Strontium zirconate SrZrO3 is a multifunctional material studied in literature as a luminescent material, proton-conductor, thermal barrier coating, and dielectric ceramics. In our study this material was sprayed by a high feed-rate water-stabilized plasma torch WSP 500 at its standard electric power 150kW. The as-deposited coatings exhibited lamellar microstructure with relatively high porosity over 13%. Archimedean (water immersion) specific weight was 4.54g/cm3. Annealing was done in air at 1350°C. Annealed coating exhibited an interesting response to UV light, including after-glow luminescence. After excitation by 225nm light the sample exhibited after-glow exponential luminescence decay with 5s characteristic lifetime. Diffused reflectance of the coatings was measured as well. The infrared reflectance is slightly lower after annealing, whereas in the ultraviolet band it is higher.
Un material multifuncional, el zirconato de estroncio (SrZrO3), estudiado en la literatura como material luminiscente, conductor de protones, recubrimiento de barrera térmica y cerámica dieléctrica se proyectó mediante un plasma estabilizado con agua WSP 500 de alta velocidad de alimentación sobre sustratos de acero inoxidable. Se produjeron revestimientos con un espesor de aproximadamente 1,2mm. El revestimiento recocido exhibió una respuesta interesante a la luz ultravioleta, incluida la luminiscencia después del resplandor. También se midió la reflectancia difusa de los revestimientos. La reflexión infrarroja es ligeramente más baja después del recocido, mientras que en la banda ultravioleta es más alta.
SrZrO3 belongs to the perovskite family with general formula A2+B4+O3. According to classification of perovskites on the basis of the ionic radii of A and B cations, SrZrO3 and its analogue CaZrO3 have orthorhombic structures [1]. At room temperature SrZrO3 was found to crystallize with either pseudocubic or orthorhombic symmetry [2], and exhibit a volume change due to a phase transition from orthorhombic to pseudotetragonal phase at 730°C [3]. Detailed investigation of phase transformations was done on SrZrO3 produced using metal chelates as starting materials [4]. Strontium zirconate seems to be really multifunctional. SrZrO3 powder was studied as a photocatalysts for hydrogen evolution from water splitting [4]. Doped SrZrO3 single crystal could also work as a proton conductor [5]. Complex oxides containing strontium are often used as optical materials, mainly as luminescent pigments [6]. Recently, polycrystalline ceramics have been developed to the point where they can compete with the performance of single crystals for light emitting applications [7–9].
Only several reports exist about strontium zirconate prepared by plasma spray process. Porosity of SrZrO3 varying from 8% for high torch power to 37% for low torch power as well as deposition efficiency were correlated with torch input power and spray distance in terms of the particle temperature and velocity [10]. Medium-size (10–50nm) intragranular pores in SrZrO3 coating (studied by small angle neutron scattering, SANS [11]), found in plasma sprayed coatings, annihilated after annealing. The in situ SANS revealed that this process occurs at 1000°C at a post-deposition thermal exposure. At 900°C nanopores are created while at 1100°C the pores begin disappearing again. Different but related materials to SrZrO3, as Dy-doped Yttria-stabilized Zirconia, were applied as phosphorescent coatings produced by plasma spray [12]. SrZrO3 coating behaves similarly to Cerium-doped Yttrium-Aluminum garnet (YAG) coating studied by us earlier [13]. Ability of Neodymum-doped YAG to serve as a luminophore for white light production in solid state light emitters was examined [8]. In the case of YAG spray coating [13], the light conversion efficiency was so far rather low for e.g. scintillator application. This is because of defects, not completely healed by the thermal annealing of plasma sprayed ceramic material. Similar situation is expectable also in the case of SrZrO3.
The aim of our actual paper is to spray strontium zirconate by a high power and high heat flux torch and study the optical character of the coating. This paper presents after-glow luminescence (AGL) experiments unique in case of thermally sprayed SrZrO3.
