Infants are born immunologically immature. However, breastfeeding mothers retain an immunological link to their infants. While it is generally accepted that infants are at an immunological advantage when compared with formula-fed infants, the benefit of long-term exclusive breastfeeding by atopic mothers remains controversial. Inconsistency in the conferral of benefit may be due to differences in the immunological constituents passed to the recipient infant. The aim of this investigation was to examine the profile of human milk cells and cytokines from asthmatic compared to non-asthmatic mothers.
MethodsTwenty-five exclusively breastfeeding mothers with a clinical diagnosis of asthma were postpartum age matched in a double-control 2:1 design with 50 non-asthmatic controls. Each mother provided a single milk sample which was assayed for cell differential by flow cytometry, for ex vivo cytokine production in culture and for aqueous phase cytokines.
ResultsMilks from asthmatic mothers differed from non-asthmatics in that they contained a higher proportion of polymorphonuclear (PMN) cells and lower proportion of lymphocytes, predominantly CD3+/CD4+ T helper cells, reflected by a decrease in the chemokine CCL5 in the milk aqueous phase. More PMN and lymphocytes from asthmatic mothers expressed the adhesion molecule CD11b and lymphocytes the IgE receptor CD23, than those from non-asthmatic mothers.
ConclusionsChanges to human milk leucocyte prevalence, activation state and cytokines due to maternal asthma may result in changes to immunological priming in the infant. Consequently, the protective effect of long-term breastfeeding may be altered in these mother-infant pairs.
Breastfed infants are at an immunological advantage when compared with formula fed infants, suggesting a biological link between a mother and her infant, at least in part due to the factors supplied in the milk. However, the protective role of breastfeeding against the development of atopic disease in the recipient infant remains controversial, particularly in the context of prolonged breastfeeding by atopic mothers.1
Atopy influences systemic leucocyte populations as well as significantly altering the cytokine profiles produced by immunoactive cells. The peripheral blood mononuclear cells of atopic adults produce higher levels of T helper (Th) 2 cytokines, and reduced levels of Th1 cytokines, demonstrating a Th2 bias.2 While T lymphocytes from infants born to atopic parents have upregulated expression of Th2 cytokines when compared with infants born to non-atopic parents, studies have generally found a greater influence of maternal than paternal atopy, supporting a potential influence of pregnancy and breastfeeding.3,4 This apparent dysregulation may similarly affect the immunological components of human milk, with small studies having previously reported elevated levels of aqueous phase interleukin (IL)-4, IL-5, IL-8 (CXCL8) and CCL5 (RANTES) in milk from atopic mothers.5
Milk from mothers of allergic infants has demonstrated differences in both leucocyte prevalence and activation from that of non-allergic infants, with elevated proportions of neutrophils and eosinophils, fewer monocytes/macrophages expressing diminished HLA-DR, more B lymphocytes, fewer cytotoxic/suppressor CD8+ T lymphocytes, and a significantly larger proportion of the total lymphocytes expressing the low affinity IgE receptor (CD23+).6–8 However, there have been no studies that have examined the effects of maternal atopy on either the proportion of cell types or the activation state of these cells in human milk.
We therefore aimed to investigate the cellular composition of human milk, including the expression of activation markers and cytokine production in response to allergic stimuli, in clinically diagnosed asthmatic mothers.
MethodsParticipantsThe project was approved by the Southern Adelaide Clinical Human Research Ethics Committee and conducted in accordance with the National Statement on Ethical Conduct in Human Research. Breastfeeding mothers >18 years and with infants <12 months of age with self-reported, clinically-diagnosed asthma requiring regular use of bronchodilators, were post-partum age matched against non-asthmatic controls in a 1:2 design and attended an assessment clinic at Flinders Medical Centre, Australia. Data were collected relating to maternal and infant health during the preceding week and on the day of sample collection, including any medication used. All participants were asked to refrain from taking any non-essential medication or from smoking prior to the clinic.
A venous blood sample (5mL) was collected for determination of total serum IgE using the Imx Total IgE assay (IU/mL; 1IU=2.4ng referenced against WHO-IgE standards; Abbott Laboratories, Abbott Park, IL, USA).
