In Phase Three of the International Study of Asthma and Allergies in Childhood (ISAAC), we investigated the relationship between breast feeding in infancy and symptoms of asthma, rhinoconjunctivitis and eczema in 6–7 year old children.
MethodsParents or guardians of 6–7 year old children completed written questionnaires on current symptoms of asthma, rhinoconjunctivitis and eczema, and on a range of possible asthma risk factors including a history of breast feeding ever. Prevalence odds ratios were estimated using logistic regression, adjusted for gender, region of the world, language, per capita gross national income, and other risk factors.
ResultsIn all 206,453 children from 72 centres in 31 countries participated in the study. Reported breast feeding ever was not associated with current wheeze, with an odds ratio (adjusted for gender, region of the world, language, per capita gross national income, and factors encountered in infancy) of 0.99 (95% CI 0.92–1.05), current rhinoconjunctivitis (OR 1.00, 95% CI 0.93–1.08), current eczema (OR 1.05, 95% CI 0.97–1.12), or symptoms of severe asthma (OR 0.95, 95% CI 0.87–1.05). Breast feeding was however associated with a reduced risk of severe rhinoconjunctivitis (OR 0.74, 95% CI 0.59–0.94) and severe eczema (OR 0.79, 95% CI 0.66–0.95).
ConclusionsThere was no consistent association between breast feeding use in the first year of life and either a history or current symptoms of wheezing, rhinoconjunctivitis or eczema in 6–7 year old children, but possibly an effect on severe symptoms of the latter two conditions.
It has been debated for 50 years whether low rates of breast feeding are associated with asthma, wheezing and other manifestations of allergy.1–3 The controversy is compounded by poorly defined phenotypes, different populations studied, and the impact of breast feeding on infections and immunity which are profoundly affected by numerous influences from the environment. Breast feeding is essential for protection against infections in babies living under poor hygienic conditions in developing countries.4,5 Furthermore, asthma symptoms are most commonly caused by infections6 especially in young children,7 while in older children allergy is a common cause in affluent but not in non-affluent countries.8,9 Similarly, rhinitis in some countries is mainly due to infections, while hay fever is the major contributor in other countries.8,10 Very recently, Nagel et al. reported breast feeding to be associated with a lower prevalence of non-atopic wheeze among school children living in low income, but not in high income countries.10 Breast feeding was not related to any measure of allergy.
We have extended these studies to a global analysis and here report the association between a history of breast feeding and symptoms of asthma, rhinoconjunctivitis and eczema in 6–7 year old children from Phase Three of the ISAAC study.
MethodsPhase Three of ISAAC is a multi-centre, multi-country, cross-sectional study of two age groups of schoolchildren (6–7 year old children and 13–14 year old adolescents) chosen from a random sample of schools in a defined geographical area.11 The Phase Three survey included two standardised questionnaires. The first questionnaire obtained data on symptoms of asthma, rhinoconjunctivitis and eczema, and was identical to that used in Phase One of the ISAAC study.12–14 The second questionnaire asked about a wide range of possible environmental risk factors for the development of asthma and allergic disorders.15 The current analysis is restricted to the younger age-group, since this involved parental reporting and it was therefore possible to ask about exposures in the first year of life. In addition to questions related to current exposure and socioeconomic conditions, the questionnaire included questions on birth order, birth weight, maternal smoking during the infancy of the child, breast feeding, immigration status, antibiotic and paracetamol use in the first year of life, as well as exposure to cat and dog at home and regular contact with farm animals in the first year. The complete questionnaire can be found on the ISAAC website (http://isaac.auckland.ac.nz).
The question relating to breast feeding was: “Was your child breast fed?” (Yes/No).
Symptoms of ‘current wheeze’ were determined by positive answers to the written question: “Has your child had wheezing or whistling in the chest in the past 12 months?” (Yes/No).
Current symptoms of rhinoconjunctivitis were determined by positive answers to two questions: “In the past 12 months has your child had a problem with sneezing or a runny or blocked nose when he/she DID NOT have a cold or the flu?” (Yes/No). If yes, “In the past 12 months has this nose problem been accompanied by itchy watery eyes?” (Yes/No).
Current symptoms of eczema were determined by positive answers to two questions: “Has your child had this itchy rash at any time in the past 12 months?” (Yes/No) (This question was preceded by the question “Has your child ever had an itchy skin rash which was coming and going for at least 6 months?” [Yes/No]). If yes, “Has this itchy rash at any time affected any of the following places – the folds of the elbows, behind the knees, in front of the ankles, under the buttocks, or around the neck, ears or eyes?” (Yes/No).
Current symptoms of severe wheeze were defined as current wheeze (as above) with four or more attacks of wheezing in the past 12 months, or wheezing causing sleep disturbance one or more nights per week in the last 12 months, or wheezing limiting speech in the last 12 months. Current symptoms of severe rhinoconjunctivitis were defined as current symptoms of rhinoconjunctivitis (as above) with nose symptoms affecting activities “a lot” in the last 12 months. Current symptoms of severe eczema were defined as current symptoms of eczema (as above) with rash causing sleep disturbance one or more nights per week in the last 12 months.
