The aim of this paper is to discuss the current evidence regarding short and long-term health respiratory effects of nutrients and dietary patterns during the first 1000 days from conception. Population of interest included children from birth to two years and their mothers (during pregnancy and lactation). Studies were searched on MEDLINE® and Cochrane database, inserting individually and using the Boolean ANDs and ORs, ‘nutrients’, ‘micronutrients’, ‘LC-PUFA’, ‘Mediterranean Diet’, ‘human milk’, ‘complementary food’, ‘pregnancy’, ‘respiratory disease’, ‘pulmonary disease’, ‘asthma’, ‘epigenetics’, ‘first 1000 days’, ‘maternal diet’ and ‘respiratory health’. All sources were retrieved between 01-09-2015 and 07-12-2016. While unhealthy maternal dietary patterns (high fat intake) during pregnancy can result in alteration of foetal lung development, with increased risk of respiratory disorders, Mediterranean diet has been associated with a lower risk of allergic sensitisation and allergic rhinitis. Breastfeeding has beneficial effects on respiratory infections while evidences about its protective effect on allergic disorders are unclear. During complementary feeding there is no evidence to avoid or encourage exposition to ‘highly allergenic’ foods to have modification of tolerance development. In children from birth to two years of age, Mediterranean diet has been associated with a lower risk of atopy, wheezing and asthma. Micronutrients, antioxidant and LCPUFA supplementation is not recommended and a whole food approach should be preferred, except for Vitamin D.
Nutrition and dietary patterns are recognised as modifiable risk factors able to influence the respiratory disease development and progression as they can regulate the immune system. Therefore, nutrients can have a high impact on individual respiratory health and contribute to the development of the organism, and should not be considered only as an energy source.1 Prenatal and early post-natal periods have a critical role in the individual outcome, as Barker affirmed: “Much of human development is completed during the first 1000 days after conception”. The first 1000 days of life from conception to two years are considered critical in nutrition for enhancing short and long-term health outcomes. Early nutrition may influence the risk of developing non-communicable diseases (NCDs) later in life, including heart disease, hypertension, type 2 diabetes (T2DM) and chronic respiratory diseases (such as asthma) through epigenetic mechanisms.2
Furthermore, diet and nutrition may influence the development of the microbiota which may influence the immune, inflammatory and allergic mechanisms and thus can contribute to the development of many diseases.3
Aim of the reviewCurrently, there is a lack of reviews amalgamating short and long-term effects of nutrition early in life on respiratory health. The aim of this paper is to discuss the current evidence regarding the short and long-term respiratory health effects of nutrients and dietary patterns during pregnancy, breastfeeding and complementary feeding and the potential underlying mechanisms.
MethodologyEligibility criteriaThe populations of interest were children from birth to 18 years and their mothers (during pregnancy and lactation). Inclusion criteria were: type of article (multicentre study, review, systematic review, observational study, case–control study, longitudinal/prospective study, retrospective study, randomised controlled trial), publication date (2011–2016), species (both human and animal), English language. Texts available only in abstract form were excluded.
Information sources, search strategy and study selectionPublications were searched on MEDLINE® and Cochrane database inserting terms individually and using the Boolean ANDs and ORs. Other publications coming from the reference list of studies extracted from MEDLINE, Cochrane database and from the personal reference databases of the authors were also evaluated. In the search strategy the following terms were included: ‘nutrients’, ‘micronutrients’, ‘LC-PUFA’, ‘Mediterranean Diet’, ‘human milk’, ‘complementary food’, ‘pregnancy’, ‘respiratory disease’, ‘pulmonary disease’, ‘asthma’, ‘epigenetics’, ‘first 1000 days’, ‘maternal diet’ and ‘respiratory health’.
All sources were retrieved between 01-09-2015 and 07-12-2016. The data screening and extraction were conducted independently by two authors and any variances resolved between them.
