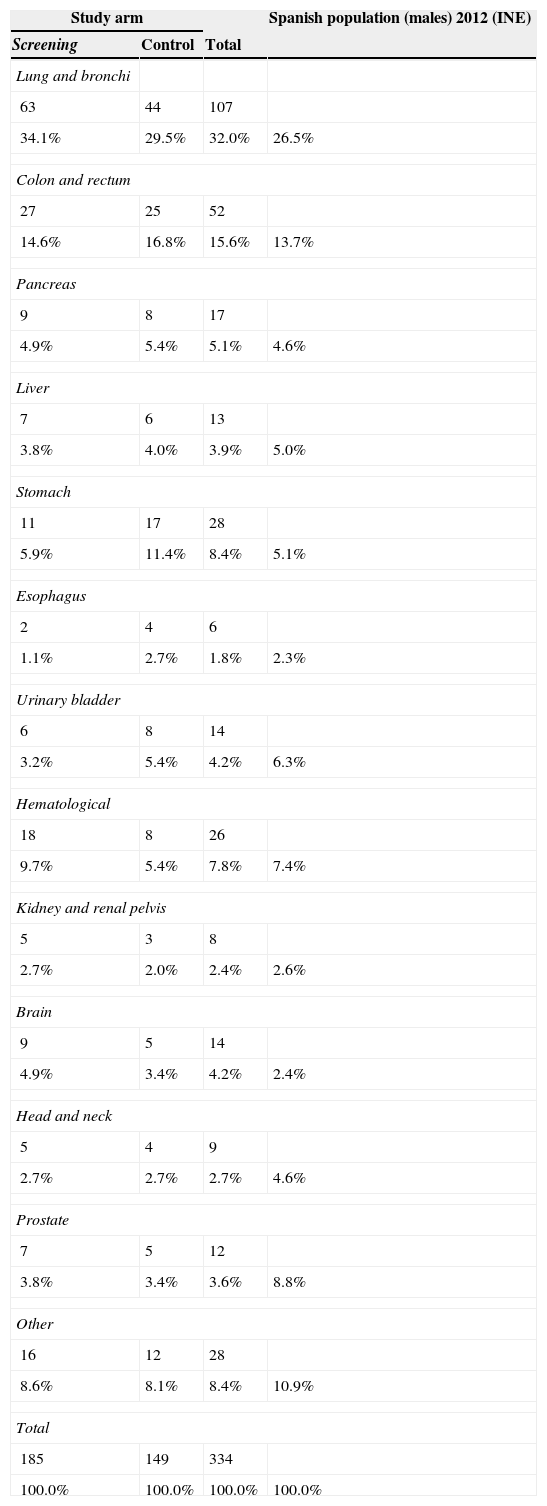
ResultsThe study was performed with 4276 men (2415 in the screening arm and 1861 in the control arm). The median age and serum PSA level were 57 years and 0.90ng/mL, respectively. The median follow-up time was 15.8 years. A total of 242 PCs were diagnosed, 162 (6.7%) in the screening arm and 80 (4.3%) in the control arm (p<.001). Of these, 214 (88.4%) had an organ-confined clinical stage at onset (91.4% in the screening arm vs. 82.5% in the control arm; p=.024). A total of 112 patients (46.3%) underwent radical prostatectomy, 53 (21.9%) underwent prostate radiation therapy, 24 (9.9%) underwent hormone therapy and 47 (19.4%) were kept under observation. A total of 18 PCs progressed to advanced disease (M+ or PSA levels >100ng/mL), with no differences between the study arms (p=.938). A total of 618 (14.5%) patients died during follow-up: 340 (14.1%) in the screening arm and 278 (14.9%) in the control arm, with no differences between the arms in terms of cancer-specific (p=.907) or all-cause (p=.399) mortality. The main causes of death were neoplasia (54.0%), cardiovascular (17.6%), respiratory (8.7%) and gastrointestinal (4.0%), with no difference between study arms. Of the 334 patients who died from neoplasia, only 12 (3.6%) died from PC.
ConclusionsPC screening results in a shifting of the diagnosis toward earlier stages. Nevertheless, we have not demonstrated a benefit in terms of overall or cancer-specific survival after more than 15 years of follow-up. The low mortality from this disease in our community could be one of the main factors that explain these results.
En la actualidad el papel del screening del cáncer de próstata (CaP) está cuestionado. El estudio aleatorizado europeo de screening del cáncer de próstata (ERSPC) tiene como objetivo demostrar si el screening del CaP reduce la mortalidad por esta enfermedad. Se muestran los resultados en la rama española de este estudio: mortalidad global y cáncer-específica, características de los tumores detectados, de los tratamientos primarios y de la progresión a enfermedad avanzada.
Material y métodosSe invitó a participar a 18.612 varones entre los 45 y 70 años de edad, excluyendo aquellos con una expectativa de vida inferior a 10 años. Se llevó a cabo aleatorización a brazo screening (determinación de PSA sérico) y brazo control (no pruebas diagnósticas). Se indicó biopsia prostática aleatorizada sextante dirigida por ecografía transrectal en los varones del brazo screening con PSA≥3ng/ml. Se identificaron los CaP detectados (estadio y tratamiento primarios), así como los fallecimientos producidos (fecha y causa de la muerte).
ResultadosEl estudio se llevó a cabo con 4.276 varones (2.415 brazo screening, 1.861 brazo control). Las medianas de edad y PSA sérico fueron de 57 años y 0,90ng/ml respectivamente. El tiempo de seguimiento (mediana) fue de 15,8 años. Se diagnosticaron 242 CaP, 162 (6,7%) en el brazo screening y 80 (4,3%) en el control (p<0,001). De ellos 214 (88,4%) comenzaron con estadio clínico organoconfinado (91,4% brazo screening vs 82,5% control, p=0,024). Un total de 112 pacientes (46,3%) fue sometido a prostatectomía radical, 53 (21,9%) a radioterapia prostática, 24 (9,9%) a tratamiento hormonal y 47 (19,4%) a observación. Un total de 18 CaP evolucionaron a enfermedad avanzada (M+ o PSA>100ng/ml), sin diferencia entre los brazos del estudio (p=0,938). Un total de 618 (14,5%) fallecieron a lo largo del seguimiento: 340 (14,1%) en el brazo screening y 278 (14,9%) en el control, sin diferencias entre brazos en términos de mortalidad cáncer-específica (p=0,907) ni por todas las causas (p=0,399). Las principales causas de muerte encontradas fueron neoplasia (54,0%), cardiovascular (17,6%), respiratoria (8,7%) y digestiva (4,0%), sin diferencia entre brazos. De los 334 pacientes fallecidos por neoplasia tan solo 12 (3,6%) murieron por CaP.
ConclusionesEl screening del CaP produce una migración del diagnóstico hacia estadios más precoces. No obstante, no hemos demostrado un beneficio en términos de supervivencia global ni cáncer-específica tras más de 15 años de seguimiento. La baja mortalidad por esta enfermedad en nuestro entorno podría ser uno de los principales factores para explicar estos resultados.