Sample collectionFollowing a minimum one hour feeding abstinence, in the morning, a complete expression from one breast was collected into sterile containers using an electric pump (Ameda Hollister, Chicago, IL, USA). All samples were processed within 15min from collection by centrifugation (900×g, 30min), the fat removed and aqueous phase aliquoted and stored at −80°C, until analysis by ELISA. The cell pellet was washed in PBS before resuspension in sterile RPMI 1640 with 10% foetal bovine serum (FBS) (CSL Biosciences, Australia) and total viable cell number determined by direct microscopy with trypan blue staining, as previously. 9
Flow cytometryCells were stained with fluorescently-labelled monoclonal antibodies for the determination of leucocyte subsets, as well as expression of markers of cellular activation, as previously.10 Briefly, cells (1×106) were incubated at 4°C for 15min with directly-conjugated phycoerythrin (PE)-Cy 5, PE or FITC mouse monoclonal IgG antibodies before analysis on a FACScan flow cytometer (Becton Dickinson, San Jose, CA, USA). Each sample was live gated to collect 1×104 CD45+ events and cells differentiated using CD45 fluorescence and sidescatter parameters.
Cell cultureTotal cells were cultured (2.5×106cells/mL) in RPMI, as above, in the presence or absence of 150μg/mL Con A, 4mM histamine or 100μg/ml ovalbumin grade V (Sigma–Aldrich, MO, USA) for 48h at 37°C in 5% CO2. Cells were lysed by a single freeze-thaw cycle (−80°C) and the medium harvested, centrifuged (900×g, 5min) to pellet cell debris, aliquoted and stored at −80°C until analysis by ELISA.
ELISAIFN-γ, IL-2, IL-4, IL-10, CXCL8 (IL-8) and CCL5 (RANTES) (R&D Systems Inc, Minneapolis, MN, USA) were measured in milk aqueous phase, serum and cell culture supernatants, as described previously.9 The minimum detectable level for each assay was 8, 8, 5, 5, 20 and 5pg/mL, respectively. Samples below this were assigned a value of half that level for statistical analysis. All samples were analysed within six weeks of storage at −80°C.
StatisticsNumerical data are expressed as mean±standard deviation and analysed by Independent t test following log transformation when necessary, and categorical variables as number (%), analysed by Pearson X2. Associations were examined using Pearson correlation. A probability of ≤0.05 was considered statistically significant and all analyses were performed using SPSS for Windows 22.0 (SPSS Inc. Chicago, IL, USA).
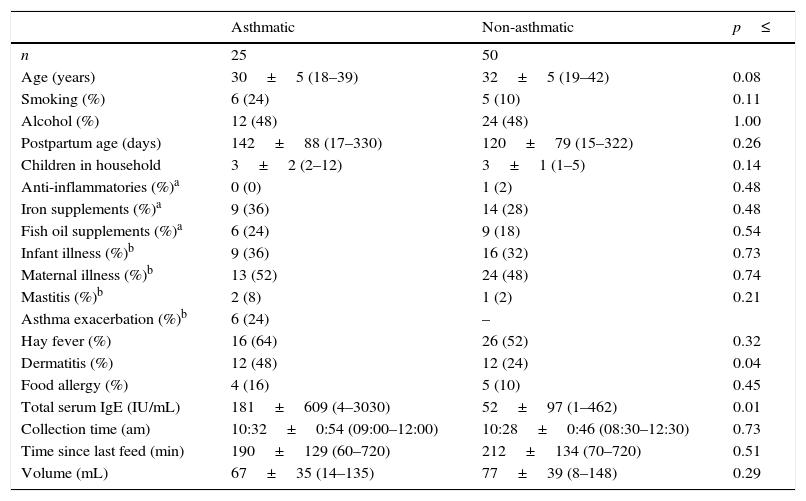
ResultsSubject and sample characteristicsMothers who participated in the study did not differ in any assessed characteristic other than those of dermatitis and serum IgE, which may indicate a predominance of atopic IgE mediated asthmatic disease (p=0.004; Table 1). Despite this, 32 non-asthmatic women reported having some form of allergic disease other than asthma (rhinoconjunctivitis, dermatitis, food allergy), 26 of whom required medication (e.g. anti-histamine).