Although the specific terms “asthma”, “hay fever” and “eczema” are interpreted differently in different countries, questions about these conditions ever were also included in the questionnaires.
Data analysisTo be included in the analysis, centres were required to have a participation rate of >70%.15 Prevalence odds ratios (ORs)16 were calculated, using generalised linear mixed models with a binomial distribution and a logit link and with the centres being modelled as a random effect. All analyses were adjusted for gender, region of the world (nine categories), language (eight categories) and per capita gross national income (four categories). The regression models allowed for the sampling by schools by scaling the size of the sample by the design effect. Regions of the world were Africa, Asia Pacific, Eastern Mediterranean, Latin America, North America, Northern and Eastern Europe, Oceania, Southeast Asia/Indian Subcontinent, Western Europe. The eight language categories reflected the proportions of their use: Arabic, Chinese, English, Hindi, Indonesian, Portuguese, Spanish and other (comprising many less frequently used languages). Centres were allocated according to socioeconomic status based on their country's gross national income categorised by the World Bank as low, lower-middle, upper-middle, and upper.17
Further multiple regression analyses were conducted to investigate whether the association between symptoms and breast feeding were confounded by other risk factors for which information was collected in the environmental questionnaire, and which had shown associations with wheezing, rhinoconjunctivitis or eczema in univariate analyses. For inclusion in these further analyses, centres were required to have at least 70% data available for all covariates. The covariates included in these multiple regression analyses were birth weight (less than or greater than 3200g), maternal education (four categories – none, primary, secondary, and tertiary), maternal smoking during the child's first year of life (Yes/No), a cat or dog at home during the first year of life, antibiotic use in the first year of life, paracetamol use in the first year of life, whether the child had one or more older siblings, maternal exposure to farm animals during pregnancy (Yes/No) and regular exposure of the child during the first year of life to farm animals (Yes/No). All analyses were carried out with SAS (version 9, SAS Institute Inc., Cary, NC, USA).
ResultsIn ISAAC Phase Three, 75 centres from 32 countries (220,408) included some or all of the environmental questionnaire for the 6–7 year age group. However, we excluded centres which did not include the breast feeding question (three centres). Thus, there were 206,453 children from 72 centres in 31 countries included in the analyses.
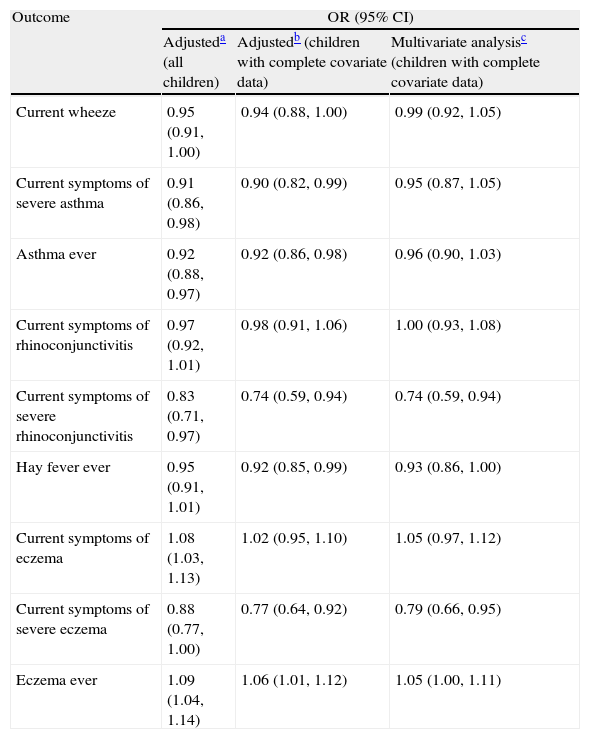
The associations between symptoms (adjusted for gender, region of the world, language and per capita gross national income) and reported breast feeding are shown in Table 1 and Fig. 1. There was no association with current wheeze with an odds ratio of 0.95 (95% CI 0.91–1.00), nor current rhinoconjunctivitis (OR 0.97, 95% CI 0.92–1.01). Current eczema, however, was slightly more common in children who had been breast-fed (OR 1.08, 95% CI 1.03–1.13).
