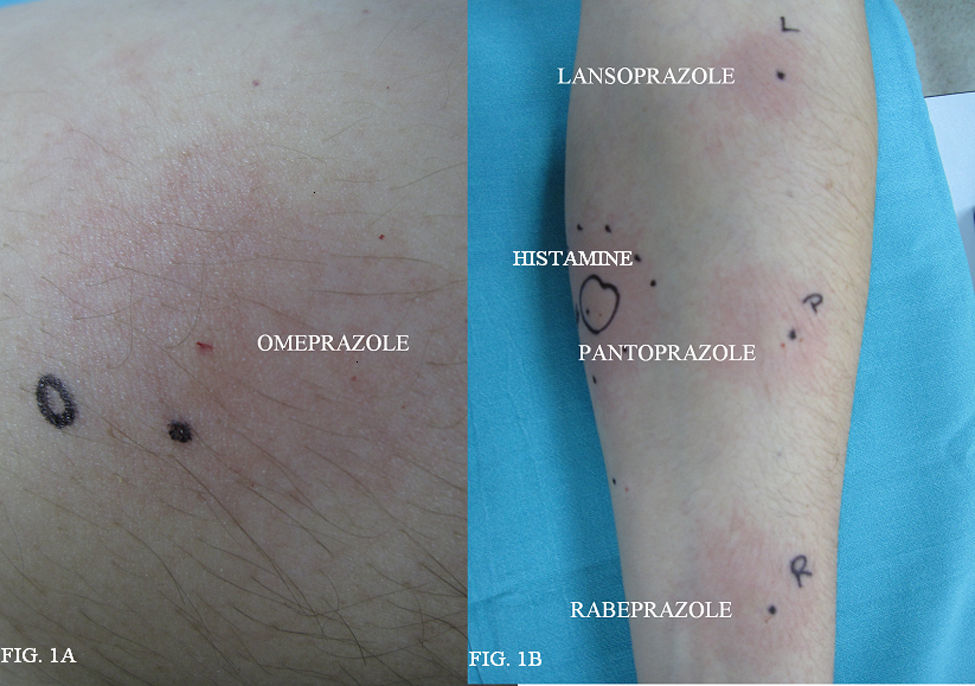
A 42-year-old non-atopic woman was referred to our Unit for evaluation. Thirty minutes after omeprazole oral intake, the patient experienced nausea, vomits, swelling of her hands, severe shortness of breath, and dysphagia. She received immediate intravenous treatment with methylprednisolone and dexchlorpheniramine, and her symptoms resolved in less than 6h. She had tolerated previous treatments with omeprazole but not with other PIPs. Skin prick and intradermal tests were performed: omeprazole was used in skin prick test at a concentration of 40mg in 1ml of saline solution.
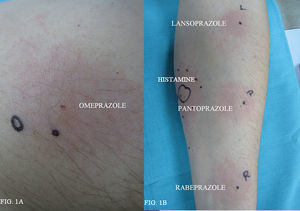
To investigate the cross-reaction patterns, a study was carried out with other PIPs. We performed skin prick tests with lansoprazole (15mg/ml) rabeprazole (20mg/ml) and pantoprazole (20mg/ml).
Intradermal skin tests were carried out with 1:100, 1:10 dilutions. Lansoprazole (0.15mg/ml and 15mg/ml), rabeprazole (0.2mg/ml and 2mg/ml), and pantoprazole (0.2mg/ml and 2mg/ml). Total serum was taken from the patient and Basophil activation test (BAT) was performed. The percentage of activated basophils which expressed the CD 63 marker after in vitro stimulation by two PIPs, omeprazole and lansoprazole, was recorded. Skin prick test with omeprazole (40mg/ml) was positive (the patient presented a wheal of 5×3.5mm) (Figure 1A). Prick tests with lansoprazole, rabeprazole and pantoprazole were negative. Intradermal tests with lansoprazole, pantoprazole, rabeprazole were positive in both concentrations tested (Figure 1B). Ten normal controls (five atopic subjects and five non-atopic subjects) were performed and were negative for prick test to omeprazole, lansoprazole, pantoprazole, rabeprazole. The BAT was performed in five healthy controls, tolerating omeprazole and pantoprazole with negative results in all cases. The patient presented a positive response to omeprazole and negative to pantoprazole in the BAT.
Previous reports described different patterns of cross-reactivity between PIPs, confirmed by skin and/or oral challenge tests.1 Different studies described cross-reactivity between omeprazole and lansoprazole2,3 and between rabeprazole and lansoprazole4 confirmed by skin tests. Lobera et al.5 reported nine patients with allergy to omeprazole and tolerance to lansoprazole in all the cases. We present a case of anaphylaxis to omeprazole, demostrated by prick test and with the flow-cytometric cellular allergen stimulation test, which suggests an IgE mediated mechanism.
Skin tests also prove the cross-reactivity with all the other PIPs tested. In accordance with other reports, we emphasize the importance of skin prick test in the diagnosis of PIPs allergy, and its usefulness to establish cross-reactivity between them. The usefulness of new diagnostic in vitro tests like the BAT is also seen.6
In our case, skin test proves cross-reactivity between all the PIPs, so due to these results, an oral challenge test with the other PIPs was not performed, and it was recommended that the use of PIPs drugs be avoided in this patient.
This work was supported by grants from Spanish Health Ministry (FIS) network RIRAAF (RD 07/0064).