Nivolumab is an immunotherapy agent that has been an approved treatment for previously treated patients with advanced renal cell carcinoma (RCC). Experience in real-life settings, especially regarding immune-related adverse events, is scarce. We present our experience with reference to the safety of nivolumab in patients with metastatic RCC (mRCC) treated in 9 hospitals in Spain.
Material and methodsRetrospective, multicenter study of patients with mRCC treated with nivolumab between 2016 and 2018. Data on baseline socio-demographic and clinical characteristics and drug-related adverse events were collected.
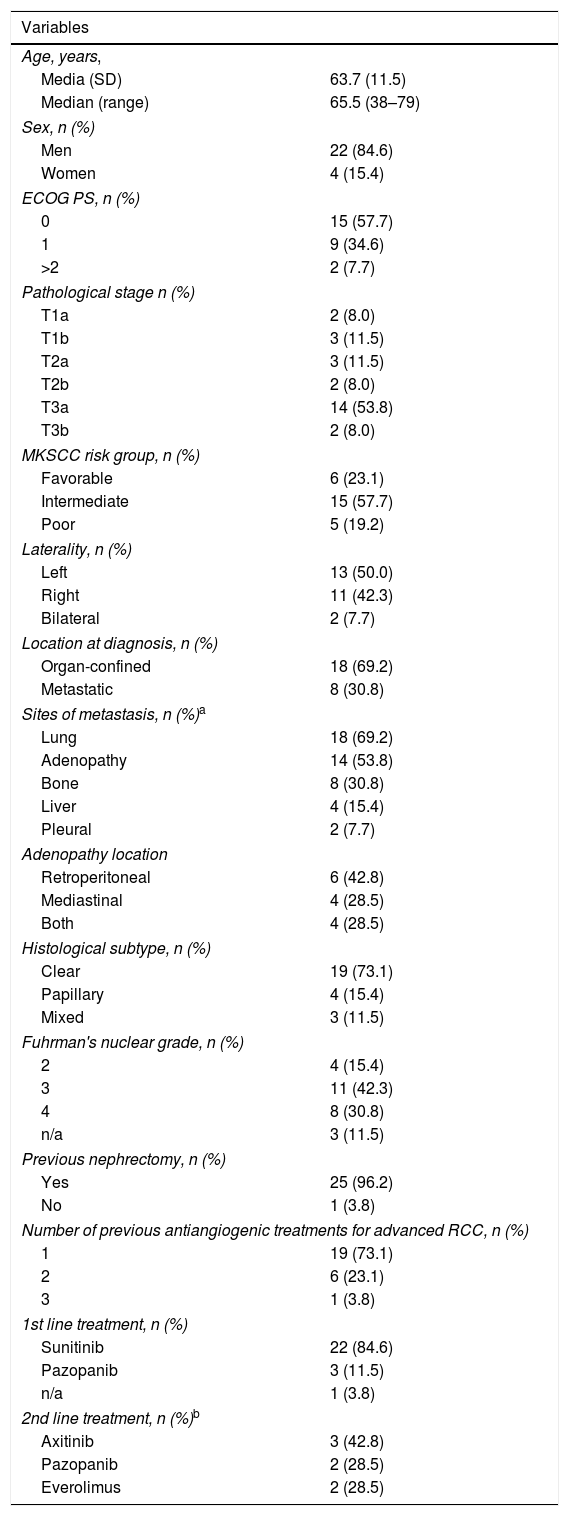
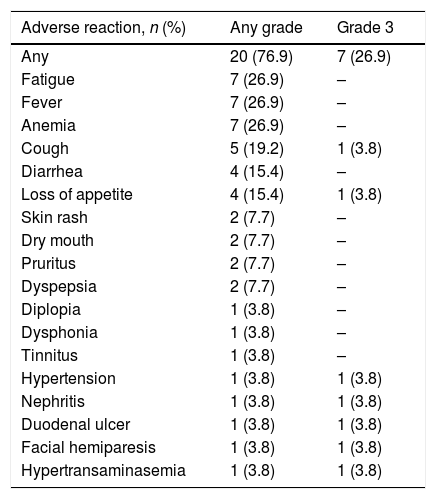
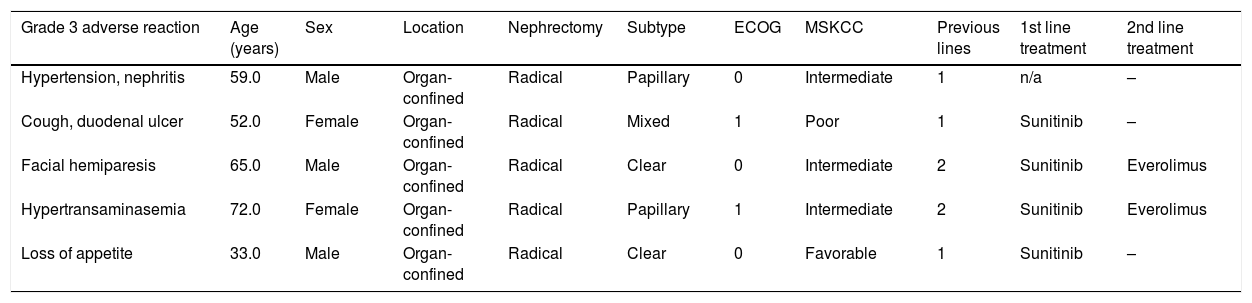
ResultsThe mean age of the 26 patients included was 63.7±11.5 years; 96% were ECOG 0–1 and 78% had favorable or intermediate MSKCC risk scores; 73% had the clear cell histological subtype and 30% metastatic disease. Median follow-up was 9 months (range 1–14). All patients experienced an adverse event at different grades, with fatigue, fever and anemia being the most common (27%). Grade 3 adverse events occurred in 23% of patients. Adverse reactions led to treatment suspension in 3 patients (11%).
ConclusionIn the real-life clinical setting, nivolumab shows favorable outcomes, similar to those reported by other studies.
Nivolumab es un agente inmunoterapéutico aprobado para el tratamiento de pacientes con carcinoma de células renales (CCR) avanzado tratados previamente. La experiencia en práctica clínica real, especialmente en lo referente a la aparición de reacciones adversas inmunorrelacionadas, es escasa. Presentamos la experiencia acerca de la seguridad de nivolumab en pacientes con CCR metastásico (CCRm) tratados en 9 hospitales de España.
Material y métodosEstudio retrospectivo, multicéntrico en pacientes con CCRm tratados con nivolumab entre 2016 y 2018. Se recogieron datos sociodemográficos y clínicos basales y las reacciones adversas relacionadas con el fármaco.
ResultadosLos 26 pacientes incluidos presentaron una edad de 63,7±11,5 años. El 96% presentaba ECOG 0-1 y el 78% un riesgo MKSCC favorable/intermedio. El 73% presentaba subtipo histológico de células claras y el 30%, metástasis de inicio. La mediana de seguimiento fue de 9 meses (rango: 1-14). El 100% de los pacientes presentó una reacción adversa de cualquier grado; las más frecuentes fueron la fatiga, la fiebre y la anemia (27%). El 23% presentó una reacción adversa de grado 3. Las reacciones adversas llevaron a la suspensión del tratamiento en 3 pacientes (11%).
ConclusiónEn la práctica clínica real, nivolumab presenta un perfil de seguridad favorable y manejable, similar al descrito en otros estudios.