Maternal diet and respiratory healthUnhealthy lifestyle (alcohol, tobacco smoking, caffeine use, etc.) during pregnancy can influence foetus growth and development. Several studies showed that a correct lifestyle, even before pregnancy, can be effective for ensuring a healthy outcome for women and their babies.2 Diet is one modifiable risk factor that women should be motivated to improve.4
Maternal dietPromoting healthy diet in childbearing age women should be a cornerstone of public health. The study of Mayor et al. has recently supported the crucial role of nutrition during pregnancy on health of offspring. Poor maternal nutrition influences placental development and foetal growth, resulting in low-birth-weight and enhanced risk of developing NCDs later in life.4 However, the most interesting result of the study is that a maternal high-fat diet before and during pregnancy increases glucose and insulin levels and induces placental inflammation resulting in placental insufficiency, foetal growth restriction and alteration of foetal lung development. The impairment of foetal lung maturation may predispose to an increased risk of respiratory distress syndrome at birth and later of chronic lung disease. These results are important, first of all because the main dietary pattern of developed countries is characterised by a high consumption of saturated fats, red meats and poor intake of fresh fruits and vegetables, whole grain and seafood (the so-called Western diet).4 This is an example of how maternal dietary habits can have long-term health consequences on infant.
The incidence of allergic diseases is increasing, both in developed and developing countries, concomitantly with the rise in living standards and the adoption of a ‘Western lifestyle’. Studies highlight the first and second trimesters of pregnancy as critical periods for the development of allergic diseases, such as asthma.5 Diets that are rich in fruit and vegetables, such as the Mediterranean diet (MD), have consistently been associated with a lower risk of allergic sensitisation (which is defined as a positive skin prick test to allergens) and allergic rhinitis in children in epidemiological studies, whereas a high maternal consumption of vegetable oils, margarine and processed foods has been associated with a higher risk of allergic sensitisation.5,6 Mediterranean diet represents a balanced diet, characterised by frequent consumption of high amounts of fibres (whole cereals, legumes, vegetables, fruits and nuts), chemical compounds with anti-oxidative properties (flavonoids, phytosterols, vitamins), fish, unsaturated fatty acids (FAs) from olive oil and low intake of red meat.7 Whole-grain foods are rich in bioactive compounds with anti-inflammatory properties, as well as vegetables and fruits. On the contrary, the consumption of red meat and foods with high glycaemic index may increase oxidative stress and chronic low-grade inflammation.8 Therefore, the Mediterranean diet may constitute a promising approach to address inflammation-driven perturbation in gut microbiota, as present in allergic conditions. Indeed, it has been observed that high adherence to a Mediterranean diet is associated with a ‘normalisation’ trend of gut microbiota.7
Additionally, the majority of recent clinical studies enrolling allergic mothers indicate that allergen avoidance (e.g. elimination of milk or eggs) during pregnancy is ineffective and unnecessary to prevent the development of allergy in the child: prolonged elimination diets indeed expose mother and infant to the risk of nutritional deficiencies.5,9
Maternal supplementationSupplementations to mothers to modulate the developing immune system is another approach that has received interest. Omega-3 polyunsaturated long-chain fatty acids (LC-PUFAs) from marine sources (fish) have been shown to have anti-inflammatory properties through several cellular mechanisms including their incorporation into cellular membranes and the synthesis of eicosanoids.1 Maternal omega-3 LCPUFAs supplementation during pregnancy has been associated in some studies with a reduced risk of allergen sensitisation, risk of asthma development, and atopic dermatitis in children although without beneficial impact demonstrated on the development of food allergy.1,5,9 However, the data examining the possible benefits of dietary omega-3 LCPUFAS supplementation on respiratory allergic disease, in particular asthma, are heterogeneous: their role in protection against allergy requires further investigation. Thus, to date there is insufficient evidence to recommend specific supplementation or a prevention diet.5
Considering micronutrients, current evidence suggests a protective effect of maternal intake of each vitamin D, vitamin E, and zinc against childhood wheeze, but is inconclusive for an effect on asthma or other atopic conditions.10
ConclusionIn summary, current data do not support maternal dietary restrictions during pregnancy to protect the infant against the development of later allergic diseases and attention to adequate nutrition, improving adherence to the Mediterranean diet, could be the most important strategy to prevent respiratory diseases. There is insufficient evidence to recommend specific nutrient supplementation in order to prevent or treat asthma or allergies. Further research in this area is needed.
Breastfeeding and the risk of respiratory infections and atopic diseasesHuman milk is the normative standard for infant feeding and nutrition; short-term and long-term advantages of breastfeeding are well documented.