Subject and expressed human milk sample characteristics.
| Asthmatic | Non-asthmatic | p≤ | |
|---|---|---|---|
| n | 25 | 50 | |
| Age (years) | 30±5 (18–39) | 32±5 (19–42) | 0.08 |
| Smoking (%) | 6 (24) | 5 (10) | 0.11 |
| Alcohol (%) | 12 (48) | 24 (48) | 1.00 |
| Postpartum age (days) | 142±88 (17–330) | 120±79 (15–322) | 0.26 |
| Children in household | 3±2 (2–12) | 3±1 (1–5) | 0.14 |
| Anti-inflammatories (%)a | 0 (0) | 1 (2) | 0.48 |
| Iron supplements (%)a | 9 (36) | 14 (28) | 0.48 |
| Fish oil supplements (%)a | 6 (24) | 9 (18) | 0.54 |
| Infant illness (%)b | 9 (36) | 16 (32) | 0.73 |
| Maternal illness (%)b | 13 (52) | 24 (48) | 0.74 |
| Mastitis (%)b | 2 (8) | 1 (2) | 0.21 |
| Asthma exacerbation (%)b | 6 (24) | – | |
| Hay fever (%) | 16 (64) | 26 (52) | 0.32 |
| Dermatitis (%) | 12 (48) | 12 (24) | 0.04 |
| Food allergy (%) | 4 (16) | 5 (10) | 0.45 |
| Total serum IgE (IU/mL) | 181±609 (4–3030) | 52±97 (1–462) | 0.01 |
| Collection time (am) | 10:32±0:54 (09:00–12:00) | 10:28±0:46 (08:30–12:30) | 0.73 |
| Time since last feed (min) | 190±129 (60–720) | 212±134 (70–720) | 0.51 |
| Volume (mL) | 67±35 (14–135) | 77±39 (8–148) | 0.29 |
Numerical data are presented as mean±SD (min–max) and analysed by Independent t test following log transformation when necessary, and categorical variables as number (%), analysed by Pearson X2.
Illness, mastitis and asthma exacerbation were based on maternal report of any incidence over the proceeding seven days, not clinical diagnosis and did not differ between the study groups. Despite six reported asthma exacerbations and 23 reports of active respiratory illness on the day of milk sample collection, 18 requiring medication; only one non-asthmatic mother reported use of anti-inflammatories during the week preceding sample collection.
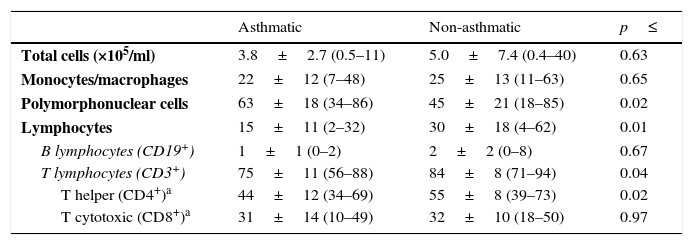
Leukocytes in human milkThe total number of cells per mL of human milk was not significantly different between asthmatic and non-asthmatic mothers, however the relative proportion of polymorphonuclear cells was elevated and lymphocytes decreased in the milk from asthmatic mothers (Table 2; p≤0.02). The decrease in lymphocyte prevalence was found to be predominantly due to decreased T lymphocytes, specifically CD45+/CD3+/CD4+ T helper cells (p≤0.04).
Leukocytes in expressed human milk.
| Asthmatic | Non-asthmatic | p≤ | |
|---|---|---|---|
| Total cells (×105/ml) | 3.8±2.7 (0.5–11) | 5.0±7.4 (0.4–40) | 0.63 |
| Monocytes/macrophages | 22±12 (7–48) | 25±13 (11–63) | 0.65 |
| Polymorphonuclear cells | 63±18 (34–86) | 45±21 (18–85) | 0.02 |
| Lymphocytes | 15±11 (2–32) | 30±18 (4–62) | 0.01 |
| B lymphocytes (CD19+) | 1±1 (0–2) | 2±2 (0–8) | 0.67 |
| T lymphocytes (CD3+) | 75±11 (56–88) | 84±8 (71–94) | 0.04 |
| T helper (CD4+)a | 44±12 (34–69) | 55±8 (39–73) | 0.02 |
| T cytotoxic (CD8+)a | 31±14 (10–49) | 32±10 (18–50) | 0.97 |
Each sample was live gated to collect 1×104 CD45+ events and cell types differentiated using CD45 fluorescence and sidescatter parameters. Data are presented as mean±SD (min–max) %CD45+ gated on side-scatter parameters.
