Changes in blood viscoelastic properties have been proposed previosuly as etiopathogenesis for severe complications in COVID-19 and some cases of Sudden Deafness (SD). This is an attempt to verify if SD cases in patients admitted for SARS-Cov-2 infection can be correlated.
Patients and methodsA prospective follow-up was carried out with COVID-19 patients, monitoring their blood viscosity (BV) at high shear rate (300 s−1) and inquiring them periodically for eventual hearing loss. This measurement was extended to cases bearing of SD in 2019 and 2020 without infection and a control group of healthy normoacoustic subjects.
ResultsThe normality range was 4,16 ± 0,62 cps. 330 cases admitted for COVID-19 were evaluated from February 24th, 2020 to March 24th, 2021, 85 of them attended in ICU. After anamnesis and Audiometric Tone Thresholds developed as soon as possible, 9 SD were detected, all belonging to ICU group. The mean BV was 4,38 ± 0,43 cps in the ward group, 4,53 ± 0,39 cps in the ICU patients without SD, and 4,85 ± 0,52 cps in the cases with SD, with statistically significant differences. Highest BV elevations in the SD cases were detected between days 6 and 10 of hospital admission. In 2019 four cases consulted with SD, and another two did it in 2020 without a diagnosis of COVID-19, with normal BV values.
ConclusionsDuring SARS-Cov-2 infection, patients may show high BV and SS, although an inpatients control group and a larger sample volume are necessary to confirm the predisposition to hyperviscosity. The incidence of hearing damage is considerable if its possible appearance is taken into account, within the limitations of critical patients with COVID-19.
La alteración en las propiedades viscoelásticas de la sangre ha sido anteriormente propuesta como etiopatogenia de complicaciones severas por COVID-19 y algunos casos de Sordera Súbita (SS). Pretendemos verificar si la aparición de casos de SSI en pacientes ingresados por infección por SARS-Cov-2 puede correlacionarse de este modo.
Pacientes y metodosEstudio longitudinal prospectivo de pacientes ingresados en nuestro Centro por COVID-19, efectuando monitorización de la viscosidad sanguínea (VS) a alta velocidad de cizallamiento (300 seg−1) y un requerimiento periódico personal sobre hipoacusia. Esta determinación se extendió a casos atendidos por SS sin infección en 2019 y 2020 y un grupo control de sujetos sanos normoacúsicos.
ResultadosEl rango de normalidad se situó en 4,16 ± 0,62 cps. Entre el 24 de Febrero de 2020 y el 24 de Marzo de 2021 se evaluaron 330 casos ingresados por COVID-19, 85 asistidos en UCI. Fueron identificadas tras anamnesis y ATL todo lo inmediata que el status clínico lo permitió hasta 9 casos con SS, todos pertenecientes al grupo de UCI. La VS media fue 4,38 ± 0,43 cps en el grupo de sala, 4,53 ± 0,39 cps en los pacientes en UCI sin SS, y 4,85 ± 0,52 cps en los casos con SS, con diferencias estadísticamente significativas. Las mayores elevaciones de la VS en los casos con SS se detectaron entre los días 6 y 10 del ingreso hospitalario. En 2019 consultaron 4 casos, y otros 2 en 2020 sin diagnóstico de COVID-19, con valores normales de VS.
ConclusionesDurante la infección por SARS-Cov-2 los pacientes pueden presentar elevación en la VS y SS, si bien se hacen necesarios un grupo control hospitalario y un volumen muestral mayor para confirmar la predisposición a la hiperviscosidad. La incidencia del daño auditivo resulta considerable si se tiene en cuenta su posible aparición en pacientes críticos con COVID-19.
SARS-CoV-2 is a new type of coronavirus transmissible between humans and first detected in December 2019 in Wuhan City, Hubei Province, China. The disease it causes is called COVID-19 and its pandemic spread resulted in more than 78 million cases and 1.47 million deaths worldwide and more than 1.85 million cases and 49,698 officially recognised deaths in Spain at the end of December 2020, just before the start of the global vaccination programme. By the end of March 2021, 3.26 million cases were reported in Spain with 5.29% of the population adequately vaccinated.
In 80% of cases it is subclinical and in 80% of symptomatic cases it produces only mild respiratory manifestations, but in its diagnosis and treatment there is evidence of increasing morbidity and mortality in relation to cardiovascular and neurological complications, mainly ischaemic.1 Of the latter, otological complications have become increasingly important, including sudden deafness (SD).
SD may be considered a syndrome due to the numerous clinical circumstances that can lead to it, as well as its varied therapeutic options. Despite its etiopathogenic diversity, the detection of cases in the COVID-19 setting suggests a relationship with the inflammatory reaction, triggering significant variations in blood flow.2 Blood viscosity (BV) is the resistance of blood to flow through vessels and its increase can lead to a condition known as hyperviscosity syndrome (HVS), dependent on both the cellular and plasma components of blood. It is considered a diagnostic and therapeutic emergency because it is potentially lethal.
Alterations in BV have been previously documented in SD, which would correlate well with the depth and prognosis of hearing loss.3 This peculiarity brings together causal theories such as viral, vascular, inflammatory or autoimmune, as the result of all of them often results in leukocytosis, thrombocytosis, hyperproteinemia and elevated acute phase reactants.1,4 These laboratory findings are often present in SD and COVID-19, especially in severe cases.