Short-term effectsRegarding short-term benefits, breastfeeding reduces morbidity and mortality due to infectious disease in childhood.11 The source of these health beneficial effects may be the peculiar composition of breast milk: important components are anti-inflammatory cytokines and pathogen neutralising secretory IgA antibodies, lysozyme, lactoferrin and galacto-oligosaccharides.12
The protective effect of breastfeeding against mortality and morbidity from respiratory infections has been widely studied. In 2012, the American Academy of Paediatrics (AAP) published a policy statement regarding the effects of human milk. Focusing on lower respiratory tract infections (LRTI), it reported that infants exclusively breastfed for four months have a consistent reduction (72%) of the risk of hospitalisation in the first year of life. Even the severity (duration of hospitalisation and oxygen requirement) of respiratory syncytial virus (RSV) bronchiolitis was reduced by 74% in infants who breastfed exclusively for four months compared with infants who never or only partially breastfed.13 The World Health Organisation (WHO) has recently published a systematic review focused on the beneficial effects of human milk. Data collected show a 30% reduction of morbidity, about 50% of hospital admissions and about 60% of mortality for LTRI, suggesting that breastfeeding affects not only the incidence but also the severity of these infections.11
Long-term effectsIn children with familiar predisposition to atopy, increased diversity of food antigen exposure in the first year of life seems to suggest the inhibition – the so-called allergic march – and so is inversely correlated with the development of atopic dermatitis, rhinitis and asthma. Consequently, the inclusion of food allergens in early childhood diet might be beneficial for the prevention of allergy and, in particular, of asthma.5
Regarding breastfeeding, several epidemiological studies have addressed the effects of human milk on the development of allergic disease during childhood. Probably human milk can modulate the onset of allergies: this can be attributed principally to compounds that facilitate development of host defence mechanisms (secretory IgA, IgG, and factors that actively stimulate the infant immune system).14 However, the evidence is not so clear: no association between breastfeeding and food allergies, while some evidence of protection against allergic rhinitis in children younger than five years have been observed. Moreover, the evidence is inconclusive for the association between breastfeeding and the risk of eczema, wheezing or asthma.14,15 Among the most recent clinical studies, some have concluded that exclusive breastfeeding for at least three months reduces the risk of asthma between two and four years of age, evidencing a stronger protection in children born in atopic families.4 Other studies, by contrast, have reported no benefit of exclusive breastfeeding in children from non-atopic families16 or in decreasing the risk of asthma in children at five years of age.17
Therefore, not all results of studies are conclusive in showing a beneficial effect of breastfeeding on the risk of asthma and atopic disease. The underlying biological mechanisms are not entirely clear, but long-chain PUFAs, which are contained in breast milk, might play a major role.18 In a recent study, asthma prevalence (defined as ever having asthma up to age 10 years) decreased with increasing duration of exclusive breastfeeding. Moreover, in a second analysis stratified by FADS genotypes (polymorphisms in the fatty acid desaturase genes) asthma prevalence was significantly reduced only in children who had been exclusively breastfed for at least three months and were carrying at least one copy of the minor allele of the investigated SNPs. In contrast, children being homozygous for the major allele showed no significant benefit from having been exclusively breastfed.19 These results suggest that a certain group of children with a defined genetic background are more sensitive to nutritional influences, e.g. breastfeeding. In this study, only those children who are less able to convert precursor PUFA to their longer-chain products show a benefit after at least three months of exclusive breastfeeding.
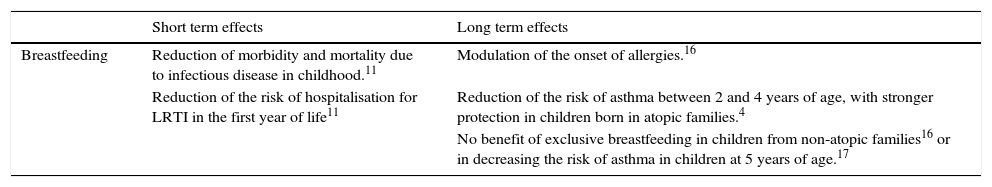
ConclusionIn conclusion, at the moment it is difficult to draw definitive indications about the beneficial effect of breastfeeding on asthma and atopic diseases.5 In any case, breastfeeding should be encouraged because of its several benefits, including protection against pulmonary infections. WHO recommends exclusive breastfeeding up to six months of age, with continued breastfeeding along with appropriate complementary foods up to two years of age or beyond20 (Table 1).