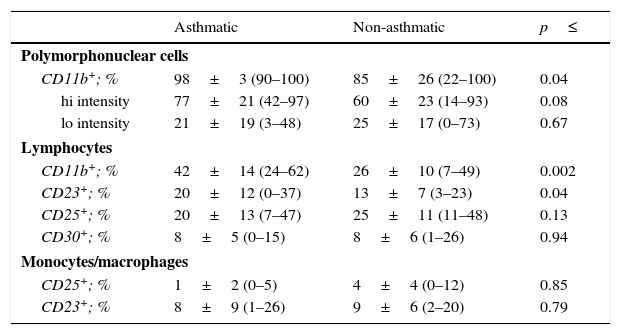
Examination of the activation state of these cells demonstrated an increase in CD11b+ polymorphonuclear cells in milks from asthmatic mothers (Table 3; p=0.04), with a trend towards higher intensity of expression in these cells (p=0.08). Similarly, the percentage of lymphocytes expressing CD11b was also higher in asthmatic when compared to non-asthmatic mothers (p=0.002). A greater proportion of lymphocytes from asthmatic mothers were also found to express the low-affinity IgE receptor, CD23 (p=0.04).
Leucocyte activation markers in expressed human milk.
| Asthmatic | Non-asthmatic | p≤ | |
|---|---|---|---|
| Polymorphonuclear cells | |||
| CD11b+; % | 98±3 (90–100) | 85±26 (22–100) | 0.04 |
| hi intensity | 77±21 (42–97) | 60±23 (14–93) | 0.08 |
| lo intensity | 21±19 (3–48) | 25±17 (0–73) | 0.67 |
| Lymphocytes | |||
| CD11b+; % | 42±14 (24–62) | 26±10 (7–49) | 0.002 |
| CD23+; % | 20±12 (0–37) | 13±7 (3–23) | 0.04 |
| CD25+; % | 20±13 (7–47) | 25±11 (11–48) | 0.13 |
| CD30+; % | 8±5 (0–15) | 8±6 (1–26) | 0.94 |
| Monocytes/macrophages | |||
| CD25+; % | 1±2 (0–5) | 4±4 (0–12) | 0.85 |
| CD23+; % | 8±9 (1–26) | 9±6 (2–20) | 0.79 |
Each sample was live gated to collect 1×104 CD45+ events and cell types differentiated using CD45 fluorescence and sidescatter parameters. Data are presented as mean±SD (min–max). Variables were tested for normality by Shapiro–Wilkes test and log transformed when necessary before analysis by Independent t test.
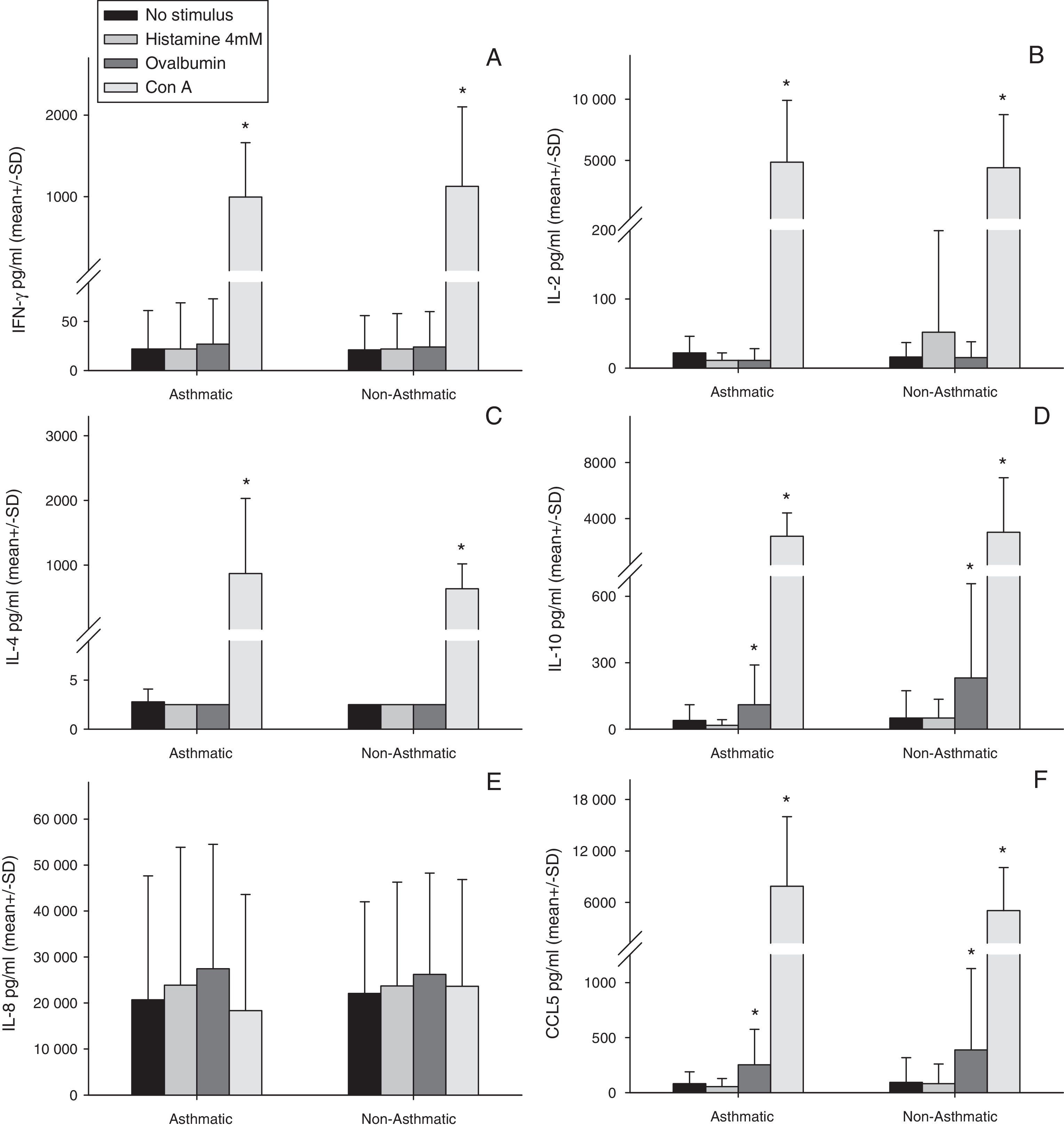