The signs and symptoms of HVS are linked to circulatory disturbances caused by increased resistance to blood flow and overlap with the manifestations of the underlying disease causing it. Classically characterised by a triad of ophthalmological, neurological and coagulopathic changes, HVS can also be multisystemic with a range of clinical expression from mere influenza to multi-organ failure.1,4
The interest of the present study lies in the prospective nature of the review carried out in cases admitted to our centre in which BV was monitored, regardless of the severity of the clinical picture, maintaining alertness in the detection of SDS during their hospital stay.
The aim of the present study was to assess possible abnormalities in the viscoelastic properties of detectable blood that debut with SD during infection with SARS-CoV-2. This would make it possible to aetiologically relate this hearing disorder to HVS, to identify COVID-19 patients with a higher risk of developing SD and, finally, to verify whether its incidence is higher than that usually described, between five and 27 cases/100,000 inhabitants/year.5,6
Patients and MethodsThis work is part of a multidisciplinary follow-up of the largest possible volume of patients diagnosed with COVID-19 and admitted to our Centre – with healthcare coverage for 66,000 inhabitants – since the beginning of the pandemic. Epidemiological information and conventional and research clinical and laboratory findings have been and continue to be collected from this group. A specific database was created with a numerical code assigned to each case in accordance with data confidentiality regulations, once approval had been obtained from the Centre's Clinical Research Ethics Committee.
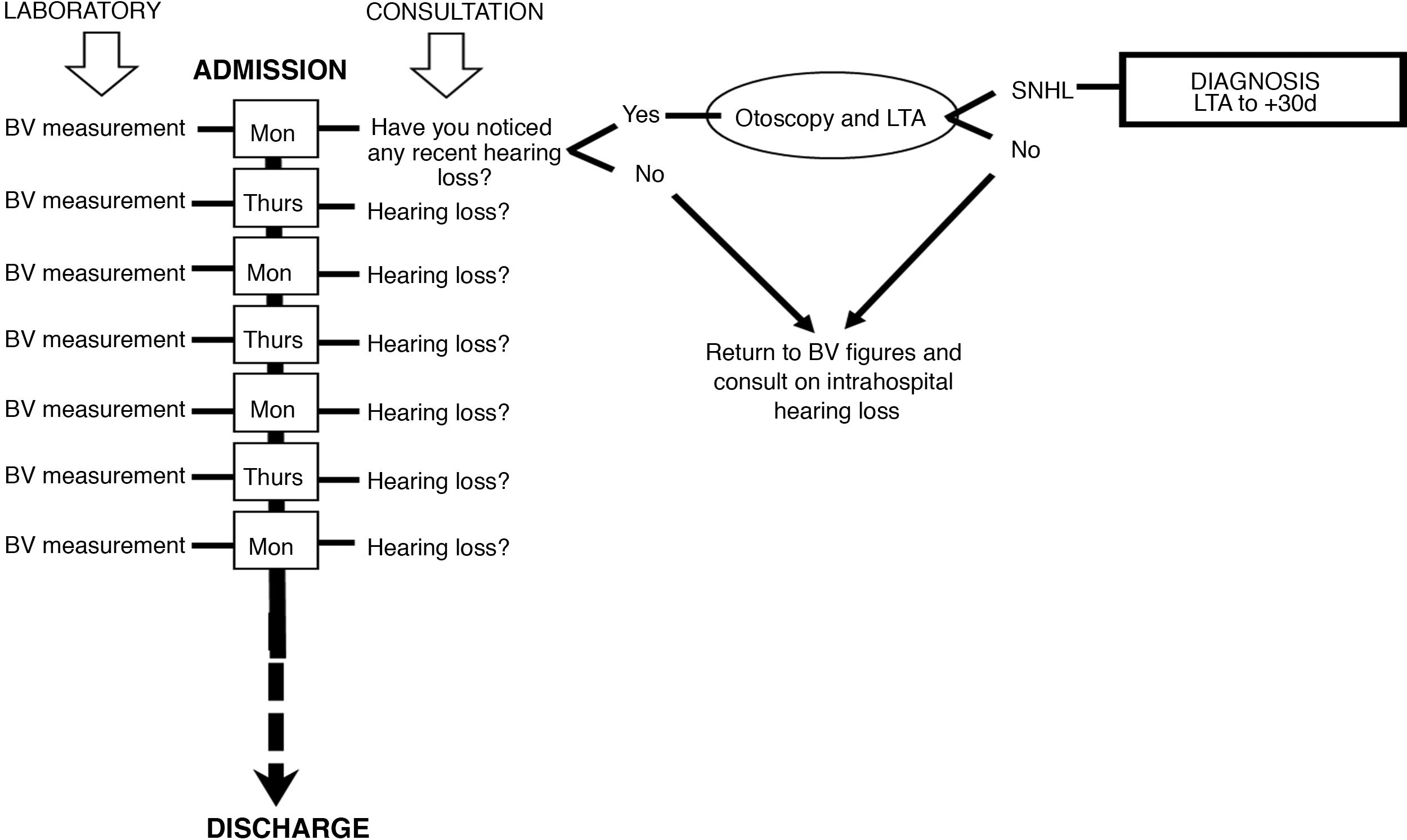
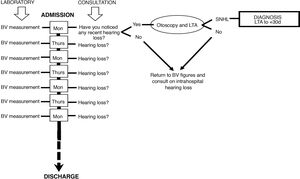
SubjectsIn a longitudinal, prospective follow-up, all patients were included who, since the onset of SARS-CoV-2 infection, gave signed informed consent (themselves or their guardians, relatives or legal representatives) for monitoring of the BV profile during their hospital stay. Thus, between 24th February 2020 and 24th March 2021, 330 of the 412 admissions were included in the study. All of them were asked twice a week if they noticed hearing loss or difficulty in understanding whenever they were receptive to a conversation, according to the flow chart illustrated in Fig. 1. Those who answered affirmatively were assessed by Tone Liminal Audiometry (LTA) when their clinical situation allowed it and again 30 days later. SD was considered as perceptual hearing impairment with losses of 30 dB HL or more in three or more consecutive frequencies of the auditory spectrum in less than 72 h. Hearing recovery was based on the criteria of the Working Group of the Audiology Commission of the Spanish Society of Otorhinolaryngology (SEORL for its initials in Spanish)7 shown in Table 1.
