To study the relationship between quantitative mRNA determination (hTERT) in patients with bladder tumor, history of bladder tumor, and in subjects without a history of this neoplasia.
Material and methodsA prospective randomized controlled study with 91 subjects included. The value of mRNA-hTERTN was determined in 63 patients with a history or suspicion of bladder tumor and in 28 controls. Urine samples were sent for evaluation of the mRNA level (hTERT), the cytological study and the NMP22 result.
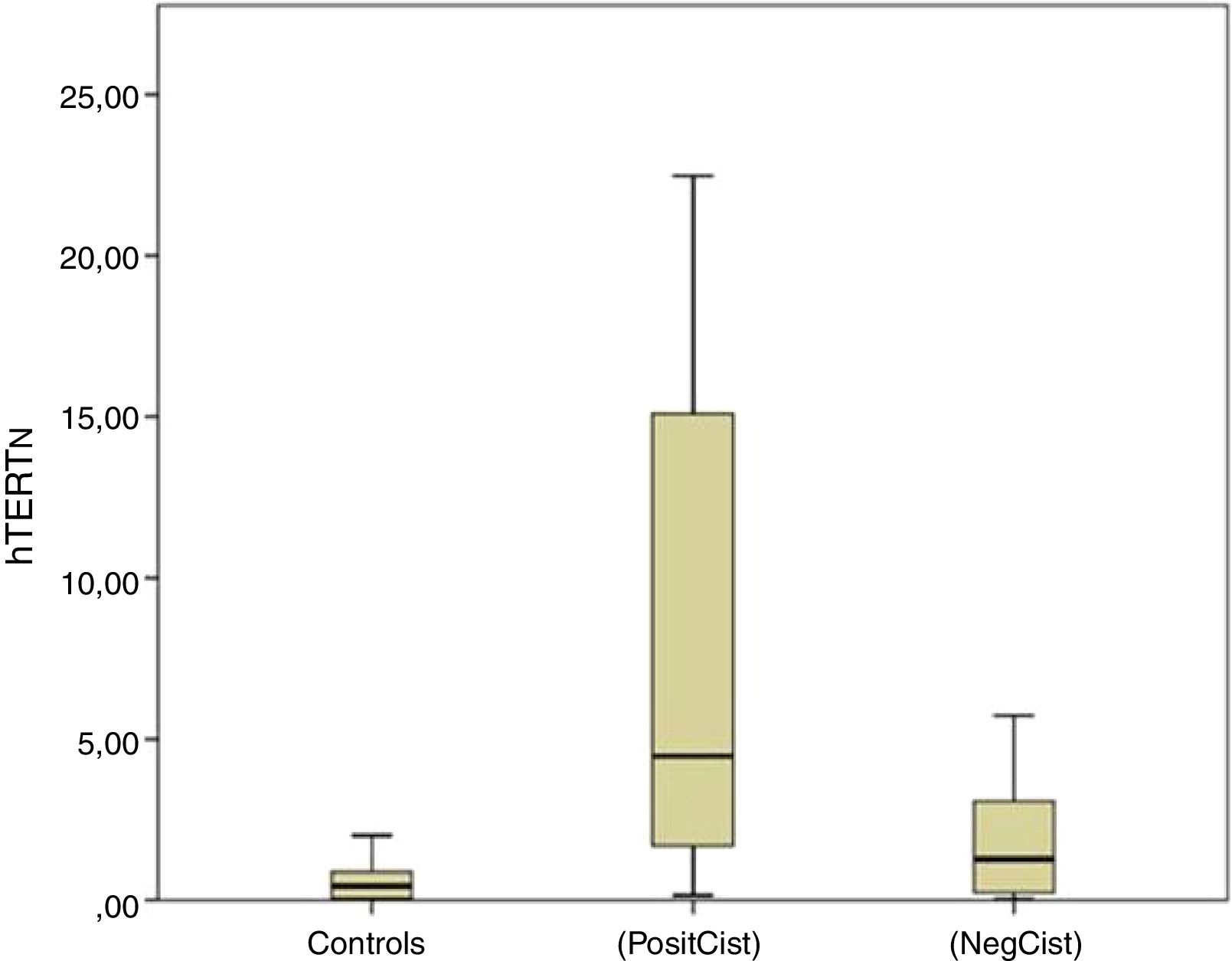
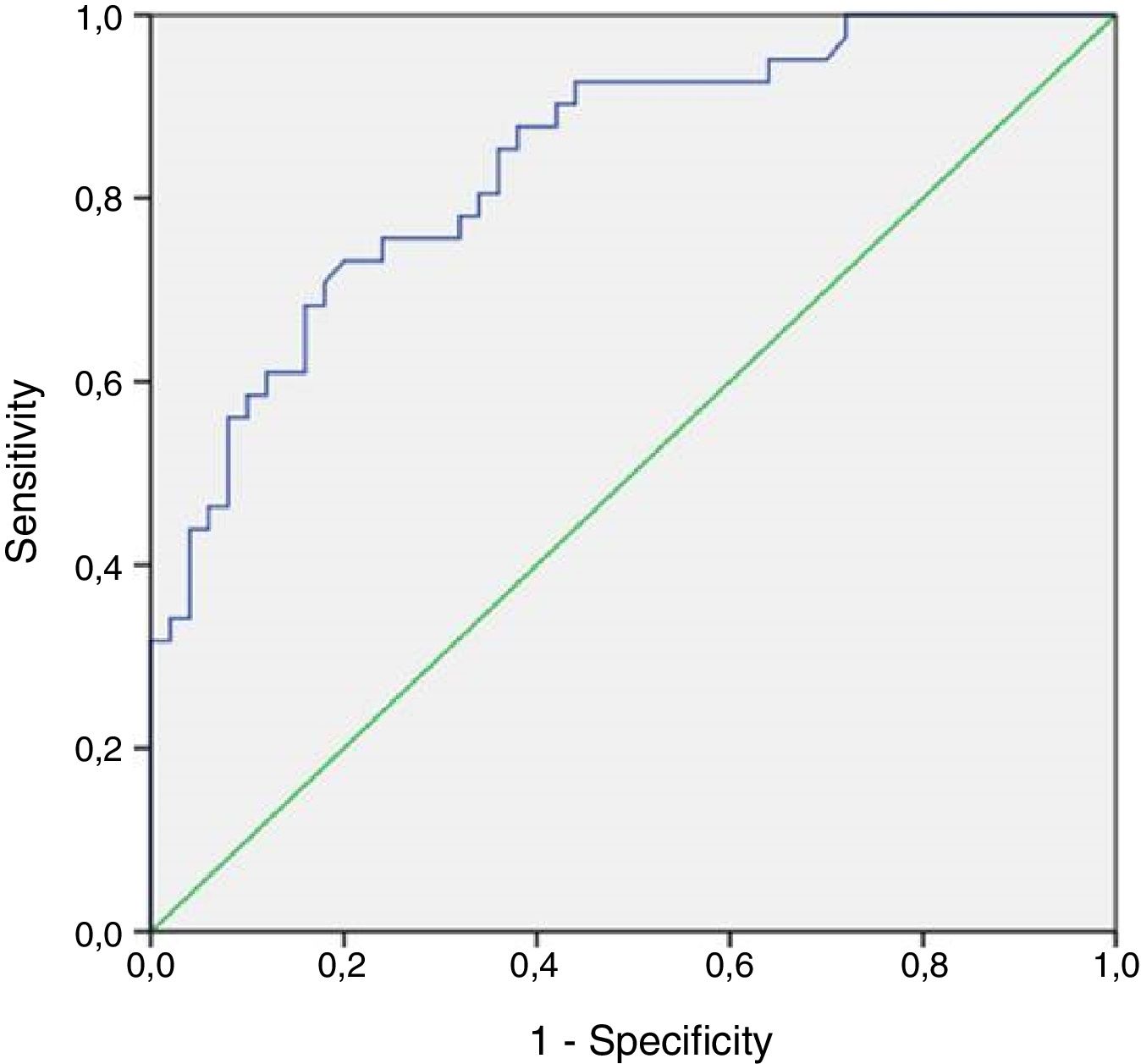
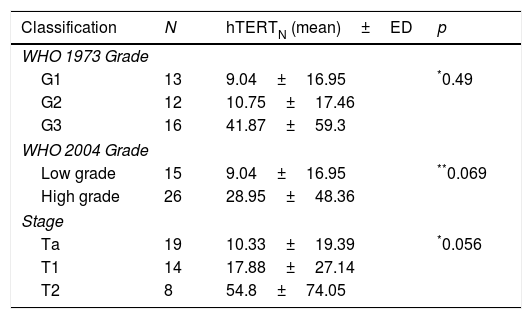
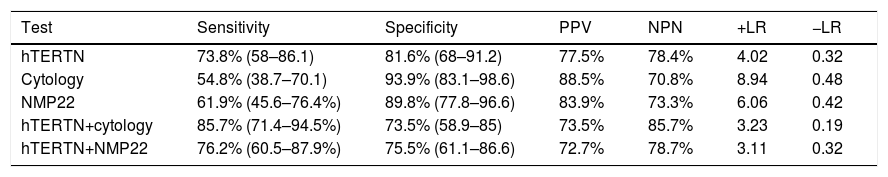
ResultsDifferences were observed in mean hTERTN levels in each of the groups: tumor presence 21.33±40.66, tumor history 2.16±2.67, controls 0.9±1.75 (p<0.001). In patients with tumor, there was no difference in mean hTERTN levels between the different grades and stages, although there was a tendency: low grade tumor 9.04±16.95, high grade 28.95±48.36 (p=0.069), stage Ta 10.33±19.39, T1 17.88±27.14, T2 54.8±74.05 (p=0.056). In