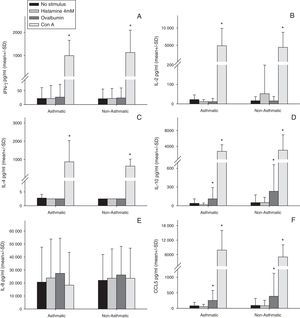
Stimulation of human milk cells from both asthmatic and non-asthmatic mothers with Con A for 48h significantly increased expression of all cytokines examined above constitutive, with the exception of IL-8 (Fig. 1). Ovalbumin stimulation increased both IL-10 and CCL5 above constitutive from cells of both asthmatic and non-asthmatic mothers (Fig. 1 D&F). There was no increase in all other cytokines following ovalbumin stimulation, nor was there any effect of 48h incubation with histamine in either of the groups.
Cytokine production by total cell isolates from expressed breast milk (EBM) samples at 48h of culture ex vivo. Data were compared between asthmatic and non-asthmatic mothers by Independent t test and between each stimulant and non-stimulated control by paired t test. * Difference from unstimulated cells by Paired t test (p≤0.05). Con A for 48h significantly increased expression of all cytokines above constitutive, with the exception of IL-8. Ovalbumin stimulation increased both IL-10 and CCL5 above constitutive. There was no increase in all other cytokines following ovalbumin stimulation, nor was there any effect of 48h incubation with histamine in either of the groups. There were no statistically significant effects of maternal asthma on cytokine production by human milk cells either constitutively or in response to any stimulant.
There were no statistically significant effects of maternal asthma on cytokine production by human milk cells either constitutively or in response to any stimulant (Fig. 1A–F).