Flowchart of action in COVID-19 admissions. Every Monday and Thursday of the hospital stay, blood viscosity (BV) was determined and questioning on the perception of hypoacusis was carried out. When there was an impression of hearing impairment, otoscopy and Liminal Tone Audiometry (LTA) were performed to assess the existence of sudden onset sensorineural hearing loss (SNHL).
2019 SEORL Criteria on Hearing Improvement in SD, Considering the Degree of Recovery by the Average Hearing Threshold From 250 to 8000 Hz in the Second LTA Within 30 Days of the First LTA.
| Recovery Concept | Degree or Recovery |
|---|---|
| Complete | Auditory threshold at final LTA less than or equal to 10 dB HL of the previous threshold and a Verbal Reception Threshold at most 5%–10% worse than the unaffected ear. |
| Partial | Improvement greater than 10 dB HL at final LTA or greater than 10% of the VRTof the unaffected ear, without reaching full recovery. |
| Mild | Improvement of less than 10 dB HL in final LTA. |
LTA: liminal tonal audiometry; SD: sudden deafness; SEORL: Spanish Society of Otorhinolaringology; VRT: verbal reception threshold.
The clinical-epidemiological characteristics of the subjects assessed included diagnosis of acute COVID-19 infection, associated underlying pathology, number of maintenance treatments, length of stay and hospital location, regardless of the reason for admission. This resulted in two groups of individuals studied:
- •
Those admitted to hospital with a diagnosis of SARS-CoV-2 infection in the Intensive Care Unit (ICU)
- •
Those admitted to hospital with a diagnosis of SARS-CoV-2 infection in the ward.
Data on infected patients were processed for individualised reporting of COVID-19 cases to the National Network of Epidemiological Surveillance through the SiViEs computer application, counting the cases reported according to the alert strategy in force at the time, which until 10th May 2020 included confirmed and probable cases, detected by any of the available laboratory techniques (polymerase chain reaction [PCR], ELISA serological test, rapid antibody test or antigen test). As of 11th May, the Ministry of Health made it compulsory to count confirmed cases diagnosed by PCR and in some specific cases those diagnosed by IgM by ELISA, based on individualised records communicated by the Autonomous Community through SiViEs, in accordance with the Strategy for Diagnosis, Surveillance and Control in the Transitional Phase of the COVID-19 Pandemic.
Additionally, patients who in 2019 and in the study period consulted for SD with no other reason for attendance and who also underwent diagnostic testing for SARS-CoV-2 were controlled.
Finally, a BV determination was performed on a control group of 99 healthy individuals in the time period between 1st June 2019 and 1st March 2020, the results of which allowed a range of normality to be established.
In patients with SD on admission, no specific therapies were used as they had been treated at the Centre with intravenous corticosteroids at doses of no less than 1 mg/kg/day of methylprednisolone and low-molecular-weight heparin. In the absence of response, a posteriori intratympanic corticosteroid therapy (ITC) was proposed as a protocol of a weekly dose of .9 cc of a 40 mg vial of methylprednisolone, for three consecutive weeks.
The inclusion criteria required for the study, in addition to informed and signed consent in accordance with the specifications of the Ethics Committee of our Centre, were the documentation of acute or past SARS-CoV-2 infection status, their BV profile and the performance of the LTA examinations.
ViscosimetryWells-Brookfield cone-plate rotational viscometers with temperature control system from 0 to 100C model DV3TLVCP and angled cone spindles 4 model CPA-40Z were used for the determination of BV for basal shear rate (SR) measurements of 7.5 s−1. The rotation of the conical vessel at a predetermined speed generates a rotational movement in 5 cc of the blood sample under study anticoagulated with ethylenediaminetetraacetic acid (EDTA). The viscometer bases its measurement on the force required to rotate the spindle inside the sample at a predetermined and constant rotational speed of 300 s−1, which is achieved by amplifying the rotation of the cone to 40 rpm. The cones are removable for cleaning and sterilisation. The unit of measurement for BV was the centipoise (cps), which is equivalent to 1 milliPascal per second (mPa s).
