Fish is a potent food allergen. The aim of this work is to demonstrate that dogfish, a small shark, has low allergenicity in both its clinical tolerance as well as its molecular structure.
MethodsWe present a study of 34 paediatric patients with IgE-mediated immediate reactions after eating fish. The diagnosis of several fish allergies was demonstrated by skin prick techniques and determination of specific IgE, in all the cases excluding sensitisation to Anisakis simplex. Open oral challenge test was checked with dogfish. Analysis was by SDS-PAGE of dogfish and other fish (megrim, shark, hake, sole, cod, anchovy and tuna) and Western-blot with “pool” of patients polysensitised sera against extracts of dogfish and other fish, and ELISA – inhibition with the “pool” sera.
ResultsThe prick-prick with raw dogfish was slightly positive in six patients, however cooked was negative in 34 cases. The specific IgE showed in the 34 cases class ≥2 for megrim, hake, sole, cod and anchovy, class 0 for tuna in 26 patients, class 0 for emperor in 18 patients and class 0 to Anisakis simplex in all cases. The IgE binding capacity for proteins of allergenic extracts of tested fish revealed, in immunoblotting, the absence of IgE-mediated recognition abstract dogfish by the “pool” of polysensitised patient sera.
ConclusionsTesting in vivo and in vitro demonstrated the low allergenicity of dogfish. Dogfish brings an alternative to eating fish in polysensitised patients.
Fish is a potent food allergen.1 In countries like Spain, with high consumption of fish, it is one of the main causes of food allergy, ranking third in incidence after milk and egg. In terms of prolonged persistence it is ranked second in prevalence after egg.1–3 Many species involved have been described, but variations depend mainly on availability and consumption.1 Thus, fish food allergy is more prevalent in geographic areas where fish is an important component of the diet, such as Spain, Portugal and the Scandinavian countries.4 Many cross-reactions between different species have also been documented.4,5 The first clinical manifestation occurs in the first two years of life coinciding with the introduction of food in the diet.6 The clinic will start within 30–60min after ingestion and the most common symptoms are urticaria and angio-oedema.1,3 Episodes of bronchospasm after inhalation of vapours have also been described. The major allergens are fish parvalbumins, proteins of molecular weight about 20kDa present in the muscle tissue of a wide variety of species, but other allergens have also been described.6,7
The aim of the present work is to demonstrate that the dogfish is a small shark with low allergenicity.
Material and methodsThe current study includes 34 children with immediate reactions (urticaria and/or angio-oedema) after IgE-mediated fish intake (megrim, hake, cod, anchovy). Fifty-three percent tolerated intake of canned tuna and emperor, only 23.5% of canned tuna and the remaining 23.5% did not tolerate any kind of fish. The mother of a polysensitised patient told us in a review if the child could eat dogfish, and thus we performed prick-by-prick technique with raw and cooked dogfish. Given the negativity of the two tests and well tolerated in open controlled oral challenge with cooked dogfish, we decided to incorporate it into the battery of fish and thus the first series of 34 patients was created.
Prick-by-prick is a common allergy test performed in food allergies using fresh foods instead of commercial extracts. It is a simple, safe, and quick test, providing results within 15–20min.
We performed skin prick test with commercial extracts of battery white and blue fish and Anisakis simplex. The specific IgE levels of the patient's serum against several species of fish (megrim, hake, sole, cod, anchovy, tuna and emperor) and Anisakis simplex were measured by fluorescence enzyme immunoassay (ImmunoCAP system). Briefly, the proteins of interest were covalently coupled to the cellulose solid phase and bound to the specific IgE of the patient's serum. The fluorescence signal obtained was measured according an IgE-calibrator range of 0.35–100kU/L.
Protein in denaturing and reducing conditions from megrim, dogfish, shark, hake, sole, cod, anchovy, and tuna were separated by molecular mass in an SDS polyacrylamide gel according to the Laemmli9 procedure with minor modifications of protocol in accordance with the manufacturer's instructions. Afterwards, proteins were transferred onto PVDF membrane. Blocking was carried out for one hour at room temperature with PBS containing 0.1% Tween 20 (PBST). Patient's serum was diluted in PBST and incubated overnight at 4°C. Bound antibodies were visualised by anti-IgE antibodies conjugated with peroxidase (Sigma–Aldrich) and subsequent chemiluminescence detection.
The procedures used on patients were carried after obtaining informed consent.
We followed the protocols of our health centre to access details of clinical records, in order to publish their manuscript for divulgation purposes for the scientific community.