addition, the sensitivity of hTERTN was superior to that of other test (76%), although specificity and positive and negative predictive values were better for cytology (94%, 88.4% and 72.3% respectively) and NMP22 (88%, 80.6% and 73.3% respectively).
ConclusionshTERTN mRNA levels in urine were higher in patients with bladder tumors compared to patients with a history of bladder tumor and with negative cystoscopy, as well as in the control group. This determination showed a higher diagnostic yield compared with the detection of NMP22 and urinary cytology.
El objetivo fue estudiar la relación entre la determinación cuantitativa de ARNm (hTERT) en pacientes con tumor vesical, antecedentes de tumor vesical y en sujetos sin antecedentes de esta neoplasia.
Material y métodosSe trata de un estudio prospectivo, aleatorizado y controlado con 91 sujetos incluidos. El valor de ARNm-hTERTN se determinó en 63 pacientes con antecedentes o sospecha de tumor vesical y en 28 controles. Se enviaron muestras de orina para evaluar el nivel de ARNm (hTERT), el estudio citológico y el resultado de NMP22.
ResultadosSe observaron diferencias en los niveles medios de hTERTN en cada uno de los grupos: presencia de tumor 21,33+/- 40,66, antecedente del tumor 2,16+/- 2,67, controles 0,9+/- 1, 75 (p<0,001). En pacientes con tumor, no hubo diferencias en los niveles medios de hTERTN entre los diferentes grados y estadios, aunque hubo una tendencia: tumor de bajo grado 9,04+/- 16,95, grado alto 28,95+/- 48,36 (p=0,069), estadio Ta 10,33+/- 19,39, T1 17,88+/- 27,14, T2 54,8+/- 74,05 (p=0,056). Además, la sensibilidad de hTERTN fue superior a la de otras pruebas (76%), aunque la especificidad y los valores predictivos positivos y negativos fueron mejores para la citología (94%, 88,4% y 72,3% respectivamente) y NMP22 (88%, 80,6% y 73,3% respectivamente).
ConclusionesLos niveles de mRNA de hTERTN en la orina fueron más altos en pacientes con tumores vesicales en comparación con pacientes con antecedentes de tumor de vejiga y con cistoscopia negativa, así como en el grupo de control. Esta determinación mostró un mayor rendimiento diagnóstico en comparación con la detección de NMP22 y citología urinaria.