Effects of breastfeeding on respiratory infections and asthma.
| Short term effects | Long term effects | |
|---|---|---|
| Breastfeeding | Reduction of morbidity and mortality due to infectious disease in childhood.11 | Modulation of the onset of allergies.16 |
| Reduction of the risk of hospitalisation for LRTI in the first year of life11 | Reduction of the risk of asthma between 2 and 4 years of age, with stronger protection in children born in atopic families.4 | |
| No benefit of exclusive breastfeeding in children from non-atopic families16 or in decreasing the risk of asthma in children at 5 years of age.17 |
The introduction of complementary foods during infancy has a discussed role in the risk of developing allergy. No effect of delayed introduction of solid foods on the prevalence of food allergies has been suggested by the results of a number of prospective birth cohort studies, e.g. the GINI Study,21 LISA Study22 and the KOALA Study.23 Regarding the early exposure to solid foods during infancy, while it was previously associated with the development of allergic diseases, currently oral tolerance induction is being investigated to determine if early introduction of highly allergenic foods (cow's milk, egg, fish, peanuts), after at least 3–4 months of exclusive breastfeeding will result in the reduced prevalence of food allergies. In order to provide a conclusive answer on preventive effectiveness and the safety of early administration of allergenic foods different randomised trials have recently been conducted.24–26 Unfortunately, they are burdened by major methodological problems. In the Prescott study (early egg exposure)24 the final sample size corresponds to less than half compared to that calculated a priori (87 vs. 206), due to the impossibility (financial constraint) of new patients enrolling. Moreover, the different incidence of egg allergy and of SCORing Atopic Dermatitis (SCORAD) index in atopic dermatitis between the group that introduced the egg to four months and the control group was not statistically significant, but this result could be affected by the small sample size at the end of the study.
The study LEAP (early exposure to peanuts),25 was planned to compare the effects of peanut avoidance until three years of age with early peanut introduction in 640 high risk infants (age 4–10 months) with atopic dermatitis and/or egg allergy. However, children having one SPT to peanut >4mm, theoretically more at risk, were excluded. Moreover, the timing of peanut introduction, considered “early”, was actually between four and 11 months.
The latest study, the EAT (Enquiring about Tolerance) study, was designed to determine whether an early (at three months of age) introduction of six allergenic foods (cow's milk-based yoghourt, egg, fish, wheat, sesame, peanut) into the diet of unselected infants, together with breastfeeding, compared with later (at six months of age) introduction while breastfeeding could have an impact on the risk of food allergies at three years of age.26 This study is burdened by a series of bias among which the most important are the very low compliance to the intervention (less than 40%) and a protocol that does not allow the proper registration of all cases of allergy. For these reasons we cannot consider the results of this study reliable.
ConclusionIn conclusion, the available evidence does not suggest that early exposures may modify tolerance development and further research on these exposures has to be continued. In 2014, the European Academy of Allergy and Clinical Immunology's (EAACI) Taskforce on Prevention published the EAACI Guidelines for Food Allergy and Anaphylaxis. For primary prevention, once complementary feeding has started, there is no evidence to avoid or encourage exposition to ‘highly allergenic’ foods, irrespective of atopic heredity: timing of introduction of complementary foods should be identical.27
In children with familiar predisposition to atopy, the timing of introduction of highly allergenic foods during complementary feeding has to be further investigated to better evaluate the effect of an early food antigen exposure on the risk of the allergic march, associated with the development, in particular of rhinitis and asthma.5,28
Quality of diet in the first two years and respiratory disease's protectionAfter the complementary feeding, a type of diet such as MD has been associated with a protection against atopy, wheezing and asthma also in children, not only in adults. On the contrary, the so-called Western diet has been associated with increased risk of asthma, wheezing and airway hyperresponsiveness.1 Recently, it has been observed that low consumption of fruits and high intake of meat by children in the first years of life was associated with negative effects on wheezing, rhinitis or dermatitis at preschool age.29
Quality of diet, in term of intake of micronutrients (iron, zinc, selenium, vitamin A, vitamin D, vitamin C, vitamin E, flavonoids and carotenoids present in fruit and vegetables and omega-3 LC-PUFAs) has an important role in the protection of respiratory tract.