In this way, a normal range of BV at 300 s−1 was calculated with the control group. Between cases, BV measurements were systematically taken on Mondays and Thursdays of admission, thus providing a chronological profile of the evolution of the variable throughout the hospital stay in the ward and/or ICU. In the subjects attended for SD and who did not present infection by the virus, BV was also determined at the time of diagnosis and 30 days after diagnosis.
Statistical AnalysisFor the comparison of groups and cohorts, the technique of factorial analysis of variance (ANOVA-test) was used to study the effect of one or more factors. This was processed in the formulation offered by Microsoft Office Excel 2013 and the statistical package SPSS 26.0. The main variable to be compared was BV monitored over time.
When the comparison of independent data was made between different individuals belonging to two population groups, the Student's t-test was used, with comparison of means and standard deviations. The quantitative variable had to be normally distributed in each of the cohorts, verified by the Kolmogorov-Smirnov test with groups having more than 50 events and with the variances within the groups approximately equal in all of them.
With P values greater than .001, insufficient evidence was assumed to consider that at least two means are different, implying that a small effect size would have been observed in the study and the inference techniques did not find statistical significance to reject. Statistical significance was thus considered when P < .001.
ResultsDuring the 13 months of the study, 330 patients admitted with a diagnosis of SARS-CoV-2 infection were followed up. Of these, 245 only required in-patient ward care, 69 others started in-patient care but required transfer to the ICU and up to 16 cases were admitted directly to the ICU. In an attempt at simplification, the latter two groups were considered as ICU group (n = 85). The annual prevalence of infection in our Health Department was 5.59%.
During this time, 33 patients responded affirmatively to the impression of uni- or bilateral hearing loss. In nine of them, seromucous otitis was found with LTA confirming conductive hearing loss, in five patients LTA did not detect a hearing deficit and in another 10 it was not possible to perform an audiometric examination. In the remaining nine patients who were consulted, LTA confirmed a unilateral or asymmetrical sensorineural hearing loss congruent with SD, considering the contralateral ear as the reference threshold on which to compare. All of them had been seen in ICU (2.2% of all patients admitted with COVID-19 and 10.5% of those seen in ICU). They were six men and three women, aged between 42 and 77 years. Of these, three cases were associated with tinnitus in the affected ear and one had an instability crisis referred to as a spinning sensation. Two other male individuals aged 49 and 59 years consulted for SD without COVID-19 while these results were collected in 1266 hospital visits on 1006 patients. In 2019, four cases had been diagnosed with SD in 3966 visits performed on 3691 patients.
The group of healthy controls (n = 99), aged between 25 and 66 years and with a male/female ratio of 1.25, hearing impaired, showed a mean BV of 4.16 ± .62 cps at a SR of 300 s−1, for a normal distribution of the parameter with a confidence interval of .95.
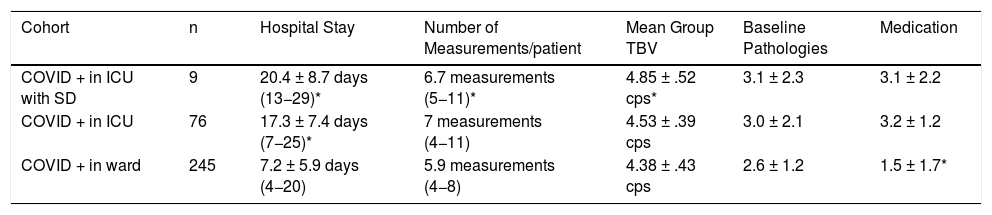
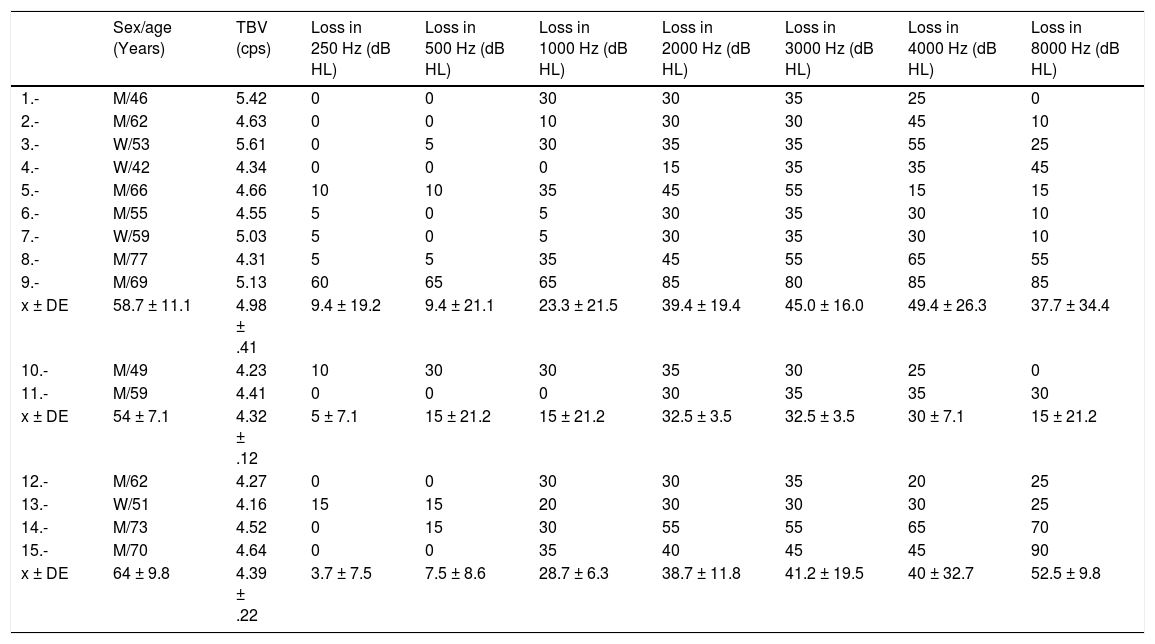