ExperimentalFeedstock and sprayingPlasma spray grade strontium zirconate powder supplied by Cerac Incorporated (Wisconsin, USA) was used as the feedstock. The powder size was from 74 to 150μm. Plasma spraying was done by the water-stabilized plasma (WSP) torch [14] at 150kW power (500A, 300V). WSP torch has the advantage of combining stabilizing system and cooling system in one. Water is fed into a specially shaped chamber, where it creates a swirl around the walls. Electric arc is burning between the graphite cathode inside the chamber and a rotating anode placed outside the chamber. Feedstock powder is introduced into the plasma jet outside of the torch using two injectors. These injectors can be positioned at various distances from the exit nozzle for different feedstock chemistry and size.
The feeding distance used was 80mm and spray distance 350mm. Compressed air was used as the feeding gas. The substrates were preheated to 460°C and the spray run started. Next pass was started after cooling to 170°C. After each pass of the torch the temperature rose to 350°C and was pushed down to 170°C by a compressed air flux before the next pass started. Stainless steel coupons were used as substrates. Coatings with thickness about 1.2mm were produced.
Thermal post-treatment and characterization techniquesAnnealing was done without the substrate, removed mechanically by a careful machining. Annealing of the sample used for the luminescence tests was done in air at 1350°C for 2h. Because of numerous structural defects inherently present in the plasma sprayed material it was expectable that for luminescent behaviour certain healing of the defects must be reached and the mentioned annealing regime was selected based on differential thermal analysis as a proper one.
Differential thermal analysis (DTA) curves were obtained by simultaneous thermal analyzer (TG-DTA, Bähr, Germany). A heating rate of 10K/min was applied up to 1400°C in air atmosphere (flow rate 5l/h). The weight of the feedstock powder was 45mg and it was measured in an alumina crucible. The results were corrected by blank subtraction.
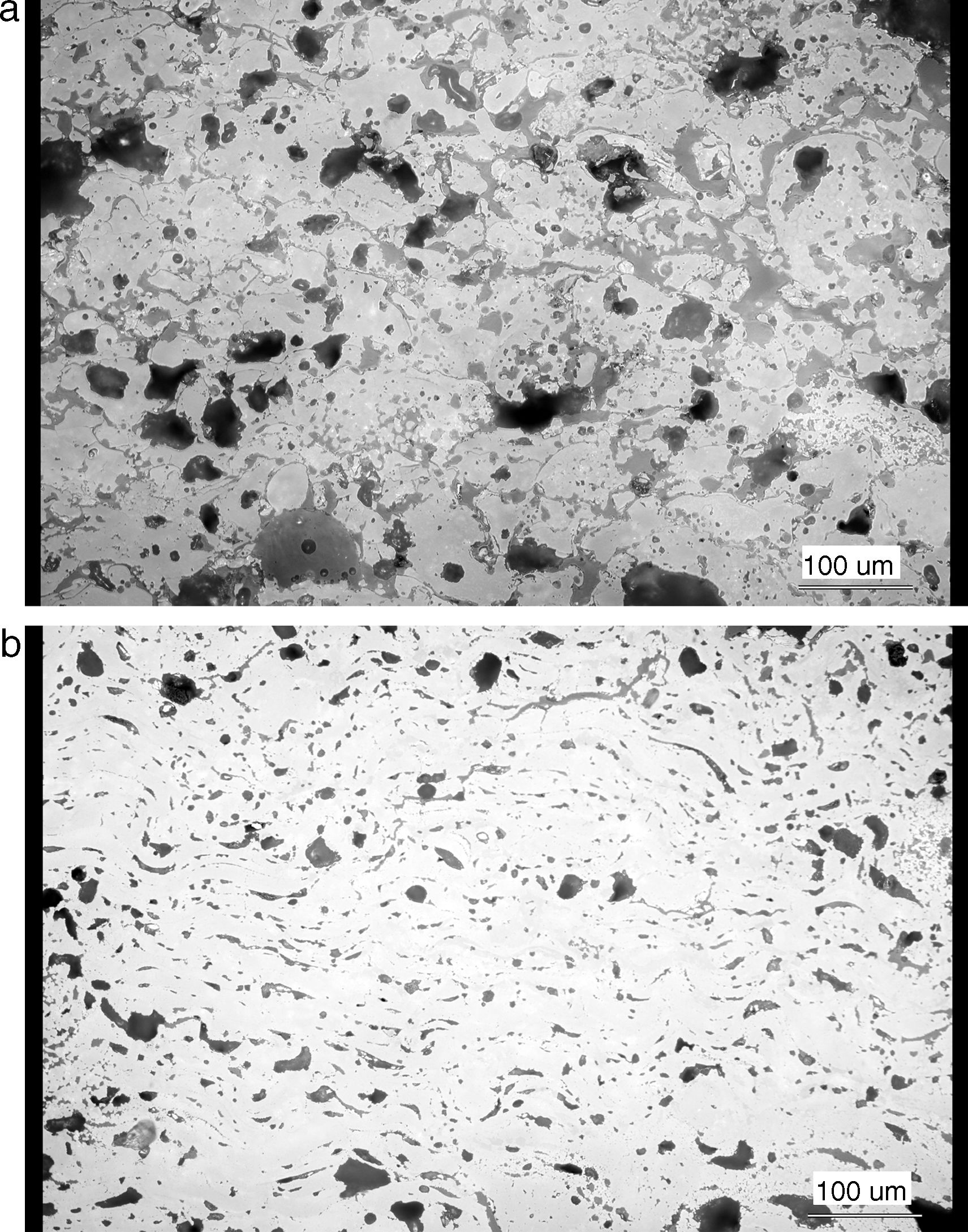
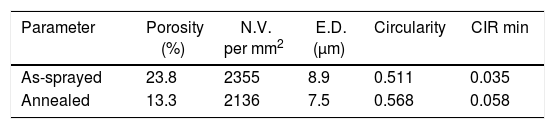
Polished cross sections of the coatings were prepared for microscopic analysis. For a better description of pores, image analysis of light micrographs was applied, and additional criteria besides the porosity percentage were introduced, Table 1. N.V. denotes the Number of Voids per unit area of the cross section and E.D. denotes Equivalent Diameter