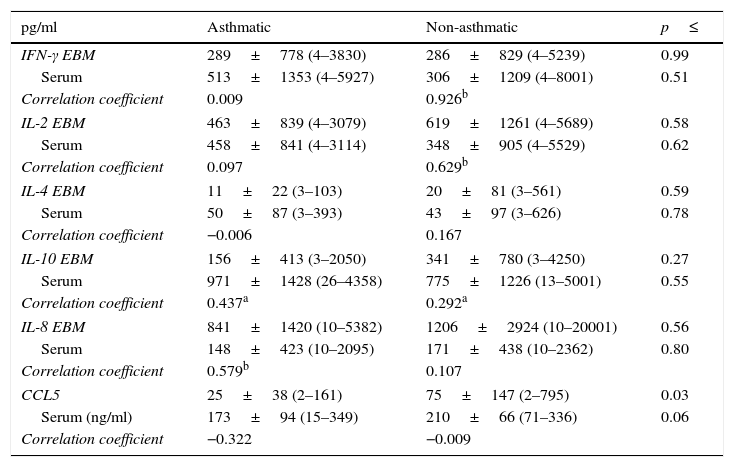
Aqueous phase and serum cytokinesThe concentration of the chemokine CCL5 was significantly lower in the aqueous phase of milks from asthmatic mothers when compared to non-asthmatic mothers (Table 4; p=0.03), which was reflected in a trend towards lower serum CCL5 in this group (p=0.06). There was no statistically significant difference in the concentration of any other aqueous phase cytokine examined in milk between asthmatic and non-asthmatic mothers.
Soluble cytokines in expressed human milk and serum.
| pg/ml | Asthmatic | Non-asthmatic | p≤ |
|---|---|---|---|
| IFN-γ EBM | 289±778 (4–3830) | 286±829 (4–5239) | 0.99 |
| Serum | 513±1353 (4–5927) | 306±1209 (4–8001) | 0.51 |
| Correlation coefficient | 0.009 | 0.926b | |
| IL-2 EBM | 463±839 (4–3079) | 619±1261 (4–5689) | 0.58 |
| Serum | 458±841 (4–3114) | 348±905 (4–5529) | 0.62 |
| Correlation coefficient | 0.097 | 0.629b | |
| IL-4 EBM | 11±22 (3–103) | 20±81 (3–561) | 0.59 |
| Serum | 50±87 (3–393) | 43±97 (3–626) | 0.78 |
| Correlation coefficient | −0.006 | 0.167 | |
| IL-10 EBM | 156±413 (3–2050) | 341±780 (3–4250) | 0.27 |
| Serum | 971±1428 (26–4358) | 775±1226 (13–5001) | 0.55 |
| Correlation coefficient | 0.437a | 0.292a | |
| IL-8 EBM | 841±1420 (10–5382) | 1206±2924 (10–20001) | 0.56 |
| Serum | 148±423 (10–2095) | 171±438 (10–2362) | 0.80 |
| Correlation coefficient | 0.579b | 0.107 | |
| CCL5 | 25±38 (2–161) | 75±147 (2–795) | 0.03 |
| Serum (ng/ml) | 173±94 (15–349) | 210±66 (71–336) | 0.06 |
| Correlation coefficient | −0.322 | −0.009 | |
Data are presented as mean±SD (min–max). Variables were tested for normality by Shapiro–Wilkes test and log transformed when necessary before analysis by Independent t test. Bivariate correlation was examined within each group by Pearson's Correlation.
There was significant correlation between IL-10 in the aqueous fraction of the milk samples and the matching serum sample in both asthmatic and non-asthmatic mothers; however, this relationship was stronger in the asthmatic group (p≤0.04). Further correlations between aqueous phase and serum cytokines were group specific with IL-8 correlated in the asthmatic group and the Th 1 cytokines IFN-γ and IL-2 in the non-asthmatic group (p≤0.003). Analysis of the relationship between aqueous phase cytokines and constitutive cellular production in culture ex vivo was only found to be IL-8 correlated (Correlation coefficient 0.650, p≤0.001).