Association between breast feeding and current symptoms of asthma, rhinoconjunctivitis, and eczema in children 6–7 years old.
| Outcome | OR (95% CI) | ||
| Adjusteda (all children) | Adjustedb (children with complete covariate data) | Multivariate analysisc (children with complete covariate data) | |
| Current wheeze | 0.95 (0.91,1.00) | 0.94 (0.88,1.00) | 0.99 (0.92,1.05) |
| Current symptoms of severe asthma | 0.91 (0.86,0.98) | 0.90 (0.82,0.99) | 0.95 (0.87,1.05) |
| Asthma ever | 0.92 (0.88,0.97) | 0.92 (0.86,0.98) | 0.96 (0.90,1.03) |
| Current symptoms of rhinoconjunctivitis | 0.97 (0.92,1.01) | 0.98 (0.91,1.06) | 1.00 (0.93,1.08) |
| Current symptoms of severe rhinoconjunctivitis | 0.83 (0.71,0.97) | 0.74 (0.59,0.94) | 0.74 (0.59,0.94) |
| Hay fever ever | 0.95 (0.91,1.01) | 0.92 (0.85,0.99) | 0.93 (0.86,1.00) |
| Current symptoms of eczema | 1.08 (1.03,1.13) | 1.02 (0.95,1.10) | 1.05 (0.97,1.12) |
| Current symptoms of severe eczema | 0.88 (0.77,1.00) | 0.77 (0.64,0.92) | 0.79 (0.66,0.95) |
| Eczema ever | 1.09 (1.04,1.14) | 1.06 (1.01,1.12) | 1.05 (1.00,1.11) |
Adjusted for sex, region of the world, language, and gross national income. Including centres with at least 70% of data available for all covariates.
Multivariate analysis including centres with at least 70% data available for all covariates. Adjusted for sex, region of the world, language, gross national income, birth weight, maternal education, maternal smoking during the child's first year of life, presence of a cat or dog in the home during the first year of life, paracetamol use during first year of life, antibiotic use during the first year of life, older siblings, maternal exposure to farm animals during pregnancy and exposure to farm animals during the first year of life.
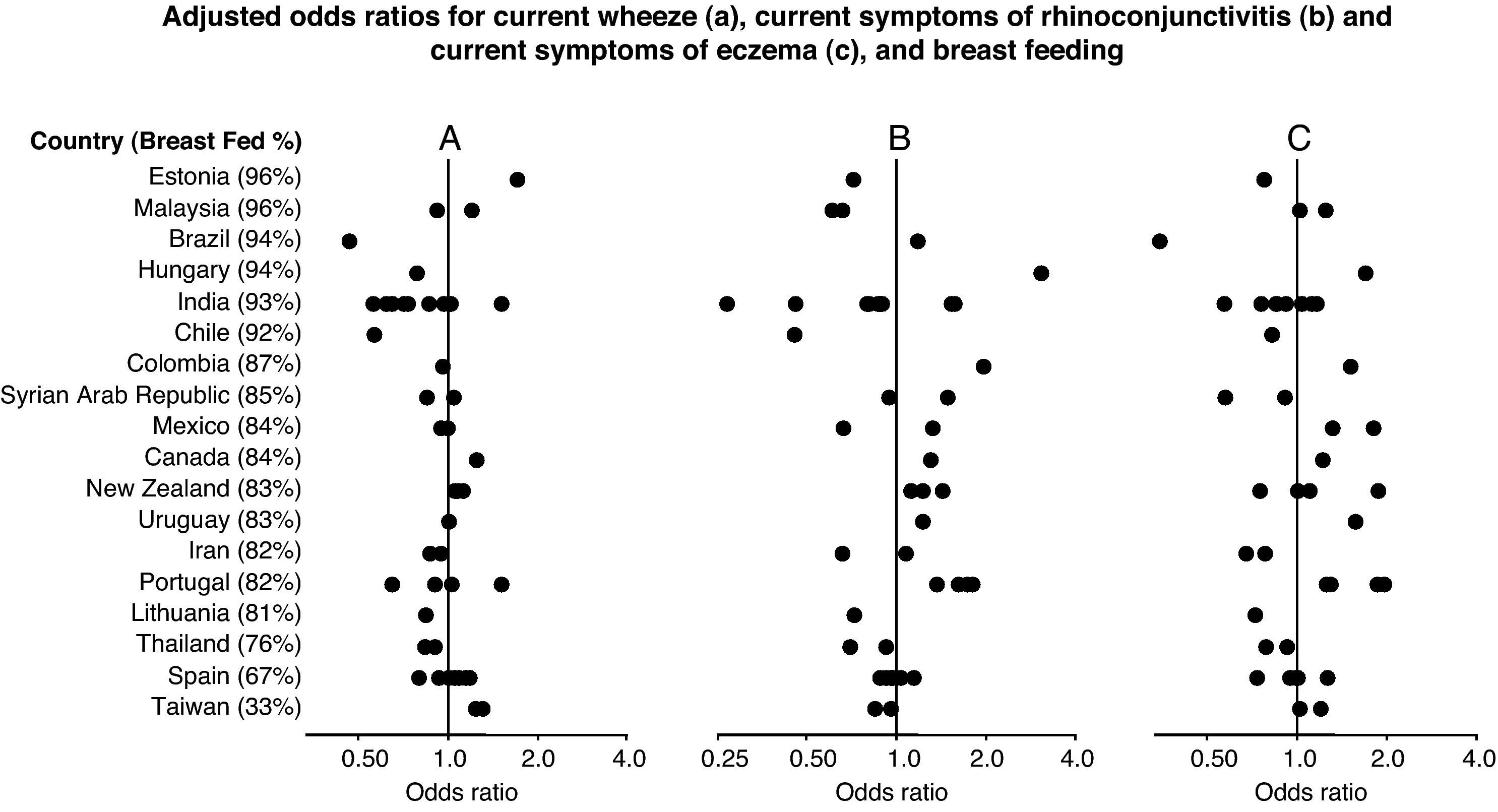
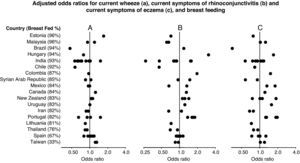
Forest plot showing the adjusted odds ratios (multiple regression analyses for children with complete covariate data) for the associations between breast feeding and current wheezing (A), rhinoconjunctivitis (B) and eczema (C) in 6–7 year old children from 45 centres in 18 countries. Each symbol represents a centre. For each country, the percentage of children who were breast fed is stated in brackets. Countries are ordered by average prevalence.