Table 2 shows the hospital circumstances in the cohorts considered. The group with the nine SD cases showed a significantly higher mean BV (4.85 ± .52 cps) compared to COVID-19 patients in ICU but without SD (n = 76; 4.53 ± .39 cps, P < .001) and to COVID-19 patients on the ward (n = 245; 4.38 ± .43 cps, P < .001). In addition, SD cases had a longer mean hospital stay than those admitted to the ward. Although the mean volume of associated pathologies was similar in the three groups, patients admitted to the ward took significantly fewer medications on average than ICU subjects, whether or not they had SD. Table 3 shows the thresholds detected in the patients who showed SD, as well as the average value of their BV during their hospital stay, not only during the study but also in the previous year. This hearing loss was always unilateral, with a total incidence of the disease in 2019 of 6/100,000 population-years (1 case per 1000 patients seen in our Service) and in the study period of 16.6/100,000 population-years (10 per 1000 patients seen).
Characteristics of the Patients Hospitalised for COVID-19 and Grouped Into the 3 Cohorts Considered (*P < .001).
| Cohort | n | Hospital Stay | Number of Measurements/patient | Mean Group TBV | Baseline Pathologies | Medication |
|---|---|---|---|---|---|---|
| COVID + in ICU with SD | 9 | 20.4 ± 8.7 days (13−29)* | 6.7 measurements (5−11)* | 4.85 ± .52 cps* | 3.1 ± 2.3 | 3.1 ± 2.2 |
| COVID + in ICU | 76 | 17.3 ± 7.4 days (7−25)* | 7 measurements (4−11) | 4.53 ± .39 cps | 3.0 ± 2.1 | 3.2 ± 1.2 |
| COVID + in ward | 245 | 7.2 ± 5.9 days (4−20) | 5.9 measurements (4−8) | 4.38 ± .43 cps | 2.6 ± 1.2 | 1.5 ± 1.7* |
ICU: Intensive Care Unit.
Threshold Drop in the First LTA Broken Down by Frequency and Mean Value of TBV at SR at 300 s−1 for Each Case With SD. Cases 1–9 Are Admissions With COVID-19. Cases 10 and 11 Are Uninfected Outpatients in 2020. Cases 12–15 Are Cases Detected in 2019.
| Sex/age (Years) | TBV (cps) | Loss in 250 Hz (dB HL) | Loss in 500 Hz (dB HL) | Loss in 1000 Hz (dB HL) | Loss in 2000 Hz (dB HL) | Loss in 3000 Hz (dB HL) | Loss in 4000 Hz (dB HL) | Loss in 8000 Hz (dB HL) | |
|---|---|---|---|---|---|---|---|---|---|
| 1.- | M/46 | 5.42 | 0 | 0 | 30 | 30 | 35 | 25 | 0 |
| 2.- | M/62 | 4.63 | 0 | 0 | 10 | 30 | 30 | 45 | 10 |
| 3.- | W/53 | 5.61 | 0 | 5 | 30 | 35 | 35 | 55 | 25 |
| 4.- | W/42 | 4.34 | 0 | 0 | 0 | 15 | 35 | 35 | 45 |
| 5.- | M/66 | 4.66 | 10 | 10 | 35 | 45 | 55 | 15 | 15 |
| 6.- | M/55 | 4.55 | 5 | 0 | 5 | 30 | 35 | 30 | 10 |
| 7.- | W/59 | 5.03 | 5 | 0 | 5 | 30 | 35 | 30 | 10 |
| 8.- | M/77 | 4.31 | 5 | 5 | 35 | 45 | 55 | 65 | 55 |
| 9.- | M/69 | 5.13 | 60 | 65 | 65 | 85 | 80 | 85 | 85 |
| x ± DE | 58.7 ± 11.1 | 4.98 ± .41 | 9.4 ± 19.2 | 9.4 ± 21.1 | 23.3 ± 21.5 | 39.4 ± 19.4 | 45.0 ± 16.0 | 49.4 ± 26.3 | 37.7 ± 34.4 |
| 10.- | M/49 | 4.23 | 10 | 30 | 30 | 35 | 30 | 25 | 0 |
| 11.- | M/59 | 4.41 | 0 | 0 | 0 | 30 | 35 | 35 | 30 |
| x ± DE | 54 ± 7.1 | 4.32 ± .12 | 5 ± 7.1 | 15 ± 21.2 | 15 ± 21.2 | 32.5 ± 3.5 | 32.5 ± 3.5 | 30 ± 7.1 | 15 ± 21.2 |
| 12.- | M/62 | 4.27 | 0 | 0 | 30 | 30 | 35 | 20 | 25 |
| 13.- | W/51 | 4.16 | 15 | 15 | 20 | 30 | 30 | 30 | 25 |
| 14.- | M/73 | 4.52 | 0 | 15 | 30 | 55 | 55 | 65 | 70 |
| 15.- | M/70 | 4.64 | 0 | 0 | 35 | 40 | 45 | 45 | 90 |
| x ± DE | 64 ± 9.8 | 4.39 ± .22 | 3.7 ± 7.5 | 7.5 ± 8.6 | 28.7 ± 6.3 | 38.7 ± 11.8 | 41.2 ± 19.5 | 40 ± 32.7 | 52.5 ± 9.8 |
LTA: liminal tonal audiometry; M: man; SD: sudden deafness; SR: shear rate; TBV: total blood viscosity; W: woman.