of voids representing their size distribution. Circularity could vary between 1 (belonging to a circle which represents a globular void, i.e. pore) and 0 (i.e. a line representing a flat pore or a crack). All parameters were calculated for 10 images. Resolution of used light microscopy is sufficient to provide quantification of the porosity commonly present in plasma sprayed coatings.
The phase composition was analyzed by X-ray diffraction (XRD) with CuKα radiation. D8 Discover diffractometer (Bruker AXS, Germany) equipped with 1D detector Lynxeye was used in divergent beam geometry. Moreover, the obtained XRD patterns were subjected to Rietveld refinement in order to ascertain the weight fraction of the identified phases and calculate the average values of coherently scattering domains (CDD) reflecting the so-called crystallite size.
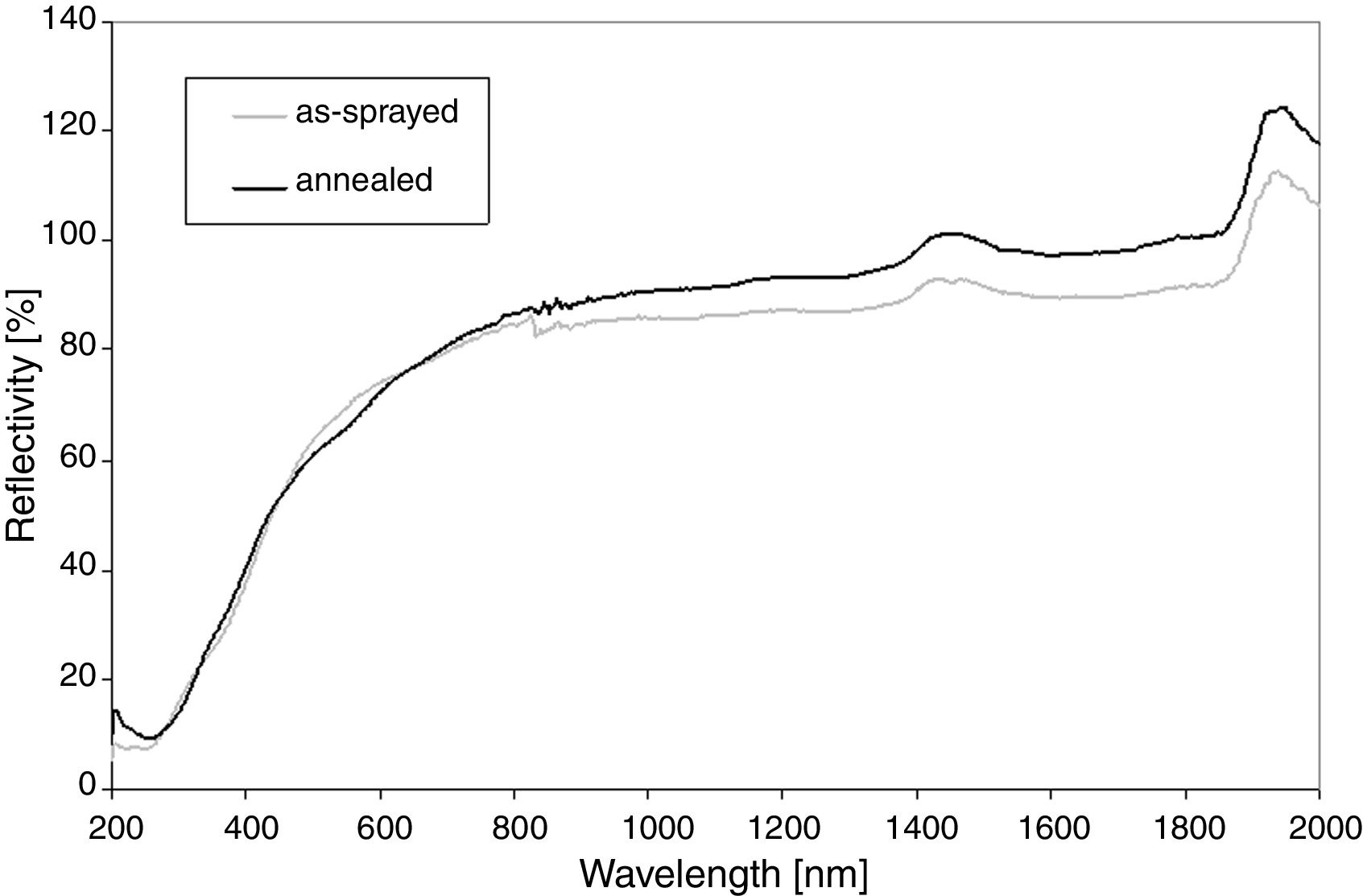
The diffuse reflectance was measured by an UV-VIS-NIR scanning spectrophotometer (Shimadzu, Japan) with a multi-purpose large sample compartment. The measured area was about 2cm2. The reflectance curves obtained between 200 and 2000nm were collected. Prior to the measurement a calibration process was conducted using a BaSO4 reference in order to minimize the error from the environment.