Breast feeding was associated with a slight reduction of severe wheeze (OR 0.91, 95% CI 0.86–0.98), severe rhinoconjunctivitis (OR 0.83, 95% CI 0.71–0.97) and severe eczema (OR 0.88, 95% CI 0.77–1.00), (Table 1 and Fig. 2). There was also a slightly reduced cumulative incidence of asthma ever (OR 0.92, 95% CI 0.88–0.97), but not of hay fever ever (OR 0.95, 95% CI 0.91–1.01) or eczema ever (OR 1.09, 95% CI 1.04–1.14).
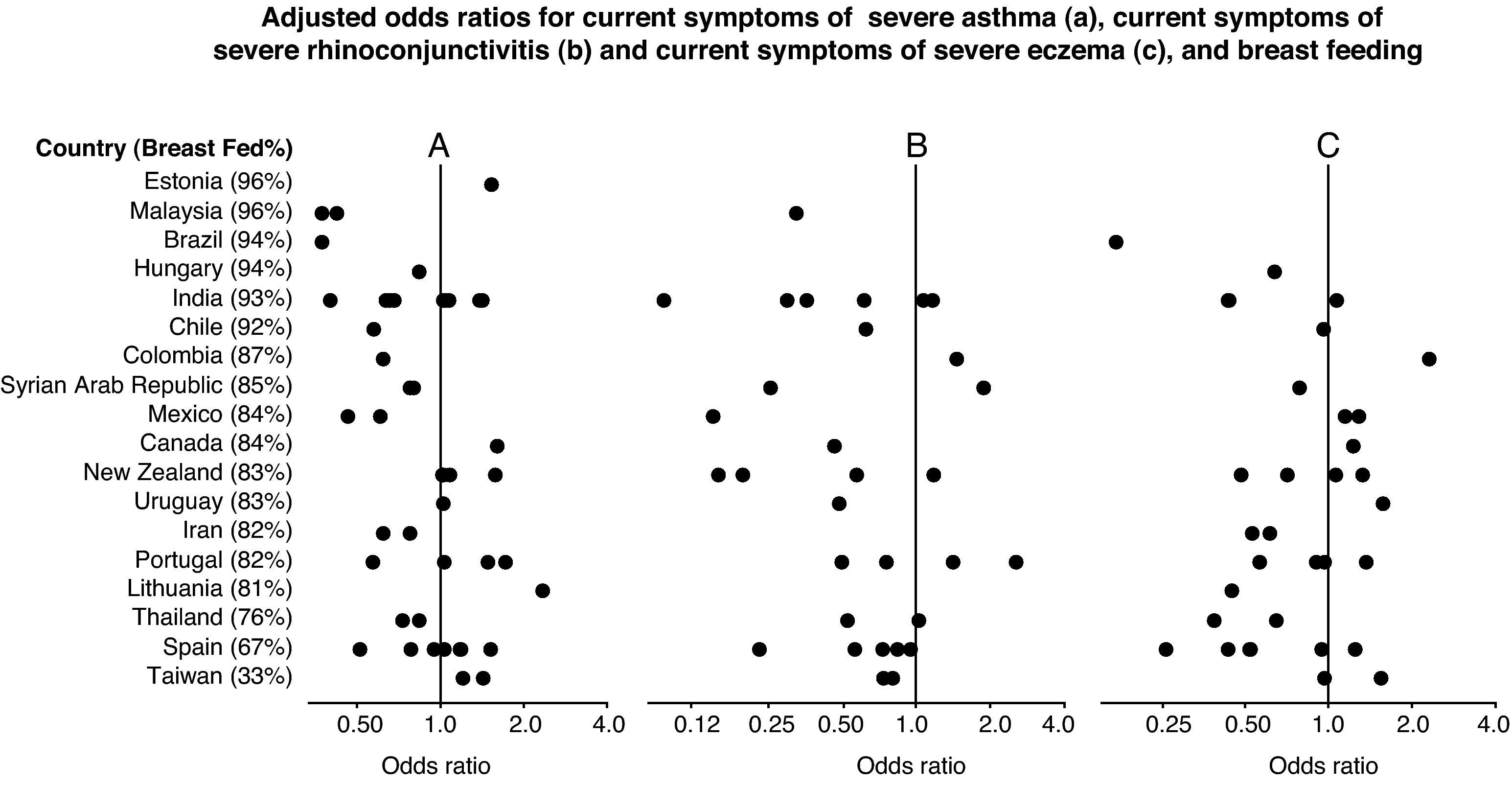
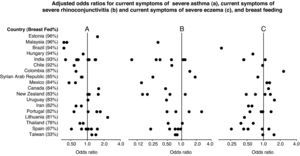
Forest plot showing the adjusted odds ratios (multiple regression analyses for children with complete covariate data) for the associations between breast feeding and current severe wheezing (A), rhinoconjunctivitis (B) and eczema (C) in 6–7 year old children from 45 centres in 18 countries. Each symbol represents a centre. For each country, the percentage of children who were breast fed is stated in brackets. Countries are ordered by average prevalence.