In particular, an improvement of antioxidant activity reachable with optimal intake of antioxidant micronutrients may represent a successful strategy for delaying the inset, influencing the development and/or reducing the gravity of asthma. Recent experimental studies and clinical data suggest that asthma is associated with enhanced production of reactive oxygen species (ROS) and reactive nitrogen species (RNS) and changes in enzymatic antioxidant activity in lung and blood, playing a central role in airway inflammation and pathogenesis of this disease. ROS and RNS are released from infiltrated inflammatory cells, particularly mast cells, eosinophils, neutrophils, and T lymphocytes. ROS regulate cellular signalling and activate redox-sensitive transcription factors, a necessary prerequisite for induction of pro-inflammatory cytokine gene expression (e.g. tumour-necrosis factor (TNF)-α and macrophage-inflammatory protein (MIP)-2); RNS further contribute to the perpetuation and amplification of airway inflammation. Both ROS and RNS enhance systemic oxidative stress, thus increasing the oxidative burden due to the alteration of systemic and blood antioxidant systems, typical of bronchial asthma. An improvement of antioxidant activity may represent a successful strategy for delaying the onset, influencing the development and/or reducing the gravity of asthma. The main antioxidants are vitamins C and E, β-carotene, selenium and coenzyme Q10, significantly reduced in children suffering from atopic asthma.30
Iron, zinc and seleniumIron and zinc are micronutrients with many important functions in the immune system. Iron deficiency seems to impair secretion of cytokines, and reduce bactericidal macrophage activity and T-cell proliferation; however, clinical studies do not convincingly show an increased risk of infections in case of iron deficiency.31 Zinc seems to inhibit viral replication or intracellular adhesion, and to enhance innate immune response at the mucosal surface: consequently, it has been investigated for treating upper respiratory infections. Data are more limited for its use in order to prevent otitis media; systematic reviews do not support a role for zinc supplements to reduce the occurrence of otitis media in healthy children. In paediatric practice, especially in settings where nutritional status is presumed to be generally adequate, use of pharmacological zinc treatment is relatively uncommon.32 Selenium is not an antioxidant per se but is required at the active sites of the antioxidant enzyme glutathione peroxidase. It can positively influence the systemic redox balance and in consequence pulmonary inflammation in asthmatic children.30
VitaminsA study population conducted on 3202 Bolivian children aged 5–12 years suggested that vitamin A is important in maintaining immune function or limiting severity of respiratory tract infections in children older than five years of age. Intervention studies are warranted to confirm that improving vitamin A status reduces the risk of morbidity in school-aged children.33
Folate plays a key role in nucleic acid and protein synthesis by supplying one-carbon units and therefore its metabolism is profoundly implicated in the DNA methylation pathway. Folate/folic acid is required for neural tube development occurring within 28 days of conception and its role in the prevention of neural tube defects (NTDs) is well established. However, high supplement levels of folic acid after the first trimester have been associated with an increase in childhood asthma and eczema. Indeed, with regard to epigenetics, inadequate folate may significantly alter the immune response, compromising the DNA methylation at genes regulating Th differentiation. Thus, too much dietary supplementation with methyl donors during a vulnerable period of foetal development might modify the heritable risk of allergic disorders in the offspring via epigenetic mechanisms. However, further investigation is warranted to draw conclusions as to the question of whether maternal folate intake during pregnancy is related to the risk of childhood allergic disorders.34
Vitamin D is a secosterol produced endogenously in the skin from sun exposure or obtained from few foods (cod liver oil, fatty fish such as salmon, mackerel and tuna, fortified foods). One of the major physiological functions of vitamin D is to maintain serum calcium and phosphorus levels in a healthy physiological range to maintain bone metabolism. However, its actions go further than calcium and bone metabolism: the discovery that many tissues and cells in the body have vitamin D receptor (VDR) has provided new insights into the function of this vitamin. The VDR is present in many organs and tissues including cells of the immune system (e.g. macrophages, T and B lymphocytes); vitamin D features its immunomodulatory effects on both the innate and adaptive immune systems.35 Vitamin D effects on innate immune system are predominantly through the toll-like receptors and on the adaptive immune system through T-cell differentiation, particularly the type 17T helper cell (Th17) response.35