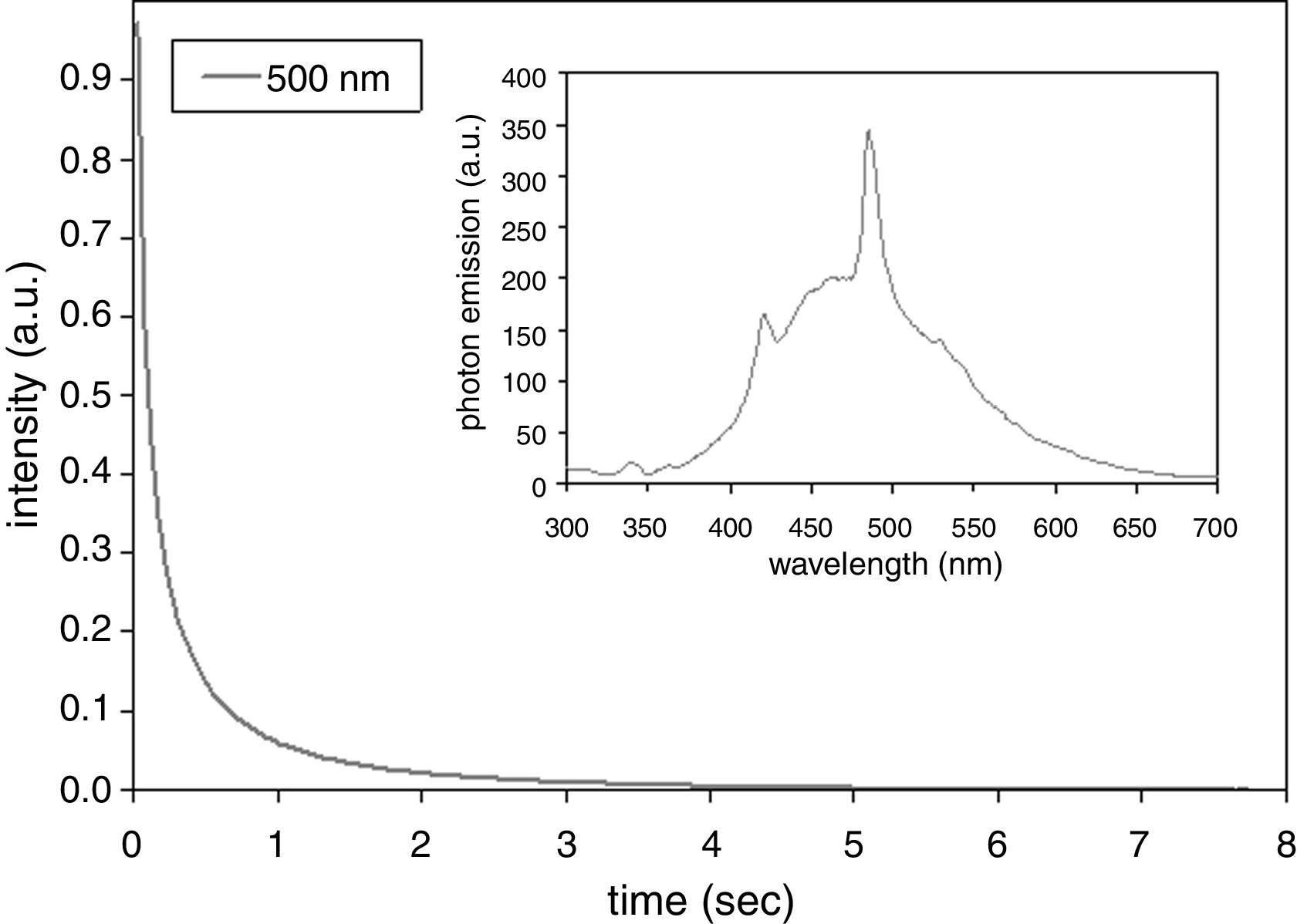
Photoluminescence experiments were performed by Varian Cary Eclipse fluorescence spectrophotometer (Agilent, USA) equipped by Xenon flash lamp and monochromator excitation source. Emission spectrum was obtained by excitation light of 250nm wavelength. Time resolved AGL curves were measured at different emission wavelengths after an excitation light of 225nm wavelength. This excitation wavelength was selected as it gave the strongest signal. The AGL curves were normalized to better compare the kinetics of the different emission wavelengths.
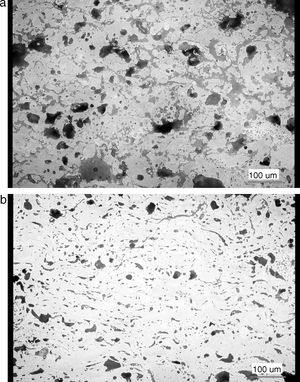
Results and discussionMicrostructure of the as-sprayed and annealed coatings is displayed in Fig. 1. The as-sprayed coating is rather porous; Archimedean (water immersion) specific weight was 4.54g/cm3, open porosity 16.03%.