There were 103,716 children from 45 centres in 18 countries with complete covariate data enabling them to be included in the further multiple regression analyses. Adjustment for numerous potential asthma risk factors (gender, language, region, GNI category, birth weight, maternal education, maternal smoking in first year of life, the presence of cat or dog in the home in first year of life, use of paracetamol in first year of life, use of antibiotics in first year of life and the presence of older siblings) did not significantly alter the odds ratios.
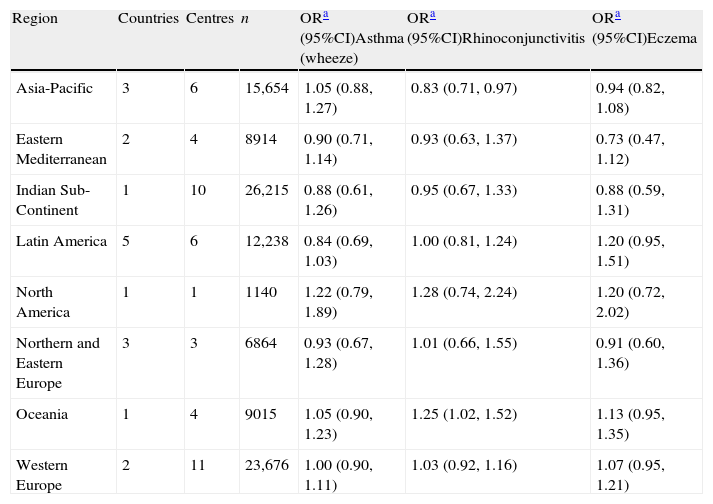
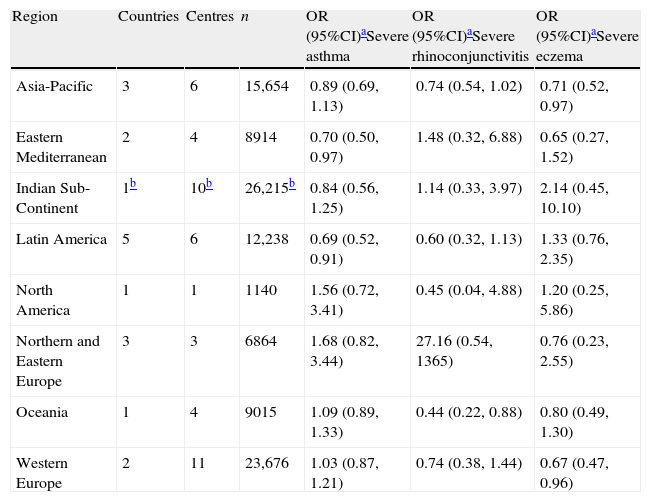
When all the regions of the world were examined after multivariate adjustment for breast feeding and current symptoms of all three conditions (Table 2), there were no relationships found apart from a significant association with reduced prevalence of rhinoconjunctivitis in Asia-Pacific. For severe symptoms (Table 3) breastfeeding was associated with reduced prevalence of severe asthma in Eastern Mediterranean and Latin America, severe rhinoconjunctivitis in Oceania and severe eczema in Asia-Pacific, and in Western Europe.