ResultsSkin prick tests were performed with commercial extracts of megrim, hake, sole, cod, anchovy, tuna and emperor and Anisakis simplex. In the 34 patients they were positive for all fish tested except for tuna in 26 patients and emperor in 18 patients, with Anisakis simplex resulting negative in all cases. The prick-by-prick with raw dogfish was slightly positive in six patients, however cooked was negative in 34 cases.
Specific IgE was determined for several species of fish (megrim, hake, sole, cod, anchovy, tuna and emperor) and Anisakis simplex using the Phadia CAP method. In the 34 cases, the results showed class ≥2 for megrim, hake, sole, cod and anchovy, class 0 for tuna in 26 patients, class 0 for emperor in 18 patients and class 0 to Anisakis simplex in all cases.
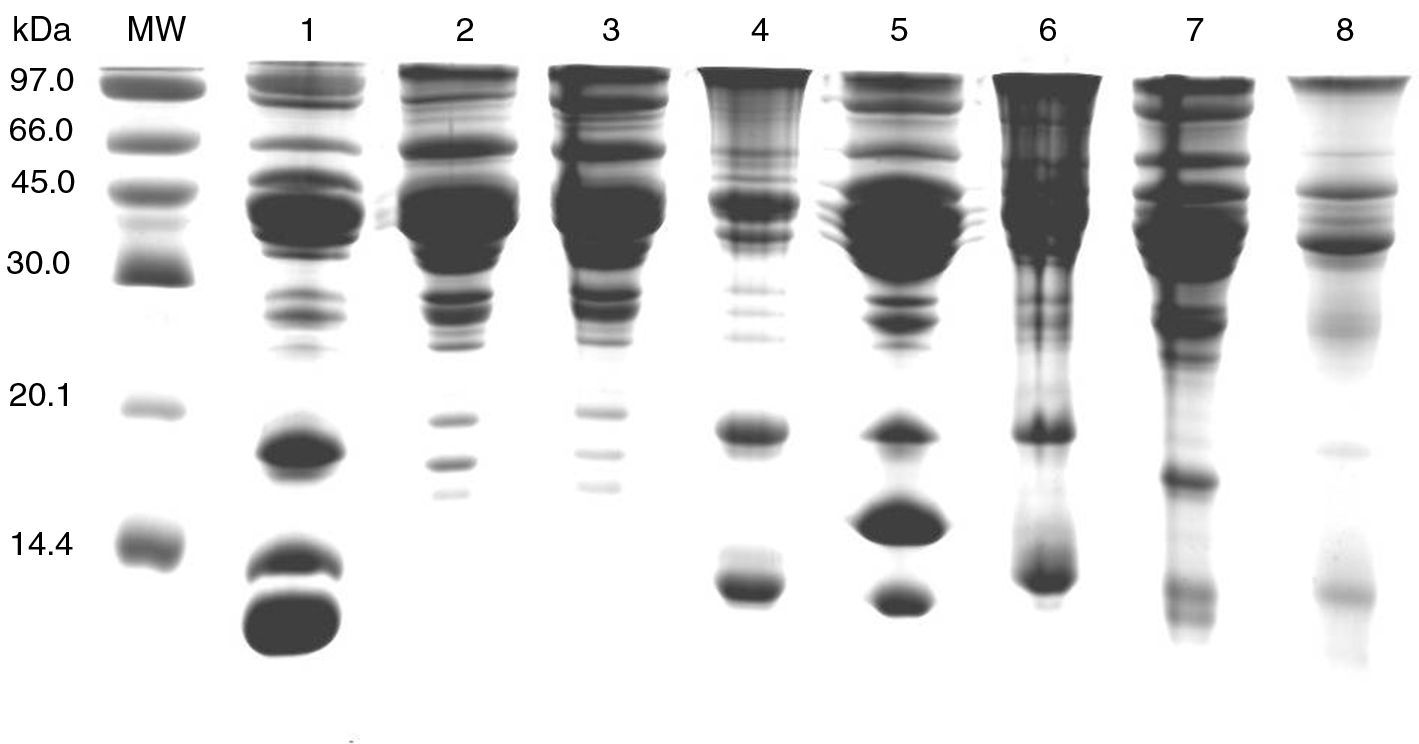
The SDS performed with several fish involved showed a pattern of protein bands with molecular weights ranging from 12 to 90kDa (Fig. 1).