According to the criteria for hearing recovery7 of the nine cases with COVID-19, three showed a complete improvement, three showed a partial improvement and three showed no improvement at all – despite the administration in three of them of ITC courses – and it can be concluded that in two thirds of the cases there has been no reversal of the disease. In none of the three cases with tinnitus, the tinnitus has subsided. The balance disorder in one of the patients disappeared within 30 days.
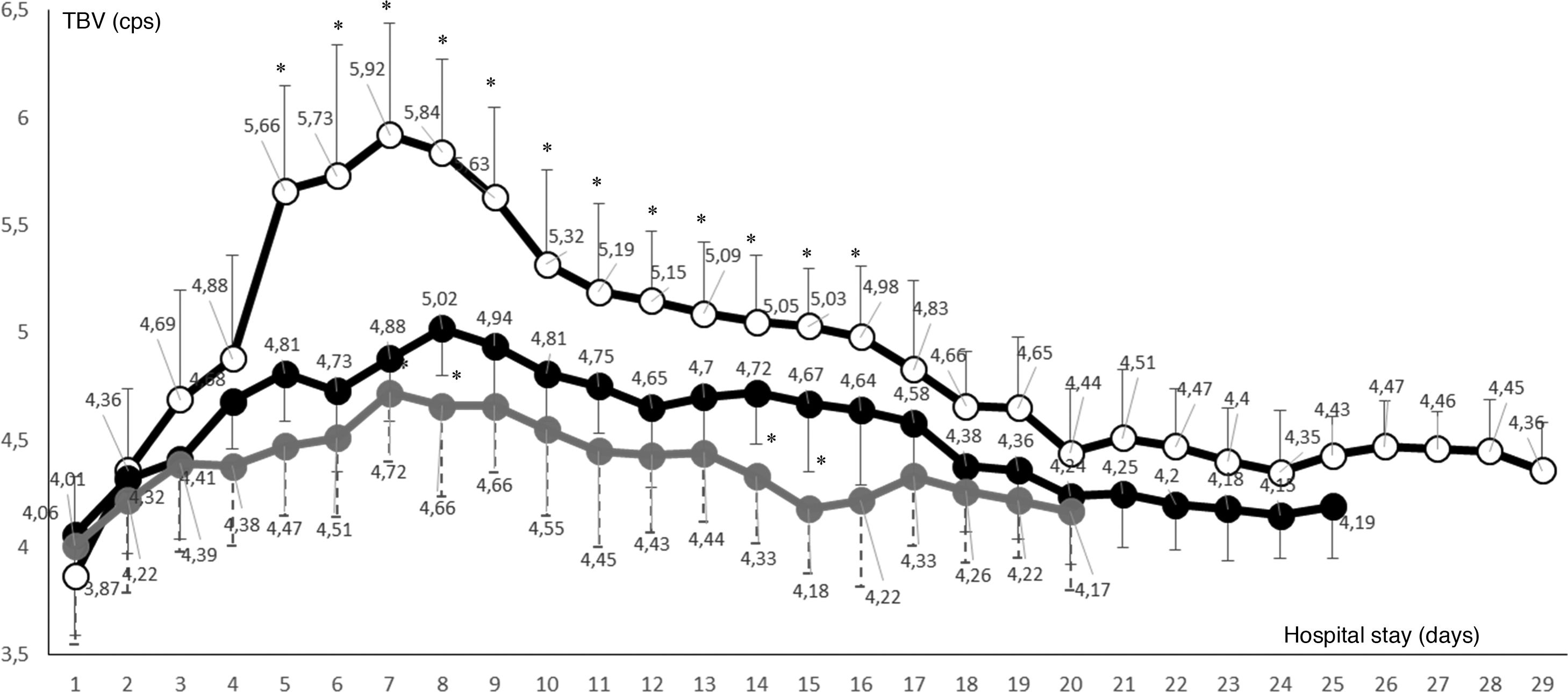
Fig. 2 represents the temporal variability in the mean BV of all cases with SARS-CoV-2 infection, considered by cohorts according to whether their hospital stay was in the ward or in the ICU. It can be seen to be elevated among the cases in which SD was detected, specifically from day 5–16, with statistically significant differences compared to the group of patients with COVID-19 admitted to ICU, but without hearing impairment, as well as compared to patients on the ward. In cases with SD, the determination of their BV was always higher than the upper value of the normal range, with a maximum peak on day seven of admission (n = 9; 5.92 ± .52 cps).