DiscussionHuman infants are born immunologically compromised, but those who are breastfed are at an immunological advantage when compared with formula fed infants, as evidenced by both decreased incidence of infections during infancy and diminished propensity for a number of long term conditions.11 However, the relationship between human milk feeding and protection against the development of atopy is uncertain and often controversial. Despite studies and epidemiology suggesting a protective association between breastfeeding and the development of atopic disease, reiterated by systematic reviews and meta-analyses, studies which have indicated a potential for increased risk, particularly with longer duration of exclusive breastfeeding by atopic mothers, continue to raise questions.1 While these conflicting results could be due to protocol differences and heterogeneity, or confounding due to mothers extending breastfeeding duration due to indications of allergy, the possibility that they may be due to differences in the milk of individual mothers warrants investigation.12
Human milk has the potential to affect the development of atopy in the recipient infant by a number of mechanisms. The transfer of allergens in human milk is one area under substantial scrutiny with maternal dietary avoidance of potentially allergenic substances being undertaken in many trials. However, the Cochrane Reviews of this subject conclude that allergen avoidance diet does not conclusively decrease the incidence of allergic disease.13,14 Alternatively, immunoactive constituents in the milk of mothers may be exerting effects on the development of infant atopy, either directly during allergen exposure or more generally through enhanced maturation of the immune system.15
Changes to human milk leucocyte prevalence, activation state and cytokines due to maternal asthma, as demonstrated in this study, have the potential to result in changes to immunological priming in the infant. Infants who were breastfed have peripheral blood lymphocyte prevalence more akin to that of an adult, and elicit enhanced responses to vaccination when compared to formula fed infants of the same age.16–18 While the source within the mother of human milk cells has not been adequately identified, there is evidence that at least some of the cells have homed directly from mucosal sites following activation by intestinal pathogens, with the second major source either directed or non-specific migration from peripheral blood.19 Therefore, it may be hypothesised that a proportion of the cells passed to the recipient infant in human milk may retain a differentiation bias induced by systemic conditions such as asthma.
CD23 plays a role in the regulation of IgE synthesis demonstrated through increased IgE production following CD23 knockout and decreased production with CD23 overexpression.20 However, the milk from mothers of cow's milk allergic infants has been found to contain fewer T lymphocytes with a larger proportion of these lymphocytes expressing the low affinity IgE receptor (CD23+), raising the implication of a protective, causal effect on the development of allergic disease in infancy due to the cellular milieu received via breastfeeding.6 This profile of decreased numbers of lymphocytes, with more expressing CD23 was reflected in our study, however, the parallel findings of Jarvinen et al. that the predominant change in T lymphocytes involved decreasing cytotoxic CD8+ and that there was an increase in B lymphocytes, was not.6 Rather, in our cohort of asthmatic mothers a decrease in the T helper CD4+ population was responsible for the decrease in T cells and very few B lymphocytes were found. As we did not follow up the infants of the mothers involved in our study we can make no associations between the changes observed in the lymphocyte populations and subsequent development of allergic disease. However, it should be noted that Jarvinen et al. found no difference in the cell differentials between atopic and non-atopic women in their cohort.
Alteration in the lymphocyte differential in milks from asthmatic mothers in our study was offset by an increase in polymorphonuclear cells. Although not specifically labelled, these cells are likely to be predominantly neutrophils, with a small contribution of eosinophils.21 Exacerbations of asthma can result in increased number of activated neutrophils in both peripheral blood and airways, associated with increased expression of CD11b.22 This is particularly apparent in peripheral blood of asthmatics presenting with a delayed rather than immediate response to allergen, so may reflect the trafficking of more mature neutrophils or, as the lymphocyte population was also found to express CD11b, may indicate an adhesion mechanism through which these cells are taken up into the mammary gland.23
Contention remains as to the functional potential of human milk cells within the recipient system. Cells isolated from human milk retain the ability to constitutively produce a number of cytokines ex vivo which has been further demonstrated here, and previously, to be increased after stimulation with many mitogens, although not to the same levels as peripheral blood cells.24,25 Alteration in cytokine production by human milk cells, as well as difference in aqueous phase cytokines, have been associated with development of allergy in the infant.26–29 Similarly, we have previously demonstrated significant alteration in cytokine production in response to infant illness, suggesting communication between infant and mother.9