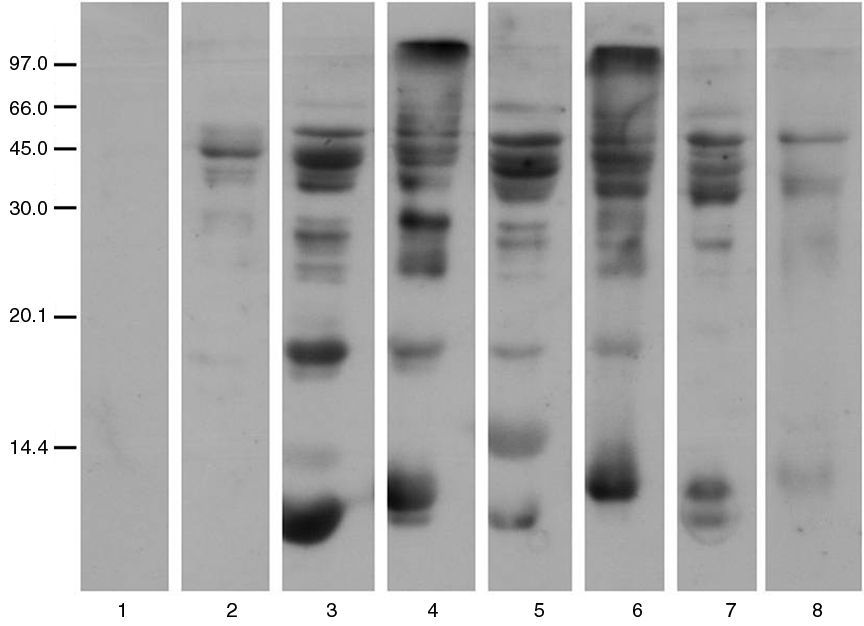
The IgE binding capacity for proteins of allergenic extracts of tested fish revealed in immunoblotting the absence of IgE-mediated recognition abstract dogfish by the pool of polysensitised patient sera (Fig. 2).
In view of the results found, the subjects underwent controlled open oral provocation with cooked dogfish beginning with a skewer, in increasing amounts every 20min to complete the full amount of a ration. After the last shot, there was a waiting period of 90min. Cooked dogfish tolerance was obtained in 100% of the cases.
DiscussionFood allergy is perceived as a major health problem in children. There is a real variation in the prevalence of food allergy in different areas of the world, as well as an increase in recent decades. Many foods have been implicated in IgE-mediated reactions, among which are the proteins of cow's milk, eggs, nuts, fish and shellfish.1,2 Allergic sensitisation is defined by the presence of specific IgE antibodies to a particular allergen.1–8
Food allergy is defined by the presence of clinical manifestations during or after the ingestion of foods that the patient showed allergic sensitisation to. The dogfish is a small shark with a slender body size and maximum weight, of two metres and 35kg, respectively. Its use is common in the area of Andalusia where it is highly prized and is known as “bienmesabe”.
The study of the allergens of various species by SDS-PAGE shows the absence of low molecular weight proteins in dogfish, another kind of white shark, similar to the protein structure of tuna. These proteins of molecular weight between 15 and 20kDa bands correspond to parvalbumins, major allergens of the fish.7,8 This would explain the negative results of skin tests with dogfish prick-by-prick and tolerance of it by 34 patients.
Therefore, apart from the emperor and canned tuna, dogfish is better tolerated in polysensitised fish patients. We facilitate the consumption of dogfish, which could be included in the battery study because of its proven low molecularly allergenicity and excellent clinical tolerance.
Conflicts of interestPineda F and Pérez R are employees of Diater Laboratories, Madrid, Spain. The rest of the authors have no conflicts of interest to disclose.
Ethical disclosuresPatients’ data protectionConfidentiality of Data. The authors declare that they have followed the protocols of their work centre on the publication of patient data and that all the patients included in the study have received sufficient information and have given their informed consent in writing to participate in that study.
Right to privacy and informed consentThe authors have obtained the informed consent of the patients and/or subjects mentioned in the article. The author for correspondence is in possession of this document.
Protection of human subjects and animals in researchThe authors declare that the procedures followed were in accordance with the regulations of the responsible Clinical Research Ethics Committee and in accordance with those of the World Medical Association and the Helsinki Declaration.
This case was studied in the Hospital Universitario Niño Jesús in Madrid, in collaboration with Diater, Madrid laboratories, Spain. In memoriam of Dra. Carmen Muñoz Martínez, thank you for your initiative and enthusiasm.