Daily variation in mean blood viscosity of all admissions with SARS-CoV-2 infection, considered by cohorts ((•ward, •ICU without sudden deafness, oICU with sudden deafness). All three groups showed blood viscosity values higher than the normal range obtained. The COVID group in ICU with sudden deafness showed a significantly higher mean blood viscosity than patients in ICU without sudden deafness during days 5 to 16 of their stay (*P < .001).
In the group of patients admitted to ICU without hearing damage a less pronounced elevation was seen, with a maximum peak on day eight (n = 76; 5.02 ± .22 cps), the only day where the difference in this value was statistically significant, although between days four and 16 the BV figures also exceeded the normal range. Among COVID-19 patients admitted to the ward (n = 245) BV was also above normal, but below the two ICU subgroups.
DiscussionThe most commonly accepted aetiologies of SD are viral, autoimmune and vascular. The pathogenesis of all of these would include processes of increased BV documented in tumour, inflammatory and circulatory disorders, often leading to single or multiple organ failure. Local endovascular hyperviscosity is likely to generate dysfunction in the endothelium and the three blood cell lines and thereby secondarily activate coagulation mechanisms that promote the phenomenon of microthrombosis.8 Where an organ depends exclusively on a monocariant circulation – such as the labyrinth – its overriding may result in rapid and potentially irreversible deterioration of its function.9
Blood is a non-Newtonian fluid and varies in viscosity depending on its composition and flow velocity. This is the resultant of the ratio between the force acting parallel to the vessel surface at each flow unit or shear rate (SR) and the difference in velocities between two adjacent layers of this flow or SR. In turn, SR is related to the flow velocity and the internal radius of the vessels according to the formula SR = 4.v/r, and has a value between 101.1 and 102.2 s−1 in arterioles and 102.2 and 102.5 s−1 in capilliaries.10 Thus, with small SR the forces acting on red blood cells are also reduced and they aggregate in rouleaux, greatly enhanced by circulating fibrinogen, so that BV rises.2 If the SR is elevated, the rouleaux are disaggregated and the red cells are deformed from discoid to oval with a major axis in the longitudinal direction of the blood stream, which provides minimal resistance to flow. Under physiological conditions the SR of the blood is several hundred second−1 throughout most of the vascular tree, so the HR is high enough to disaggregate red cells and bring them to a state of maximal deformation that minimises viscosity and optimises cardiac work. BV rises in slow flows, promoting red cell aggregability, and decreases in fast flows because of its deformability.11
The labyrinthine circulation is terminal and depends on an arteriole whose regulation is not a function of cerebral perfusion, but its flow can be influenced by variations in BV if some of the factors affecting it are altered, such as haematocrit, erythrocyte deformability and aggregability, platelet aggregability, leukostasis, hyperfibrinogenemia and hyperlipaemias.11,12 BV at high RH attempts to emulate inner ear perfusion conditions and is documented to be elevated in individuals who have had SD by reducing blood filterability and tissue oxygenation. The values obtained for the control group in our work are similar to those detected in 1997 working with less sophisticated viscometers, showing values of approximately 4.2 cps for high SR4. It is accepted that plasma viscosity, not studied in this work, does not exceed 2 cps and is not influenced by variations in RS.
Blood hyperviscosity has previously been observed in presbycusis, rapidly progressive sensorineural hearing loss and SD. In the latter, correlations could also be established between reduced erythrocyte deformability and blood filterability indices and high fibrinogen levels with greater hearing loss and lower recovery capacity,9,11,13 and specific actions have been proposed as part of the treatment of SD, including haemoreactive agents, normovolaemic haemodilution and lipid and fibrinogen apheresis.4,14–16
Among the various pathways in which SARS-CoV-2 has been found to cause tissue damage is the deoxygenation capacity of red blood cells by deconstructing haemoglobin chains and increasing the rigidity of the lipid bilayer of their membrane.7 Moreover, the presence of angiotensin-converting enzyme-2 (ACE-2) in arteriolar smooth muscle causes it to lose elasticity in the event of infection, promoting junctions between red blood cells and fibrinogen and the latter with the whole endothelium, even before platelets act locally and enhance microvascular stasis.1,18 All this facilitates erythrocyte aggregability and elevates BV in small vessels, such as the internal auditory artery, leading to activation of haemostasis much earlier than coagulation. Other mechanisms such as neurotropism of the virus and its access via cerebrospinal fluid (CSF) to the VIII pair and ototoxicity of conventional therapies in the infection have also been proposed.19. The middle ear involvement detected in our study has also been documented by Fidan,20 but lacks the clinical significance of the present case.