The changes of porosity character after annealing could be described as transformation towards a more homogeneous microstructure. The total porosity dropped from 23.8% to 13.3%, number of voids dropped from 2355 per mm2 to 2136 per mm2, and mean equivalent diameter of a pore from 8.9μm to 7.5μm. This means that pores are after annealing less frequent and finer. Certain degree of sintering took place. The circularity rose from 0.511 to 0.568 and minimal circularity from 0.035 to 0.058. The pores became more globular. This is also an indication of sintering. The microstructure is considerably different, cf. Fig. 1a and b.
The XRD indicated presence of phases with tetragonal and orthorhombic symmetry. Due to the similarity of lattice plane distances and XRD signal intensity ratios, the tetragonal phase is isomorphous with commonly plasma sprayed tetragonal zirconia (t-ZrO2) stabilized at room temperature by Y2O3. Proximity of Y and Sr elements in the periodic table caused similarity of recorded patterns. The crystallite size of as-sprayed coating was relatively large, 413nm.
Just a small endothermic peak on the DTA curve at about 1290°C (onset) was detected, thus the coating was annealed to even higher temperature for better phase stabilization.
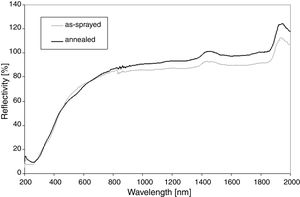
Reflectance, Fig. 2, shows that at the wavelengths over 800nm the value is near 100% because sample is non-absorbing in this region. The only absorptance seen as peaks at 1450nm and 1950nm are because of adsorbed water, always present in air, since the sample is placed in ambient atmosphere during the test. The slightly unstable run between 800 and 900nm is due to an instrumental problem. For the annealed sample any pronounced changes in the reflectivity curve were recorded. Its IR reflectivity is slightly lower, whereas in the UV band is it higher than the as-sprayed SrZrO3. This fact corresponds with the luminescent activity of the annealed SrZrO3.
The high annealing temperature was applied to a coating released from the carbon steel substrate. Being UV irradiated, the annealed (2h in air at 1350°C) sample responded by reflecting yellow visible light whereas the other, i.e. as-sprayed, samples were “UV blind”, Fig. 3.
After annealing in air the treated SrZrO3 coating exhibited changed optical properties, however not very homogeneously dispersed (c.f. Fig. 3 with some darker and lighter islands on the surface of annealed coating) due to numerous defects in the original coating structure.
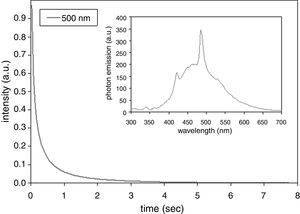
After-glow luminescence (AGL) of the annealed sample was quantified, Fig. 4. There is 10 times decrease of the emitted light intensity within the first second after ceasing of the excitation. The band at 500nm was selected as that one with the highest photon emission intensity; see the inset in Fig. 4. For other observed bands 400–700nm (100nm step) the lifetime was nearly the same. Compared to SrZrO3 prepared using polymeric precursor method [15] (420nm after excitation by 355nm [15]) the photon emission intensity maximum of our coating is shifted to higher wavelength. Compared to doped SrZrO3 produced by a gel-combustion technique (with decay times from 500μs to 1.2ms depending on dopant type) [16] our sample exhibits longer AGL response. Our present paper is, according our best knowledge, the first report on existence of the AGL phenomenon in SrZrO3 coating sprayed by plasma. Further improvements in the annealing (dwell time, atmosphere – e.g. oxygen instead of air) would offer interesting large area coatings with promising optical properties.
SummaryStrontium zirconate SrZrO3 was sprayed by a high feed-rate water-stabilized plasma torch (WSP). The as-sprayed coatings exhibited lamellar microstructure and relatively high porosity over 13%. This multifunctional material, studied in the form of bulk ceramics, thin film and recently also thick coatings as a luminescent material, thermal barrier coating, proton-conductor, and dielectric ceramics, showed interesting luminescent performance also in the form of plasma sprayed coating. After excitation by 500nm light the sample exhibited 5s after-glow luminescence with an exponential decay.
I acknowledge help of Jakub Holovský with photoluminescence measurements and support of MEYS grant no CZ.02.1.01/0.0/0.0/15_003/0000464 Centre of Advanced Photovoltaics.