Associations between breast feeding and current symptoms of asthma, rhinoconjunctivitis, and eczema in children 6–7 years old in different regions of the world. Multiple regression analyses in children with complete covariate data.
| Region | Countries | Centres | n | ORa (95%CI)Asthma (wheeze) | ORa (95%CI)Rhinoconjunctivitis | ORa (95%CI)Eczema |
| Asia-Pacific | 3 | 6 | 15,654 | 1.05 (0.88,1.27) | 0.83 (0.71,0.97) | 0.94 (0.82,1.08) |
| Eastern Mediterranean | 2 | 4 | 8914 | 0.90 (0.71,1.14) | 0.93 (0.63,1.37) | 0.73 (0.47,1.12) |
| Indian Sub-Continent | 1 | 10 | 26,215 | 0.88 (0.61,1.26) | 0.95 (0.67,1.33) | 0.88 (0.59,1.31) |
| Latin America | 5 | 6 | 12,238 | 0.84 (0.69,1.03) | 1.00 (0.81,1.24) | 1.20 (0.95,1.51) |
| North America | 1 | 1 | 1140 | 1.22 (0.79,1.89) | 1.28 (0.74,2.24) | 1.20 (0.72,2.02) |
| Northern and Eastern Europe | 3 | 3 | 6864 | 0.93 (0.67,1.28) | 1.01 (0.66,1.55) | 0.91 (0.60,1.36) |
| Oceania | 1 | 4 | 9015 | 1.05 (0.90,1.23) | 1.25 (1.02,1.52) | 1.13 (0.95,1.35) |
| Western Europe | 2 | 11 | 23,676 | 1.00 (0.90,1.11) | 1.03 (0.92,1.16) | 1.07 (0.95,1.21) |
Multivariate analysis including centres with at least 70% data available for all covariates. Adjusted for sex, region of the world, language, gross national income, birth weight, maternal education, maternal smoking during the child's first year of life, presence of a cat or dog in the home during the first year of life, paracetamol use during first year of life, antibiotic use during the first year of life, older siblings, maternal exposure to farm animals during pregnancy and exposure to farm animals during the first year of life.
Associations between breast feeding and current symptoms of severe asthma, rhinoconjunctivitis, and eczema in children 6–7 years old in different regions of the world. Multiple regression analyses in children with complete covariate data.
| Region | Countries | Centres | n | OR (95%CI)aSevere asthma | OR (95%CI)aSevere rhinoconjunctivitis | OR (95%CI)aSevere eczema |
| Asia-Pacific | 3 | 6 | 15,654 | 0.89 (0.69,1.13) | 0.74 (0.54,1.02) | 0.71 (0.52,0.97) |
| Eastern Mediterranean | 2 | 4 | 8914 | 0.70 (0.50,0.97) | 1.48 (0.32,6.88) | 0.65 (0.27,1.52) |
| Indian Sub-Continent | 1b | 10b | 26,215b | 0.84 (0.56,1.25) | 1.14 (0.33,3.97) | 2.14 (0.45,10.10) |
| Latin America | 5 | 6 | 12,238 | 0.69 (0.52,0.91) | 0.60 (0.32,1.13) | 1.33 (0.76,2.35) |
| North America | 1 | 1 | 1140 | 1.56 (0.72,3.41) | 0.45 (0.04,4.88) | 1.20 (0.25,5.86) |
| Northern and Eastern Europe | 3 | 3 | 6864 | 1.68 (0.82,3.44) | 27.16 (0.54,1365) | 0.76 (0.23,2.55) |
| Oceania | 1 | 4 | 9015 | 1.09 (0.89,1.33) | 0.44 (0.22,0.88) | 0.80 (0.49,1.30) |
| Western Europe | 2 | 11 | 23,676 | 1.03 (0.87,1.21) | 0.74 (0.38,1.44) | 0.67 (0.47,0.96) |
Multivariate analysis including centres with at least 70% data available for all covariates. Adjusted for sex, region of the world, language, gross national income, birth weight, maternal education, maternal smoking during the child's first year of life, the presence of a cat or dog in the home during the first year of life, paracetamol use during first year of life, antibiotic use during the first year of life, older siblings, maternal exposure to farm animals during pregnancy and exposure to farm animals during the first year of life.
In this large global study breast feeding ever was not associated with parental-reported symptoms in 6–7 year old children of asthma, or rhinoconjunctivitis, but was associated with a slight increase in eczema. There was a reduced odds ratio for current symptoms of severe asthma, and asthma reported ever which disappeared after multivariate analysis with various risk factors. Breastfeeding was associated with reduced prevalence of current symptoms of severe rhinoconjunctivitis, however, both before and after multivariate analysis, and with current symptoms of severe eczema only after multivariate analysis. Within regions there were a small number of associations between breastfeeding and reduced symptom prevalence, but with no consistent picture.
Several studies have shown that breast feeding in developing countries is associated with protection against infections, particularly gastric infection and diarrhoea.5 The immunological properties of breast milk are significant contributing factors to the vital importance for infant health of breastfeeding in poor countries. Breastfeeding is therefore rightly promoted by authorities such as the World Health Organisation,18 but our results suggest that there is little evidence that breastfeeding protects against the development of allergy and related conditions.