In addition to pulmonary damage, COVID-19 has been characterised by a particular neurological involvement of a thromboembolic nature in patients with an unfavourable evolution, with a good correlation with fibrinogen and D-dimer levels,1,16 which reaches 36% of those admitted for the infection.21 Our study detected a significant increase in BV in patients admitted to the ICU and therefore clinically more compromised, characteristically from the sixth day of admission. This is the period of maximum release of cytokines IL-1, IL-2, IL-4, IL-7, IL-10, IL-12, IL-13, IL-17, macrophage inflammatory protein-1, interferon, tumour necrosis factor, hepatocyte growth factor and granulocyte and macrophage colony stimulators in the absence of adequate immune control upon entry of SARS-CoV-2.22 The IL-6 interceptor and IL-2 receptor promote recruitment of blood cell lines and elevation of acute phase reactants. This predisposes to endothelial damage and especially to an increase in HR and thus BV.23 The result is loss of glycocalyx in the endothelial wall, inhibition of the plasminogen system from the renin-angiotensin pathway and finally the development of VHS. This disorder, described by Ahmed et al. with turbulence in blood flow, turns out to be the basis of thrombogenesis in VHS infection.24
Hypertensives, diabetics and dyslipidaemics are more predisposed to HVS. They are also risk groups and a poor prognostic factor in severe cases of COVID-19, where the most significant laboratory findings include elevated fibrinogen, cytokines, circulating immunocomplexes and platelets.6,25 These modifications have previously been detected in blood and implicated in the aetiopathogenesis of SD. It would be reasonable to assume that the same findings among coronavirus-infected individuals could lead to the development of hearing impairment.
The documentation of SD cases in the context of COVID-19 is not exceptional. These are publications with isolated cases or very limited series that assessed patients with hearing impairment with and without infection, almost all of them summarised in the work of Saniasiaya.17 The first case described appears to be documented by Sriwijitalai et al. in Thailand in March, with no clinical or therapeutic specifications.25 Interestingly, in the first paper with a significant population volume, Chari et al. compared SD records on 75 consecutive days between March and May 2019 and 2020, reporting picture incidences of 1.77 BV. 1.91%, in a healthcare coverage area with 592, 853 individuals in Massachusetts and a prevalence of SARS-CoV-2 infection of 16.4%, suggesting a lack of SARS-CoV-2 involvement in the disorder, although they also weighed patient-dependent bias associated with fear of disease at consultation.26
Harenberg et al. however, take the opposite view, finding a higher risk of SD among patients with COVID-19, which they attribute to neurotropism due to the neuronal presence of ACE2 receptors, concomitant endothelitis and platelet aggregability.27 Maier et al. describe 15 patients attended in ICU for severe pneumonia and treated with anticoagulants for presenting mean fibrinogen values of 708 mg/dL and plasma viscosity always above its normal range between 1.4 and 1.8 cps, also verifying four thrombotic events with this parameter at 3.5 cps8 Similarly, Venter et al. detected elevations in plasma fibrinogen and viscosity of up to 1000 mg/dL and 2.8 cps, respectively, in their series of 29 cases with severe vascular complications.4 This may establish a link between inflammation, hyperviscosity and coagulopathy in critically ill patients with COVID-19 and recommend the identification of cellular or plasma components as a measure of disease severity.
Our study prospectively detects significant changes in BV as a potential cause of hearing impairment, being the first to relate viscoelasticity to SD in the COVID-19 setting. From this point of view, we agree with Hildesheimer et al. that the normalisation of this factor may become an obligatory goal of treatment aimed at hearing recovery.12 At least it seems logical to think that knowledge of these parameters is useful in the choice of treatment, the clarification of aetiology and the establishment of a prognosis. We insist on the need to ask the admitted patient as soon as possible about hearing impairment.
Today, this therapeutic offer already includes systemic corticotherapy according to the specifications of the Rapid Evidence Appraisal for COVID-19 Therapies (REACT) group of the World Health Organisation (WHO), which may make it possible to prevent the hearing disorder and reduce it to an incidence still unknown to us in this context of a new disease.28 The inherent infectious capacity of SARS-CoV-2 does not allow for early diagnosis and, if possible, more specific treatment, except for the possibility of ITC when the subject is well enough. Prospective follow-up of newly diagnosed cases and their systematic request on the occurrence of hearing loss should be the norm. Under these premises, a larger volume of cases with defined criteria of clinical circumstances, laboratory findings and, of course, therapies performed will be necessary.
ConclusionsVariations in the viscoelastic properties of blood have been detected in patients with SD and COVID-19, which may present with elevated BV. In both contexts, the elevation of fibrinogen is noteworthy. This makes it advisable to investigate the presence of hearing loss of these characteristics in COVID-19, assuming that the patient's disconnection with the environment may be high, their collaboration limited and the diagnostic and therapeutic actions deferred. Larger population samples will allow the establishment of optimal pathophysiological associations.
Conflict of InterestsThe authors have no conflict of interests to declare.
Please cite this article as: García-Callejo FJ, Balaguer-García R, Lis-Sancerni MD, Ruescas-Gómez L, Murcia-López M. Viscosidad sanguínea en pacientes COVID-19 con sordera súbita. Acta Otorrinolaringol Esp. 2022;73:104–112.