The activation of VDR on dendritic cells has been proven to modulate the tolerance of these antigen-presenting cells in adaptive immune responses. Th2 response is enhanced by Th1 inhibition and also as a result of balance shifting towards Th2. Moreover, it has been demonstrated that vitamin D inhibits interferon (IFN)-γ production and promotes interleukin (IL)-4, IL-5 and IL-10 production in a mouse model.36
Epidemiological studies showed promising associations between vitamin D and pulmonary health. Vitamin D has probably a role in the onset, progression and exacerbations of respiratory tract infections, asthma and chronic obstructive pulmonary disease (COPD). The mechanisms responsible for these effects are not completely understood,1 but vitamin D seems to be a pleiotropic regulator of immune function that might exert different effects depending on the timing of exposure, dose and possibly also genetic variability.5 Recent data suggest that the active form of vitamin D is able to influence the foetal lung maturation and airway smooth muscle cell proliferation and differentiation per via paracrina.37 Effects of vitamin D are also reported in respiratory diseases: lung function, increased corticosteroid use and asthma exacerbations have been reported in asthmatic children with hypovitaminosis D. Vitamin D probably modulates T cell driven immune responses and thus influences the onset of asthma. Another possible mechanism is the reduction of steroid responsiveness.38 Therefore, it is fundamental to ensure vitamin D supplementation in children with vitamin D insufficiency and asthma, but as yet there is not conclusive evidence for guidelines on vitamin D supplementation for asthma.37,39
Vitamin C is one of the most effective nutritional antioxidants by inhibiting the generation of ROS.
By counteracting oxidants and reducing external attacks (bacteria, viruses, toxins, xenobiotics) in the lung, vitamin C may modulate the development of bronchial inflammation and the impairment of pulmonary function. Several studies have demonstrated that vitamin C supplementations are not effective in reducing the incidence of cold in the general population, but can reduce its duration. Routine vitamin C supplementations are not yet justified; indeed, given the consistent effect of vitamin C on the duration and severity of colds in the regular supplementation studies, and the low cost and safety, it is possible to test whether vitamin C supplementation is useful for common cold patients.40 Moreover, vitamin C supplementation has shown value for the prevention of wheezing and asthma in the children of smokers.41
Antioxidant ROS scavengers in the diet reduce oxidative stress and therefore can prevent or modulate the development and progression of lung inflammatory diseases (e.g. asthma). However even though dietary antioxidants are associated with positive effects on inflammation, clinical outcomes and respiratory disease prevention, intervention studies of antioxidants do not indicate widespread adoption of supplementation. A whole food approach has the benefit of increasing intake of multiple nutrients; moreover, it can help individuals in learning a correct lifestyle.1
Polyunsaturated long-chain fatty acidsExperimental in vitro studies have shown that omega-3 LC-PUFA has anti-inflammatory effects by inhibiting inflammatory cell production of pro-inflammatory prostaglandin (PG) E2, leukotriene (LT) B4 from arachidonic acid (AA) and decreasing activity of nuclear factor-kappa B (NF-κB), a potent inflammatory transcription factor. Moreover, downregulate pro-inflammatory cell cytokine production (IL-1β, TNF-α) by monocytes and macrophages reduces expression of cellular adhesion molecules on monocytes and endothelial cells and production of ROS in neutrophils.1 Plasma fatty acids (FA) are able to modulate inflammation; specific arachidonic acid-derived mediators, called eicosanoids, contribute to the initiation of inflammation; in contrast, mediators derived from omega-3 LCPUFAs (DHA and EPA), named resolvins, protectins and maresins, have potent anti-inflammatory, tissue protective and resolution stimulating functions. The FA unbalance may therefore contribute to the inflammatory process leading to progressive lung damage.42 However, now there is no scientific evidence for omega-3 LC-PUFAs oral supplementation in preventing or treating inflammatory respiratory diseases.1
The data mentioned are summarised in Table 2.