The situation regarding a major role of breast feeding for infant health is less clear cut in affluent societies, with a higher hygienic standard. In these countries, there are reports of a reduced incidence of infections19,20 and consequently of a slightly reduced incidence of infant wheezing. With regard to allergy, the bulk of evidence indicates that breast feeding does not in a major way either decrease or increase either manifest disease or sensitisation (summarised in21,22), although there are several studies reporting both an increased23 and decreased risk.24
In the Phase Two of ISAAC, the possible relationship between breast feeding and wheezing, skin test sensitivity and other factors was addressed in some more detail.10 In that cross-sectional study data were obtained from school children in 27 centres in 20 countries. Any breast feeding was associated with less non-atopic wheeze in non-affluent countries. There was also a slightly higher predicted forced expiratory volume in breast-fed children living in affluent, but not in non-affluent countries. Breast feeding was not related to any measure of allergy, e.g. clinical symptoms of wheezing, eczema or rhinitis, nor to skin prick test results or circulating IgE antibodies.
In the present global analysis, we did not obtain objective markers of bronchial reactivity or allergic sensitisation and the question concerning breast feeding was simply a “yes” or “no”, with no information concerning the duration of any breast feeding. Despite these limitations, we believe that this global analysis lends further support to the lack of protective effects of breast feeding on allergy related symptoms beyond infancy. However, there is some evidence of protective associations between breast feeding and severe symptoms and these associations are stronger among non-affluent countries (data not shown).
There are several limitations of this study. The questionnaire data was completed by the parents of the 6–7 year old children, and was obtained retrospectively. This could potentially lead to recall bias, particularly if parents of asthmatic children were more likely to recall poor breast feeding in the first year of life than parents of non-asthmatic children. This is less likely to be of concern on a global scale, however, as a possible protective role of breast feeding has been publicly discussed in only a limited number of countries. It is conceivable that in some affluent countries the frequency could be higher in families with a family history of allergy. This would tend to neutralise a slight protective effect of breast feeding. In most countries, however, particularly in those with a low prevalence of allergies, this would not be the case. Finally, selection bias seems unlikely, as the average response rate from centres included in this analysis was 85%.
In summary, this global study has confirmed that any association is minor between a history of poor breast feeding and current symptoms of asthma, rhinoconjunctivitis and eczema in 6–7 year old children. Breast feeding is essential for health, particularly in infants living under poor hygienic conditions, but is not associated with any major increased or decreased risk for development of allergy related symptoms.
Conflict of interestThe authors have no conflict of interest to declare.
We are grateful to the children and parents who willingly cooperated and participated in ISAAC Phase Three and the coordination and assistance by the school staff is sincerely appreciated. We thank the Phase Three National Coordinators, Principal Investigators and their colleagues, who helped make ISAAC Phase Three such a success. We would like to acknowledge and thank the many funding bodies throughout the world that supported the individual ISAAC centres and collaborators and their meetings. In particular, we wish to thank the New Zealand funding bodies, the Health Research Council of New Zealand, the Asthma and Respiratory Foundation of New Zealand, the Child Health Research Foundation, the Hawke's Bay Medical Research Foundation, the Waikato Medical Research Foundation, Glaxo Wellcome New Zealand, the NZ Lottery Board and Astra Zeneca New Zealand. Glaxo Wellcome International Medical Affairs supported the Regional Coordination and the ISAAC International Data Centre. Without help from all of the above, ISAAC would not have given us all these results from so many countries. The Centre for Public Health Research is supported by a Programme Grant and Sunia Foliaki is supported by a Pacific Health Research Postdoctoral Fellowship from the Health Research Council of New Zealand.
ISAAC Phase Three Study Group
ISAAC Steering Committee: N Aït-Khaled* (International Union Against Tuberculosis and Lung Diseases, Paris, France); HR Anderson (Division of Community Health Sciences, St Georges, University of London, London, UK); MI Asher (Department of Paediatrics: Child and Youth Health, Faculty of Medical and Health Sciences, The University of Auckland, New Zealand); R Beasley* (Medical Research Institute of New Zealand, Wellington, New Zealand); B Björkstén* (Institute of Environmental Medicine, Karolinska Institutet, Stockholm, Sweden); B Brunekreef (Institute of Risk Assessment Science, Universiteit Utrecht, Netherlands); J Crane (Wellington Asthma Research Group, Wellington School of Medicine, New Zealand); P Ellwood (Department of Paediatrics: Child and Youth Health, Faculty of Medical and Health Sciences, The University of Auckland, New Zealand); C Flohr (Centre for Evidence Based Dermatology, Queen's Medical Centre, University Hospital, Nottingham, UK); S Foliaki* (Centre for Public Health Research, Massey University, Wellington, New Zealand); F Forastiere (Department of Epidemiology, Local Health authority Rome, Italy); L García-Marcos (Respiratory Medicine and Allergy Units, ‘Virgen de la Arrixaca’ University Children's Hospital, University of Murcia, Spain); U Keil* (Institut für Epidemiologie und Sozialmedizin, Universität Münster, Germany); CKW Lai* (Department of Medicine and Therapeutics, The Chinese University of Hong Kong, SAR China); J Mallol* (Department of Paediatric Respiratory Medicine, University of Santiago de Chile, Chile); EA Mitchell (Department of Paediatrics: Child and Youth Health, Faculty of Medical and Health Sciences, The University of Auckland, New Zealand); S Montefort* (Department of Medicine, University of Malta, Malta), J Odhiambo* (Centre Respiratory Diseases Research Unit, Kenya Medical Research Institute, Nairobi, Kenya); N Pearce (Centre for Public Health Research, Massey University, Wellington, New Zealand); CF Robertson (Murdoch Children's Research Institute, Melbourne, Australia); AW Stewart (Population Health, Faculty of Medical and Health Sciences, The University of Auckland, New Zealand); D Strachan (Division of Community Health Sciences, St Georges, University of London, London, UK); E von Mutius (Dr von Haunerschen Kinderklinik de Universität München, Germany); SK Weiland† (Department of Epidemiology, University of Ulm, Germany); G Weinmayr (Institute of Epidemiology, University of Ulm, Germany); H Williams (Centre for Evidence Based Dermatology, Queen's Medical Centre, University Hospital, Nottingham, UK); G Wong (Department of Paediatrics, Prince of Wales Hospital, Hong Kong, SAR China).