Few studies, until now, have examined changes to the cytokines in human milk in response to atopic disease in the mother.5 However, there does appear to be a pattern of higher levels of some of the classical Th2-type cytokines IL-4, IL-5 and possibly IL-13, and in the chemokines, IL-8 and CCL5 in the aqueous phase of milk samples from atopic women, particularly asthmatics, when compared to those of non-atopic controls, but this is inconsistent. Both these and independent studies examining Th1 cytokine concentration have found no difference in the concentration of these cytokines based on atopic status. Inconsistency in a definitive Th2/Th1 imbalance is also found ex vivo, whereby allergenic stimulation of human milk cells isolated from milks of allergic versus non-allergic mothers produced higher levels of Th2 cytokines IL-5 and IL-13, but lower IL-4, and lower Th1 cytokine IFN-γ but higher IL-10.30 While many of these studies did not differentiate between allergic diseases or severity of each, our study which examined a cohort of clinically diagnosed asthma did not demonstrate any of these previously reported differences. The single exception in our study was a decrease in the chemokine CCL5, which parallels the decrease in lymphocyte infiltration, but differs from the previous report of increased CCL5.31 Chemokines play an important role in allergic disease through the influx of cells responsible for both the release of preformed granules capable of increasing the allergic inflammatory response, as well as the release of cytokines such as IL-4 and IL-5, which promote the development of the Th2 IgE response.32 While the chemokine IL-8 was not increased in parallel with the increase in PMN in our asthmatic cohort, the discordance between milk and serum IL-8 in the non-asthmatic subjects, due in part to a few extremely high outliers in the milk of some, may indicate an alternative unreported pathology in the mammary gland of this group.
The measurement of systemic cytokine levels has allowed for examination of correlations between aqueous phase of human milk and these cytokines in matched serum samples. We previously reported no correlation between the levels of TGF-β, but positive correlation between the levels of IL-2, in milk and matched plasma/serum samples from healthy women.33,34 Relationships were found again here with IFN-g, IL-2, IL-10 and IL-8, however with the exception of IL-10 this was not consistent between asthmatic and non-asthmatic mothers. Therefore, while it appears that some systemic cytokine levels and those in the mammary gland may be related in healthy individuals, the levels found in these fluids are more often discordant in the presence of disease. This may imply differing primary sources of cytokines in peripheral blood when compared to those in human milk, or in the level of activation of cells present in the mammary gland of atopic women, suggested by the correlation of IL-8 in asthma, in parallel with the increase in neutrophils in the milk.
Limitations to the study are largely due to potential confounding factors which are represented in higher than desired numbers, for example, smoking, maternal illness and allergic diseases in the non-asthmatic group. However, there was no difference in the incidence of these variables between the study groups, with the exception of dermatitis, and post hoc analysis did not identify any of these factors as significant confounders. A further limitation may also be evident in the lack of steroid use in the week prior to sample collection among our asthmatic cohort. This may indicate either that our group of asthmatic mothers could be classified as mild to moderate asthma, or that they have been non-compliant with their medication, possibly through avoidance due to breastfeeding.
ConclusionThe changes to human milk leucocyte prevalence, activation state and cytokines due to maternal asthma described by this study may result in changes to immunological priming in the infant. Whether these changes may contribute through modulation of immune system development or through direct effects during allergen exposures requires extensive further research. In addition, the effect of these differences in the context of the inherited genetic and epigenetic background of the recipient infant should also be investigated. However, the finding of immunological differences in the components of human milk delivered to infants of asthmatic mothers may address some of the inconsistency between studies as the protective effect of long-term breastfeeding may be altered in these mother-infant pairs.
Ethical disclosuresConfidentiality of dataThe authors declare that they have followed the protocols of their work centre on the publication of patient data and that all the patients included in the study have received sufficient information and have given their informed consent in writing to participate in that study.
Right to privacy and informed consentThe authors have obtained the informed consent of the patients and/or subjects mentioned in the article. The author for correspondence is in possession of this document.
Protection of human subjects and animals in researchThe authors declare that the procedures followed were in accordance with the regulations of the responsible Clinical Research Ethics Committee and in accordance with those of the World Medical Association and the Helsinki Declaration.
Conflict of interestThe authors have no conflicts to declare.
The authors would like to thank the Channel 7 Children's Research Foundation and the Flinders Medical Centre Foundation which funded this research.