Micro and macronutrients intake and respiratory health outcomes during the first 1000 days of life.
| Micro and macronutrients intake | Respiratory health outcomes |
|---|---|
| Omega-3 LC-PUFAs | Modulation of inflammatory activity.42 |
| Iron | Regulation of secretion of cytokines and bactericidal macrophage activity and T-cell proliferation.31 |
| Zinc | Inhibition of viral replication or intracellular adhesion; promotion of innate immune response at the mucosal surface.32 |
| Selenium | Antioxidant activity (co-factor).30 |
| Folate/folic acid | Increase of the risk of childhood asthma and eczema if too much dietary supplementation after the first trimester of pregnancy.34 |
| Vitamin A | Supporting immune function and limitation of severity of respiratory tract infections in children older than five years of age.33 |
| Vitamin D | Immunomodulatory effects on both the innate and adaptive immune systems.35 |
| Vitamin C | Antioxidant effects.40 |
Diet and nutrition may influence the development of the microbiota, a collection of microorganisms including a large number of bacteria, archea, virusis and eukaryotic microorganisms that live in various part of the body and colonise skin, eyes, oral cavity and the respiratory, urinary and gastrointestinal tract. In particular, the gastrointestinal tract is the most heavily inhabited organ colonised by different communities of germs. The human microbiota is represented by approximately 300–500 of bacterial species with a total amount of genes estimated about 2–4 millions.3,43 The microbiome, that is the genes of the microbiota, may exceed the total number of human genes by a factor of about 100.44
The development of the microbiota is dynamic and principally influenced by mode of delivery, infant diet and use of antibiotics.45 Before delivery, many maternal factors can impact microbiota diversity and composition: maternal infections, use of antibiotics, diet and smoking. Concerning mode of delivery, it has been shown that neonates are born sterile and are colonised thereafter.44 However it is worth mentioning that recent studies have challenging this paradigm speculating that the gastrointestinal tract of foetus is colonised by germs before birth and indicating the maternal basal plate of placenta as a possible source of colonisation.46 Infants vaginally born have a microbiota representing the maternal vaginal and gut microbiota (Lactobacillus, Prevotella, Escherichia, Bacteroidetes, Bifidobacterium, Streptococcus); on the other hand, babies born by caesarean delivery have a reduction of Bifidobacterium and Bacteroidetes, and exhibit germs representative of maternal skin, such as Staphylococcus spp.44 Gestational age is another important factor that can affect microbial diversity: premature infants exhibit significantly bacterial diversity.47
Microbiota plays an important role in human health and is essential for gut maturation, metabolic and immunologic programming. Bacterial colonisation may impact the occurrence of disease later in life. First of all, it has an important role in the immune system, modulating expression of genes involved in mucosal barrier fortification and acting like a barrier against invasions of pathogens.48
Second, it heavily influences host nutrition. It synthesises vitamins (e.g. vitamin K and constituents of vitamin B) useful for human body; by fermenting carbohydrates, it leads to the production of short-chain fatty acids (SCFAs) important for energy harvest and storage.49
Dysbiosis of microbiota has been linked to the development of several disorders, including respiratory diseases. Indeed, evidences indicate that microbiota can influence the inflammatory status of respiratory tract, in a way called gut-airway axis.3
Fujimura et al. showed that oropharyngeal colonisation of newborns by Streptococcus, Haemophilus or Moraxella species at 4 weeks of age vs. 12 months was associated with higher risk of recurrent wheezing at childhood age. Moreover, intestinal dysbiosis, an alteration in the composition of the gut microbiota, may have a key role. Indeed, dysbiotic neonatal (3-week-old) gut microbiomes enriched with Escherichia coli or Clostridium difficile were found at significantly higher risk for childhood atopy development.50 Thus gut microbiota seems to have a crucial role, because immune cells requires it for their normal differentiation.51 Concerning the development of immune system, higher sanitation standards might be in part responsible for a decrease in early exposure to microbial agents.52 The resulting impaired maturation of immune system in early life and altered microbial flora, that does not allow the normal shift towards a predominant TH1 response, leads to an increase of allergic and atopic diseases.53 Moreover, strict hygiene practices may lead to inadequate establishment of the gut microbiota and an increase in the incidence of allergic disease.44
ConclusionThis review aimed to discuss the current evidence regarding the short and long-term respiratory health effects of nutrients and dietary patterns during pregnancy, breastfeeding and complementary feeding and the potential underlying mechanisms. It should be pointed out that it was not strictly designed a priori as a systematic review, and that the PRISMA Statement was not fully applied (including PICOS). Although this is a weakness, a descriptive approach (not including numerical values of principal summary measures) might benefit the reader with a promptly and friendly readable update of the matter.