* Regional Coordinators; † Deceased.
ISAAC International Data Centre: MI Asher, TO Clayton, P Ellwood, EA Mitchell, Department of Paediatrics: Child and Youth Health, and AW Stewart, Population Health, Faculty of Medical and Health Sciences, The University of Auckland, New Zealand.
ISAAC Principal Investigators: Barbados: ME Howitt* (Barbados); Belgium: J Weyler (Antwerp); Brazil: L de Freitas Souza (Salvador); Canada: D Rennie (Saskatoon); Chile: P Aguilar (South Santiago); Colombia: G Aristizábal (Bogotá), AM Cepeda (Barranquilla); Estonia: M-A Riikjärv* (Tallinn); Hungary: G Zsigmond* (Svábhegy); India: S Awasthi (Lucknow), S Bhave (Rasta Peth), NM Hanumante (Pune), KC Jain (Jodhpur), MK Joshi (Mumbai (16)), SN Mantri (Mumbai (29)), AV Pherwani (Mumbai (18)), S Rego (Bangalore), S Salvi (Nagpur, Pimpri), SK Sharma (New Delhi (7)), V Singh (Jaipur), TU Sukumaran (Kottayam), PS Suresh Babu (Davangere); Indonesia: CB Kartasasmita (Bandung); Iran: M-R Masjedi* (Rasht, Tehran); Isle of Man: A Steriu (Isle of Man); Japan: H Odajima (Fukuoka); Kyrgyzstan: C Imanalieva* (Bishkek); Lithuania: J Kudzyte* (Kaunas); Malaysia: BS Quah (Kota Bharu), KH Teh (Alor Setar); Malta: S Montefort* (Malta); Mexico: M Baeza-Bacab* (Mérida), M Barragán-Meijueiro (Ciudad de México (3)), BE Del-Río-Navarro (Ciudad de México (1)), R García-Almaráz (Ciudad Victoria), SN González-Díaz (Monterrey), FJ Linares-Zapién (Toluca), JV Merida-Palacio (Mexicali Valley), S Romero-Tapia (Villahermosa), I Romieu (Cuernavaca); New Zealand: MI Asher* (Auckland), R MacKay (Nelson), C Moyes (Bay of Plenty), P Pattemore (Christchurch); Nigeria: BO Onadeko (Ibadan); Panama: G Cukier* (David-Panamá); Poland: A Brêborowicz (Poznan), G Lis* (Kraków); Portugal: R Câmara (Funchal), JM Lopes dos Santos (Porto), C Nunes (Portimao), JE Rosado Pinto* (Lisbon); Singapore: DYT Goh (Singapore); South Korea: H-B Lee* (Provincial Korea, Seoul); Spain: RM Busquets (Barcelona), I Carvajal-Urueña (Asturias), G García-Hernández (Madrid), L García-Marcos* (Cartagena), C González Díaz (Bilbao), A López-Silvarrey Varela (A Coruña), MM Morales-Suárez-Varela (Valencia); Sultanate of Oman: O Al-Rawas* (Al-Khod); Syrian Arab Republic: S Mohammad* (Tartous), Y Mohammad (Lattakia); Taiwan: J-L Huang* (Taipei), C-C Kao (Taoyuan); Thailand: M Trakultivakorn (Chiang Mai), P Vichyanond* (Bangkok); Uruguay: MC Lapides (Paysandú).
* National Coordinator
ISAAC Phase Three National Coordinators not identified above: Canada: M Sears; Chile: V Aguirre; India: J Shah; Indonesia: K Baratawidjaja; Isle of Man: HR Anderson; Singapore: B-W Lee.