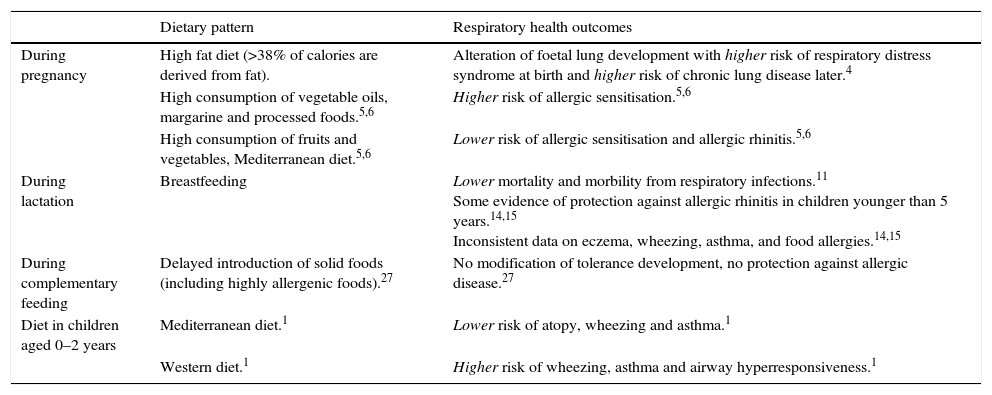
Diet and nutrition, thanks to their capacity to regulate the immune system, are modifiable behavioural risk factors influencing the respiratory disease development and progression (Table 3). Nutrition may modulate gene expression through epigenetic effects but also the so called gut-airway axis, associated with the inflammatory status of respiratory tract.
Dietary patterns and respiratory health outcomes during the first 1000 days of life.
| Dietary pattern | Respiratory health outcomes | |
|---|---|---|
| During pregnancy | High fat diet (>38% of calories are derived from fat). | Alteration of foetal lung development with higher risk of respiratory distress syndrome at birth and higher risk of chronic lung disease later.4 |
| High consumption of vegetable oils, margarine and processed foods.5,6 | Higher risk of allergic sensitisation.5,6 | |
| High consumption of fruits and vegetables, Mediterranean diet.5,6 | Lower risk of allergic sensitisation and allergic rhinitis.5,6 | |
| During lactation | Breastfeeding | Lower mortality and morbility from respiratory infections.11 Some evidence of protection against allergic rhinitis in children younger than 5 years.14,15 Inconsistent data on eczema, wheezing, asthma, and food allergies.14,15 |
| During complementary feeding | Delayed introduction of solid foods (including highly allergenic foods).27 | No modification of tolerance development, no protection against allergic disease.27 |
| Diet in children aged 0–2 years | Mediterranean diet.1 | Lower risk of atopy, wheezing and asthma.1 |
| Western diet.1 | Higher risk of wheezing, asthma and airway hyperresponsiveness.1 |
A strong link between maternal nutrition and health of the offspring exists: attention to adequate nutrition should begin during the pre-conception period and be continued during pregnancy and lactation. Unhealthy maternal nutrition influences placental development and foetal growth; a high fat diet can result in placental insufficiency, foetal growth restriction and alteration of foetal lung development, which in turn can increase the risk of respiratory disorders. Breastfeeding should be encouraged as the normal nutrition for the infants. For the moment it is difficult to draw definitive conclusions about the beneficial effect of breastfeeding on asthma. Anyway breastfeeding should be encouraged because of its several benefits, including pulmonary infections. In order to prevent the development of allergies in the offspring, maternal dietary restrictions are not recommended and in infants there is no evidence to avoid or encourage exposition to ‘highly allergenic’ foods during complementary feeding.
In conclusion, attention to adequate nutrition in the first 1000 days is very important for the respiratory health. Mediterranean Diet represents the cornerstone of a correct dietary pattern for the pregnant and breastfeeding woman and for the children aged 0–2 years. For what concern micronutrients, antioxidant and LCPUFA a whole food approach to nutrients should be preferred with respect to oral supplementation, except for vitamin D.
Conflict of interestAll the authors declare that they have no conflict of interest or financial support for this study.
Ethical disclosuresConfidentiality of dataThe authors declare that no patient data appears in this article.
Right to privacy and informed consentThe authors declare that no patient data appears in this article.
Protection of human subjects and animals in researchThe authors declare that no experiments were performed on humans or animals for this investigation.